Jubilant Generics Limited — Esomeprazole Exporter Profile
Indian Pharmaceutical Exporter · #10 for Esomeprazole · $5.2M export value · DGFT Verified
Jubilant Generics Limited is the #10 Indian exporter of Esomeprazole with $5.2M in export value and 179 verified shipments. Jubilant Generics Limited holds a 1.1% market share in Esomeprazole exports across 10 countries. The company exports 6 pharmaceutical products worth $23.9M across 4 therapeutic categories.
Jubilant Generics Limited — Esomeprazole Export Profile: Buyers & Destinations
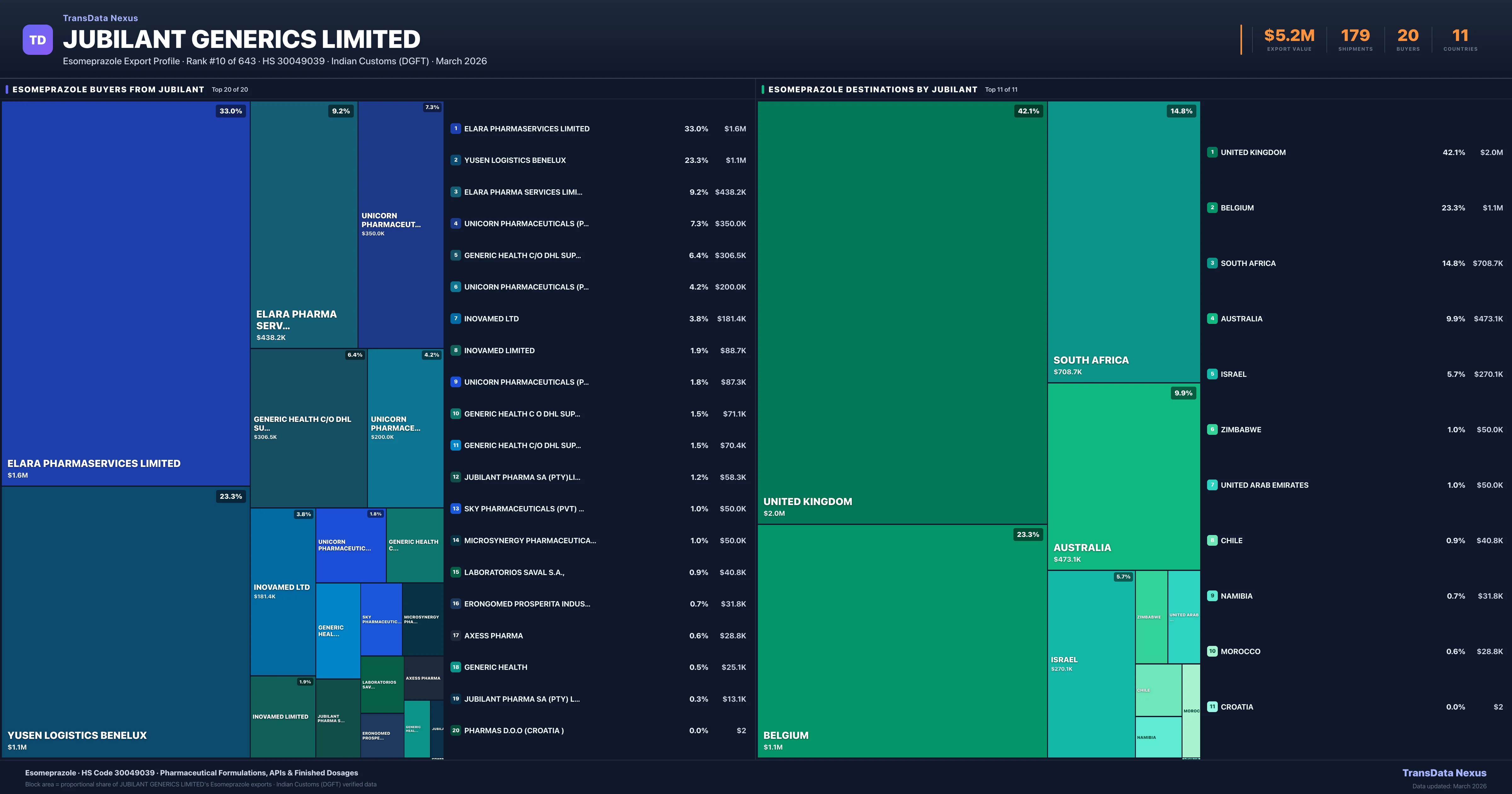
Where Does Jubilant Generics Limited Export Esomeprazole?
