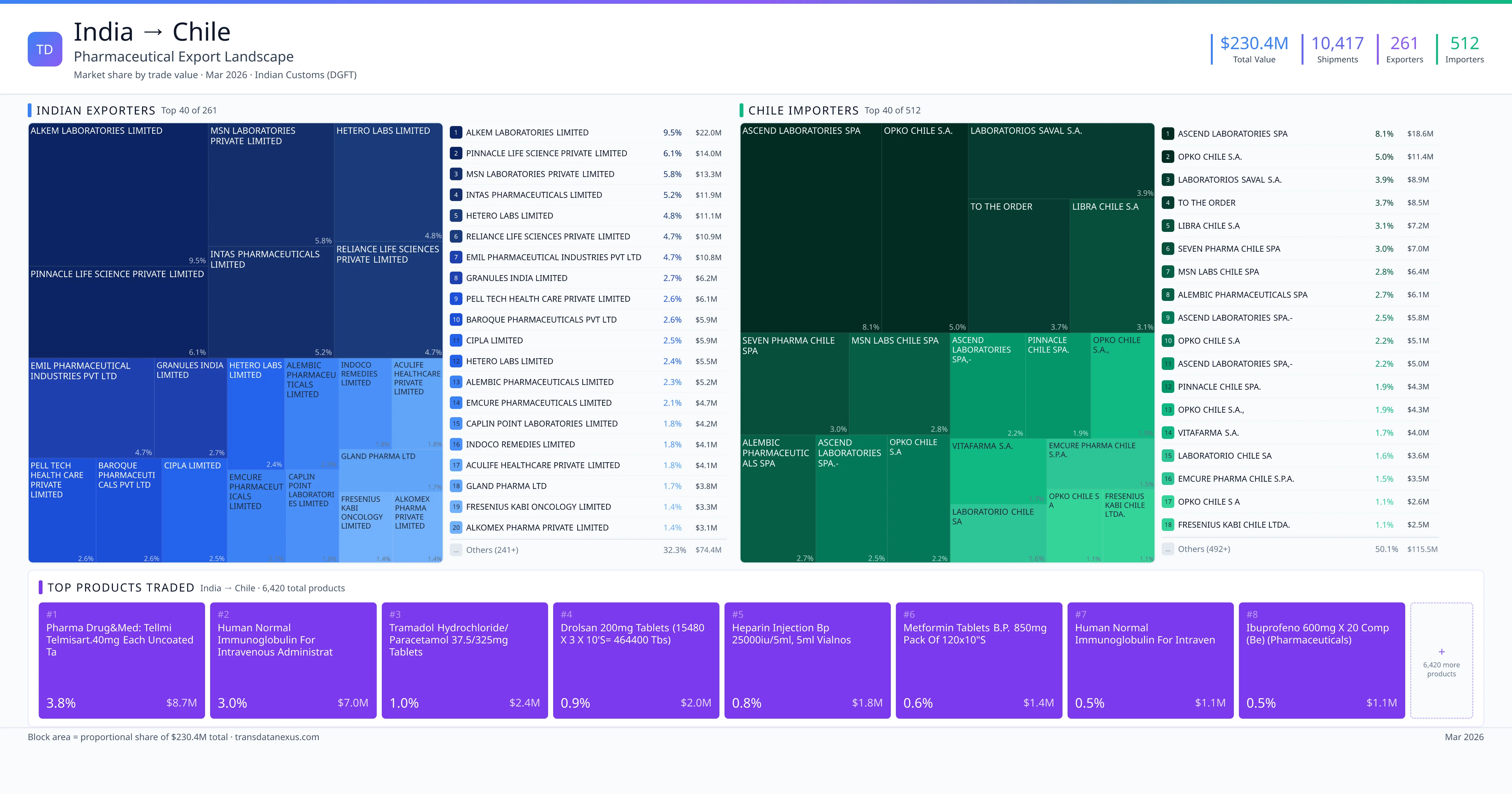
Chile to India Pharmaceutical Import
Bilateral Trade Intelligence · $60.1K Total Trade · 8 Foreign Suppliers · 6 Indian Buyers · DGFT Verified · Updated March 2026
India imported $60.1K worth of pharmaceutical formulations from Chile across 34 verified shipments, from 8 foreign suppliers to 6 Indian buyers. The top suppliers are PINNACLE CHILE SPA. ($58.8K) and SYNTHON CHILE LTDA ($391). The leading products are Paracetamol ($59K) and Lenalidomide ($391). Average shipment value: $1.8K.
