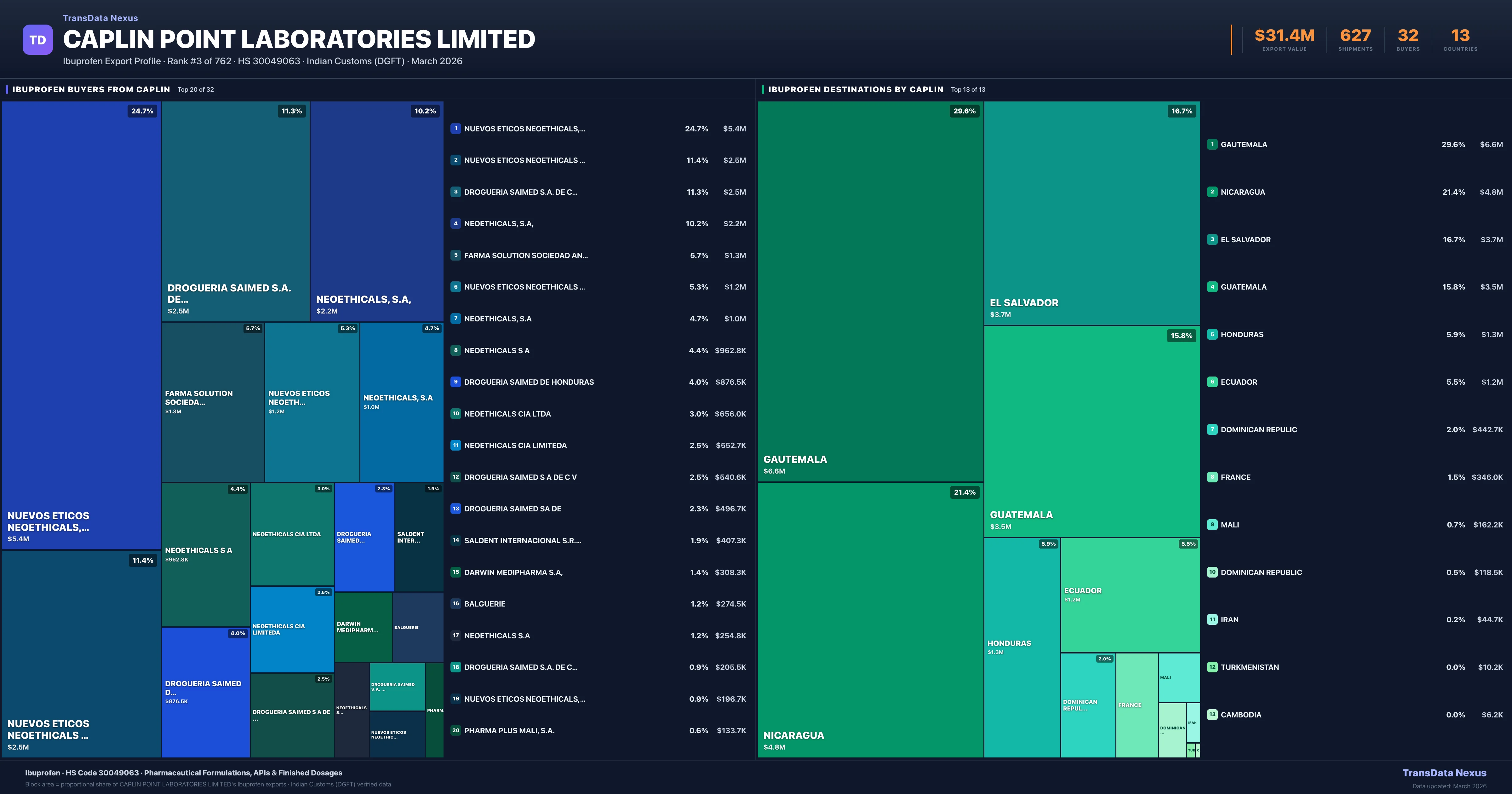
Geopolitical & Trade Policy Impact on Caplin Point Laboratories Limited's Ibuprofen Exports
The global pharmaceutical export landscape is currently navigating several geopolitical challenges that directly impact companies like Caplin Point.
The Israel-Iran conflict has led to disruptions in Red Sea shipping routes, causing maritime carriers to suspend bookings and reroute vessels around the Cape of Good Hope. This has resulted in elongated lead times and increased shipping costs, disproportionately affecting time-sensitive pharmaceutical shipments. Freight charges have surged, with transport costs doubling and additional surcharges ranging from $4,000 to $8,000 per shipment. Such disruptions pose significant risks to the timely delivery of pharmaceuticals, especially those requiring stringent cold-chain logistics. (pharmaceuticalcommerce.com)
In the United States, recent tariff implementations have introduced further complexities. The U.S. administration has imposed broad tariffs on imports from over 60 countries, including India, with rates reaching up to 39%. These tariffs affect a substantial portion of Indian exports to the U.S., including pharmaceuticals, potentially leading to increased costs and competitive disadvantages for Indian exporters. (apnews.com)
The European Union's regulatory landscape is also evolving, with stricter compliance requirements under directives like the Falsified Medicines Directive. Indian pharmaceutical companies must invest in advanced serialization and traceability systems to meet these standards, ensuring continued access to the lucrative EU market.
For Caplin Point, these geopolitical and regulatory developments necessitate strategic adaptations. Diversifying export markets, enhancing supply chain resilience, and investing in compliance infrastructure are critical to mitigating risks and sustaining growth in this volatile environment.
Caplin Point Laboratories Limited — Regulatory Compliance & Quality Standards
The global pharmaceutical industry is witnessing heightened regulatory scrutiny, particularly from agencies like the U.S. FDA and the European Medicines Agency. Compliance with Good Manufacturing Practices (GMP) and other quality standards is imperative for market access.
Caplin Point has proactively addressed these requirements. The company's subsidiary, Caplin Steriles Limited (CSL), has secured an EU GMP renewal and successfully passed audits by regulatory bodies such as the Saudi FDA. These achievements underscore Caplin Point's commitment to maintaining high-quality standards and ensuring compliance with international regulations. (whalesbook.com)
However, the broader Indian pharmaceutical sector faces challenges related to quality and compliance. The U.S. FDA conducted over 200 inspections in India in 2023, with plans to increase this number in subsequent years. Instances of substandard products have led to increased scrutiny, emphasizing the need for continuous quality improvements. (fact.net.in)
For Caplin Point, ongoing investment in quality assurance systems, employee training, and infrastructure upgrades is essential to navigate the evolving regulatory landscape and maintain its competitive edge.