| Country | Value | Shipments | Share |
|---|---|---|---|
| UNITED KINGDOM | $2.0M | 56 | 42.1% |
| BELGIUM | $1.1M | 67 | 23.3% |
| SOUTH AFRICA | $708.7K | 16 | 14.8% |
| AUSTRALIA | $473.1K | 21 | 9.9% |
| ISRAEL | $270.1K | 9 | 5.7% |
| ZIMBABWE | $50.0K | 1 | 1.0% |
| UNITED ARAB EMIRATES | $50.0K | 1 | 1.0% |
| CHILE | $40.8K | 1 | 0.9% |
| NAMIBIA | $31.8K | 2 | 0.7% |
| MOROCCO | $28.8K | 5 | 0.6% |
Jubilant Generics Limited exports Esomeprazole to 11 countries. The largest destination is UNITED KINGDOM accounting for 42.1% of Jubilant Generics Limited's Esomeprazole shipments, followed by BELGIUM (23.3%) and SOUTH AFRICA (14.8%). These destinations reflect Jubilant Generics Limited's established distribution network and regulatory approvals in key markets.
Who Are the Top Buyers of Esomeprazole from Jubilant Generics Limited?
| Buyer | Country | Value | Orders |
|---|---|---|---|
| ELARA PHARMASERVICES LIMITED | UNITED KINGDOM | $1.6M | 43 |
| YUSEN LOGISTICS BENELUX | BELGIUM | $1.1M | 67 |
| ELARA PHARMA SERVICES LIMITED | UNITED KINGDOM | $438.2K | 13 |
| UNICORN PHARMACEUTICALS (PTY)LTD | SOUTH AFRICA | $350.0K | 7 |
| GENERIC HEALTH C/O DHL SUPPLY CHAIN | AUSTRALIA | $306.5K | 16 |
| UNICORN PHARMACEUTICALS (PTY) LTD | SOUTH AFRICA | $200.0K | 4 |
| INOVAMED LTD | ISRAEL | $181.4K | 6 |
| INOVAMED LIMITED | ISRAEL | $88.7K | 3 |
| UNICORN PHARMACEUTICALS (PTY) LTD., | SOUTH AFRICA | $87.3K | 2 |
| GENERIC HEALTH C O DHL SUPPLY CHAIN | AUSTRALIA | $71.1K | 2 |
Jubilant Generics Limited supplies Esomeprazole to 20 buyers globally. The largest buyer is ELARA PHARMASERVICES LIMITED (UNITED KINGDOM), followed by YUSEN LOGISTICS BENELUX (BELGIUM) and ELARA PHARMA SERVICES LIMITED (UNITED KINGDOM). These buyers represent pharmaceutical distributors, hospital groups, and government procurement agencies in their respective markets.
What Is India's Total Esomeprazole Export Value and How Much Does Jubilant Generics Limited Contribute?
India exported $207.7M worth of Esomeprazole through 9,532 shipments from 643 suppliers to 147 countries, serving 1,634 buyers globally. Jubilant Generics Limited contributes $5.2M to this total, accounting for 1.1% of India's Esomeprazole exports. Jubilant Generics Limited ships Esomeprazole to 11 countries through 20 buyers.
What Is the Average Shipment Value for Jubilant Generics Limited's Esomeprazole Exports?
Jubilant Generics Limited's average Esomeprazole shipment value is $28.9K per consignment, based on 179 shipments totaling $5.2M. The largest destination is UNITED KINGDOM (42.1% of Jubilant Generics Limited's Esomeprazole exports).
How Does Jubilant Generics Limited Compare to Other Indian Esomeprazole Exporters?
Jubilant Generics Limited ranks #10 among 643 Indian Esomeprazole exporters with a 1.1% market share. The top 3 exporters are SUN PHARMACEUTICAL INDUSTRIES LIMITED ($82.5M), CIPLA LIMITED ($30.6M), MICRO LABS LIMITED ($20.4M). Jubilant Generics Limited processed 179 shipments to 10 destination countries.