Top Pharmaceutical Formulations — Chile to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Paracetamol | $58.8K | 97.9% |
| 2 | Lenalidomide | $391 | 0.7% |
| 3 | Empagliflozin | $285 | 0.5% |
| 4 | Amlodipine | $200 | 0.3% |
| 5 | Valsartan | $200 | 0.3% |
| 6 | Hydrochlorothiazide | $64 | 0.1% |
| 7 | Montelukast | $55 | 0.1% |
| 8 | Sodium | $55 | 0.1% |
| 9 | Metformin | $20 | 0.0% |
India imports 9+ pharmaceutical formulations from Chile with a combined trade value of $60.1K. Key products include Paracetamol ($59K), Lenalidomide ($391), Empagliflozin ($285), Amlodipine ($200), Valsartan ($200). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 34 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — Chile to India Trade Routes
These are the top pharmaceutical products exported from India to Chile, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Chile buyers, regulatory requirements, and logistics for that specific product corridor. Products include Paracetamol ($59K), Lenalidomide ($391), Empagliflozin ($285), Amlodipine ($200), Valsartan ($200) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Chile Suppliers to India
8 Indian pharmaceutical companies export finished formulations to Chile. Leading exporters include Pinnacle Chile Spa., Synthon Chile Ltda, Synthon Chile, Laboratorio Saval S.a /int. The top exporter accounts for 97.9% of total India–Chile pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from Chile
6 companies in Chile import pharmaceutical formulations from India. Top buyers include Pinnacle Life Science Private Limited, Veeda Clinical Research Limited, Azidus Laboratories Limited, Lambda Therapeutic Research Limited. The largest buyer accounts for 97.9% of India–Chile pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
India–Chile Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade corridor between Chile and India has experienced modest activity from 2022 to 2026. Over this period, India imported a total of $60,000 USD worth of finished pharmaceutical formulations from Chile, distributed across 34 shipments. This equates to an average shipment value of approximately $2,000 USD. The trade involved eight Chilean suppliers and six Indian buyers, indicating a niche yet consistent exchange in the pharmaceutical sector. The predominant product in this trade was paracetamol, accounting for 97.9% of the total import value. Other imported products included lenalidomide, empagliflozin, and amlodipine, among others. The primary Chilean supplier was Pinnacle Chile SPA, which supplied paracetamol in two shipments valued at $59,000 USD. On the Indian side, Pinnacle Life Science Private Limited was the main buyer, importing paracetamol from Chile. This trade corridor, while limited in volume, highlights the specialized nature of pharmaceutical imports from Chile to India.
2Chile's Role in India's Pharma Imports
Chile's contribution to India's pharmaceutical imports is relatively minor. In 2024, India imported pharmaceutical products worth $2.8 billion USD, with imports from Chile amounting to $60,000 USD, representing a fraction of a percent of the total. The imports from Chile were predominantly finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The focus on paracetamol suggests that Chile's pharmaceutical exports to India are concentrated in specific therapeutic areas, catering to particular market needs.
3Recent Developments
Between 2024 and 2026, there have been no significant policy changes or trade agreements specifically affecting the pharmaceutical import relationship between Chile and India. The Central Drugs Standard Control Organisation (CDSCO) in India has maintained its existing regulatory framework for foreign pharmaceutical imports, ensuring consistent standards for product safety and efficacy. Similarly, Chile's National Drug Regulatory Authority continues to uphold its WHO-GMP standards for pharmaceutical exports. The lack of major developments indicates a stable, albeit limited, trade relationship in the pharmaceutical sector between the two nations.
Chile Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1CDSCO Registration Process
For Chilean pharmaceutical companies aiming to export finished formulations to India, obtaining registration with the Central Drugs Standard Control Organisation (CDSCO) is mandatory. The process involves several key steps:
1. Application Submission: Companies must apply through the CDSCO's online portal, providing detailed information about the applicant and the products intended for import.
2. Documentary Requirements: Essential documents include:
3. Regulatory Review: CDSCO evaluates the application to ensure compliance with Indian standards. This may involve site inspections and assessments of product safety and efficacy.
4. Approval and Registration: Upon successful evaluation, CDSCO grants approval, allowing the company to export pharmaceutical products to India.
The entire process can take several months, depending on the completeness of the application and the regulatory review timeline. It's crucial for Chilean companies to ensure all documentation is accurate and comprehensive to facilitate a smooth registration process.
2GMP & Manufacturing Standards
India requires that all imported pharmaceutical products adhere to Good Manufacturing Practice (GMP) standards. Chilean manufacturing sites must comply with these standards, which are in line with the World Health Organization's (WHO) GMP guidelines. To demonstrate compliance, Chilean manufacturers should obtain WHO prequalification for their products. Additionally, CDSCO may conduct inspections of foreign manufacturing facilities to verify adherence to GMP standards. Maintaining high-quality manufacturing practices is essential for Chilean companies to ensure their products meet the stringent requirements of the Indian market.
3Import Documentation
Chilean pharmaceutical companies exporting to India must prepare and submit several key documents to CDSCO:
- Import License (Form 10): This license authorizes the import of pharmaceutical products into India.
- Registration Certificate: Issued by CDSCO, this certificate confirms that the imported products are approved for sale and distribution in India.
- No Objection Certificate (NOC): This certificate is required for certain products, such as blood products, to ensure they meet Indian standards.
- Test License: For products intended for clinical trials or testing, a test license is necessary.
- Customs Documentation: Proper customs procedures must be followed, including the submission of import declarations and payment of applicable duties and taxes.
Ensuring all documentation is accurate and complete is vital for the timely clearance of pharmaceutical imports through Indian customs.