What Esomeprazole Formulations Does Jubilant Generics Limited Export?
| Product Description / Formulation | Value | Shipments |
|---|---|---|
| ESOMEPRAZOLE 20MG GRT4X7 JUB UK ELARA, B | $550.0K | 11 |
| ESOMEPRAZOLE 20 MG GRT 4X7'S CRESCENT- U | $376.4K | 13 |
| ESOMEPRAZOLE 40 MG GRT 4X7'S CRESCENT- U | $296.0K | 13 |
| ESOMEPRAZOLE 40MG GRT4X7 JUB UK ELARA, B | $178.7K | 6 |
| ESOMEPRAZOLE 20MG GRT4X7 JUB UK ELARA,B. | $150.0K | 3 |
| ESOMEPRAZOLE GR TAB 40MG 4X7 UNICORN ZAF(ESOMEPRAZOLE ENTEIC-COATED TABLETS 40MG) B/N:EY225021A MD:02/25 ED:01/27 | $150.0K | 3 |
| ESOMEPRAZOLE GR TAB 20MG 4X7 UNICORN ZAF(ESOMEPRAZOLE ENTEIC-COATED TABLETS 20MG) B/N:EY125017A MD:02/25 ED:01/27 | $150.0K | 3 |
| ESOMEPRAZOLE GASTRO-RESISTANT TABLETS 20MG, BATCH NO. EY124019A, MFG. DT. 01.03.2024, EXP. DT. 28.02.2026 | $101.4K | 3 |
| ESOMEPRAZOLE 40MG GRT4X7 JUB UK ELARA ,B | $101.3K | 3 |
| ESOMEPRAZOLE GASTRO-RESISTANT TABLETS 20MG, BATCH NO. EY124023A, MFG. DT. 01.03.2024, EXP. DT. 28.02.2026 | $101.0K | 3 |
Jubilant Generics Limited exports 111 distinct Esomeprazole formulations including tablets, capsules, syrups, and combination drugs. The top formulation is ESOMEPRAZOLE 20MG GRT4X7 JUB UK ELARA, B with 11 shipments worth $550.0K.
Regulatory Requirements: Exporting Esomeprazole to Key Markets
What Jubilant Generics Limited must comply with to export Esomeprazole to its top destination countries
United Kingdom — MHRA
Approval Process
Post-Brexit UK Marketing Authorisation via national procedure. Separate from EU approval since January 2021.
Timeline: 12–15 months
GMP & Export Requirements
UK GMP compliance; MHRA inspection of manufacturing site
Manufacturer Import Authorisation (MIA) held by UK importer; Qualified Person (QP) batch release
Note: Post-Brexit: EU GMP certificates no longer automatically valid in UK. Separate MHRA recognition required.
South Africa — SAHPRA
Approval Process
Registration under SAHPRA guidelines (CTD format). Bioequivalence required for generics.
Timeline: 24–48 months (significant backlog)
GMP & Export Requirements
WHO GMP or PIC/S GMP compliance
Certificate of Pharmaceutical Product from CDSCO; Import permit per Section 22A of Medicines Act
Note: Known registration backlog. WHO Prequalification significantly assists the process.
Regulatory information is provided for general reference based on publicly available guidelines. Requirements vary by formulation, dosage form, and therapeutic classification. Exporters should consult qualified regulatory affairs professionals for case-specific guidance.
How Does Jubilant Generics Limited Compare to Nearest Esomeprazole Exporters?
Exporters ranked immediately above and below #10 by export value
| # | Supplier | Export Value | Shipments | Countries | Avg/Shipment |
|---|---|---|---|---|---|
| 3 | MYLAN LABORATORIES LIMITED | $8.9M | 179 | 6 | $50.0K |
| 9 | ANNORA PHARMA PRIVATE LIMITED | $5.7M | 155 | 1 | $36.6K |
| 10 | JUBILANT GENERICS LIMITED ★ | $5.2M | 179 | 10 | $28.9K |
| 13 | SUN PHARMACEUTICAL INDUSTRIES LTD | $3.1M | 63 | 8 | $50.0K |
| 14 | PELL TECH HEALTH CARE PRIVATE LIMITED | $3.0M | 72 | 5 | $42.3K |
Jubilant Generics Limited ranks #10 among 643 Indian Esomeprazole exporters. Average shipment value of $28.9K compared to the market average of $323.1K. The closest competitors by value are MYLAN LABORATORIES LIMITED and ANNORA PHARMA PRIVATE LIMITED.
Which Indian Ports Ship Esomeprazole Exports?
| Port | Shipments | % Share |
|---|---|---|
| NHAVA SHEVA SEA (INNSA1) | 829 | 8.7% |
| SAHAR AIR | 824 | 8.6% |
| SAHAR AIR CARGO ACC (INBOM4) | 707 | 7.4% |
| DELHI AIR CARGO ACC (INDEL4) | 630 | 6.6% |
| DELHI AIR | 522 | 5.5% |
| JNPT/ NHAVA SHEVA SEA | 393 | 4.1% |
| JNPT | 361 | 3.8% |
| CMA CGM LOGISTICS PARK ICD | 305 | 3.2% |
Geopolitical & Trade Policy Impact on Jubilant Generics Limited's Esomeprazole Exports
The current geopolitical landscape presents both challenges and opportunities for Indian pharmaceutical exporters like Jubilant Generics. The Israel-Iran conflict has led to disruptions in Red Sea shipping routes, causing increased freight charges and potential losses estimated between ₹2,500 crore to ₹5,000 crore in March 2026. These disruptions have particularly impacted exports to the Middle East, a key market for Indian pharmaceuticals. (fortuneindia.com)
Conversely, U.S.-China trade tensions have opened avenues for Indian exporters to capture a larger share of the U.S. market. However, recent U.S. tariffs on imports, including pharmaceuticals, pose a risk to this opportunity. The Federation of Indian Export Organisations highlighted that these tariffs could affect nearly 55% of India's exports to the U.S., potentially leading to the loss of longstanding clients. (apnews.com)
In the European Union, compliance with the Falsified Medicines Directive (FMD) remains a critical requirement. Indian exporters must invest in serialization and traceability systems to meet these stringent regulations, ensuring continued access to the EU market.
Jubilant Generics Limited — Regulatory Compliance & Quality Standards
Maintaining compliance with international regulatory standards is paramount for Jubilant Generics. The company's Roorkee facility's VAI classification by the USFDA in 2024 reflects its adherence to current good manufacturing practices. (business-standard.com) However, the Indian pharmaceutical industry faces ongoing scrutiny regarding quality and compliance. In 2023, the USFDA conducted over 200 inspections in India, with plans to increase this number in 2024, underscoring the need for continuous quality assurance. (fact.net.in)
Additionally, the company has faced legal challenges concerning intellectual property. In March 2025, a court in Uttar Pradesh restrained Jamp Pharma from using Jubilant Generics' confidential data, granting an injunction against copyright infringement. (scconline.com) This case highlights the importance of safeguarding proprietary information in the competitive pharmaceutical landscape.
About Jubilant Generics Limited
Jubilant Generics Limited exports 6 products worth $23.9M. Beyond Esomeprazole, top products include Azithromycin, Atorvastatin, Valsartan, Irbesartan, Valacyclovir. View the complete Jubilant Generics Limited profile for full portfolio analysis.
Market Intelligence
Latest developments and market context
Detailed market intelligence for Esomeprazole — including regulatory updates, trade policy changes, and competitive landscape analysis — will be available in an upcoming update. This section will feature AI-researched insights from verified industry sources, government notifications, and market reports.
Official References & Regulatory Resources
- CDSCO India
- Pharmexcil
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- India Trade Statistics (DGFT)
Verify manufacturer licensing and export certifications with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Product Matching: Esomeprazole shipments identified using fuzzy matching on DGFT product description fields, validated against Indian Pharmacopoeia nomenclature.
- 2.Supplier Identification: Jubilant Generics Limited matched across shipments using name normalization, accounting for company name formatting variations.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 4.Verified Records: This page is backed by 182 individual customs records matching Jubilant Generics Limited exporting Esomeprazole, covering 111 formulations to 11 countries via 20 buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
Global Coverage
Exports to 147+ countries, 1,634+ buyers
Expert-Reviewed
Analyzed by pharmaceutical trade specialists
Get Esomeprazole Export Data from Jubilant Generics Limited
Access shipment-level details, pricing data, buyer connections, and competitive analysis for Jubilant Generics Limited's Esomeprazole exports. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Jubilant Generics Limited
Full Company Profile →
6 products · $23.9M total trade · 4 categories
Esomeprazole Stats
Company Overview
Top Products by Jubilant Generics Limited
Related Analysis
Need Detailed Export Data?
Access shipment-level records, pricing trends, and buyer contacts for Jubilant Generics Limited.
Request DemoView Pricing →Data Source
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports for Esomeprazole. For current shipment-level data, contact TransData Nexus.