Product Categories & Therapeutic Trends — India to Chile
Dominant categories, emerging opportunities, and demand drivers
1Dominant Import Categories
The pharmaceutical imports from Chile to India are predominantly focused on specific therapeutic areas, with paracetamol being the most significant import. This suggests that Chile's pharmaceutical exports to India are concentrated in the analgesic segment, catering to the demand for pain relief medications. The limited variety of imported products indicates that Chile's pharmaceutical offerings in the Indian market are specialized, possibly due to production capabilities or strategic market positioning.
2Innovation & Specialty Imports
The current trade data indicates that the pharmaceutical imports from Chile to India are limited to standard formulations, with no significant presence of innovator drugs, novel formulations, or advanced therapies. This suggests that Chilean pharmaceutical exports to India may not yet include cutting-edge or specialized products, potentially due to regulatory challenges, market entry barriers, or a focus on more established product lines.
3Import Demand Drivers
The demand for pharmaceutical imports from Chile to India is likely driven by several factors:
- Patent Protection: Chilean pharmaceutical products may offer patent protection, providing exclusivity in the Indian market.
- Technology Gaps: Certain formulations or manufacturing technologies in Chile may not be available domestically in India, creating a niche market for imports.
- Quality Requirements: Chilean products may meet specific quality standards that align with the preferences of Indian consumers or regulatory bodies.
- Disease Burden: The prevalence of certain health conditions in India may drive the need for specific pharmaceutical products, such as paracetamol for pain management.
These factors collectively contribute to the sustained, albeit limited, demand for pharmaceutical imports from Chile to India.
Trade Policy & Tariff Intelligence — India and Chile
Tariff structure, trade agreements, IP and patent landscape
1India's Import Tariff Structure
India's import tariff structure for pharmaceutical formulations includes several components:
- Most Favored Nation (MFN) Tariff Rates: These are the standard import duty rates applied to goods from WTO member countries, including Chile.
- Basic Customs Duty (BCD): This is the primary duty levied on imported goods, calculated as a percentage of the product's value.
- Integrated Goods and Services Tax (IGST): Applied to imported goods, IGST is calculated on the total value of the goods, including customs duties.
- Health Cess: A specific levy imposed on certain imported pharmaceutical products to fund health initiatives.
The exact rates for these duties and taxes can vary and are subject to periodic revisions by the Indian government. Chilean exporters should consult the latest notifications from the Directorate General of Foreign Trade (DGFT) and the Central Board of Indirect Taxes and Customs (CBIC) to determine the applicable rates for their products.
2Trade Agreements & Preferences
As of March 2026, there are no specific Free Trade Agreements (FTAs) or preferential duty rates between India and Chile concerning pharmaceutical products. The Partial Scope Trade Agreement between India and Chile, signed in 2007 and deepened in 2017, provides tariff preferences ranging from 20 percent to 100 percent for pharmaceutical products. However, the limited volume of pharmaceutical trade between the two countries suggests that these preferences have had a minimal impact on the overall trade dynamics. Both nations continue to engage in bilateral negotiations to explore opportunities for enhancing trade relations, but no significant developments have been reported in the pharmaceutical sector.
3IP, Patents & Price Control
India's patent regime, particularly Section 3(d) of the Indian Patents Act, aims to prevent evergreening of patents by restricting the patentability of new forms of known substances unless they demonstrate enhanced efficacy. This provision can impact the import of certain pharmaceutical products from Chile if they are considered incremental innovations. Additionally, the National Pharmaceutical Pricing Authority (NPPA) in India exercises price control over essential medicines, which can affect the pricing strategies of imported drugs. Chilean pharmaceutical companies must navigate these regulatory frameworks to ensure
FAQ — India to Chile Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Chile?
India exported pharmaceuticals worth $60.1K to Chile across 34 verified shipments.
Who are the top Indian pharmaceutical exporters to Chile?
1. PINNACLE CHILE SPA. — $58.8K. 2. SYNTHON CHILE LTDA — $391. 3. SYNTHON CHILE — $285. Total: 8 suppliers.
Which companies in Chile import pharmaceuticals from India?
1. PINNACLE LIFE SCIENCE PRIVATE LIMITED — $58.8K. 2. VEEDA CLINICAL RESEARCH LIMITED — $391. 3. AZIDUS LABORATORIES LIMITED — $327. 6 buyers total.
What pharmaceutical products does India export most to Chile?
1. Paracetamol ($58.8K, 97.9%); 2. Lenalidomide ($391, 0.7%); 3. Empagliflozin ($285, 0.5%); 4. Amlodipine ($200, 0.3%); 5. Valsartan ($200, 0.3%)
Which ports handle pharmaceutical shipments from India to Chile?
Export: . Import: .
Why does Chile import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $60.1K corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Chile?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Chile pharmaceutical trade?
$1.8K per consignment across 34 shipments.
How many Indian pharmaceutical companies export to Chile?
8 Indian companies. Largest: PINNACLE CHILE SPA. with $58.8K.
How can I find verified Indian pharmaceutical suppliers for Chile?
TransData Nexus covers 8 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Chile Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 34 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Chile identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 34 verified shipments from 8 Indian exporters to 6 Chile buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
8 Exporters
6 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists