Bangladesh to India Pharmaceutical Import
Bilateral Trade Intelligence · $7.2K Total Trade · 23 Foreign Suppliers · 19 Indian Buyers · DGFT Verified · Updated March 2026
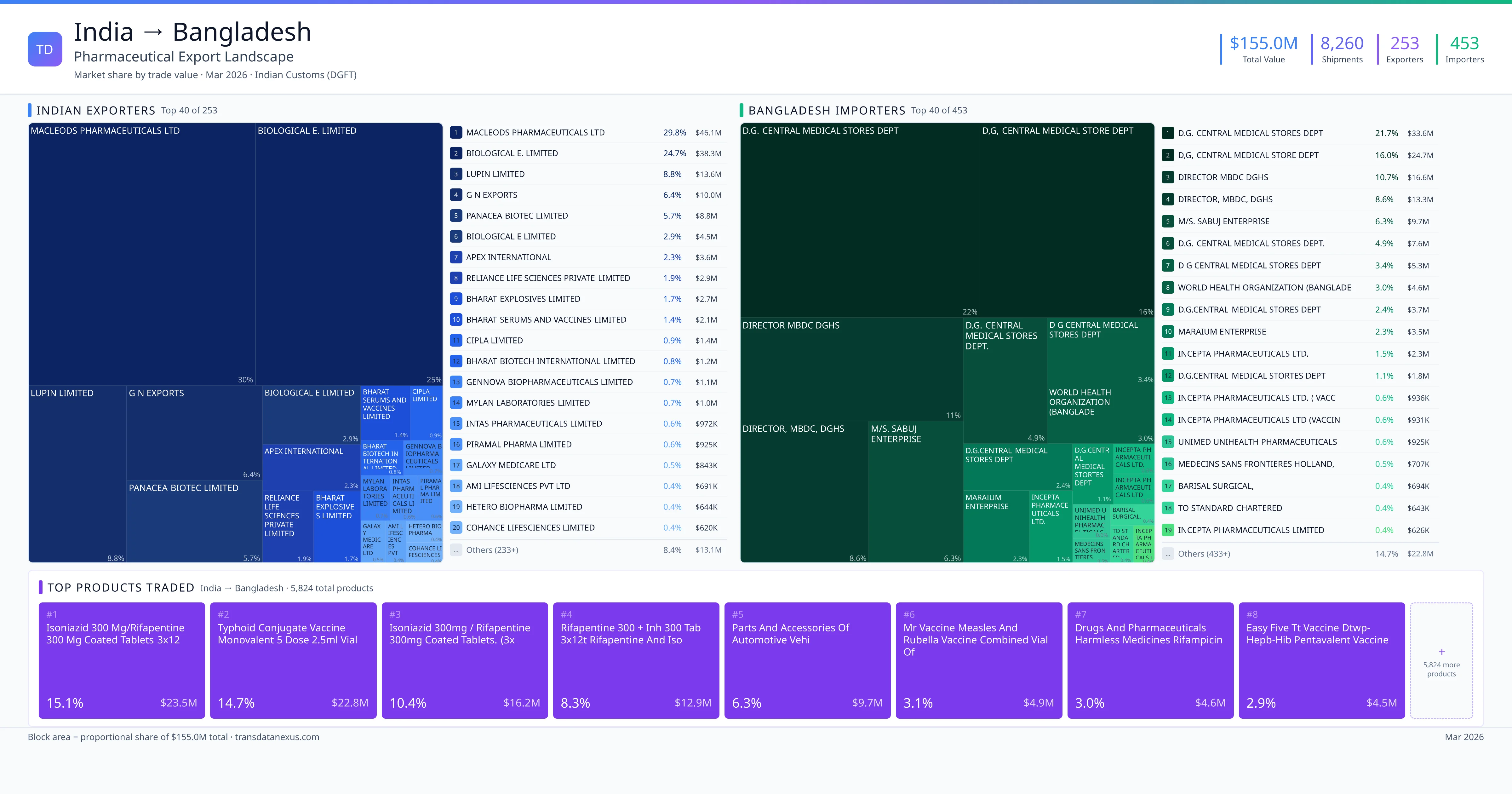
India imported $7.2K worth of pharmaceutical formulations from Bangladesh across 89 verified shipments, from 23 foreign suppliers to 19 Indian buyers. The top suppliers are SQUARE PHARMACEUTICALS LTD. ($1.5K) and SQUARE PHARMACEUTICAL LTD ($1.5K). The leading products are Famotidine ($3K) and Salbutamol ($1K). Average shipment value: $81.
Top Pharmaceutical Formulations — Bangladesh to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Famotidine | $3.3K | 46.1% |
| 2 | Salbutamol | $1.2K | 16.6% |
| 3 | Plasma | $649 | 9.1% |
| 4 | Empagliflozin | $380 | 5.3% |
| 5 | Linagliptin | $364 | 5.1% |
| 6 | Fluticasone | $268 | 3.7% |
| 7 | Vaccine | $208 | 2.9% |
| 8 | Erythromycin | $180 | 2.5% |
| 9 | Sodium | $101 | 1.4% |
| 10 | Montelukast | $72 | 1.0% |
| 11 | Bisoprolol | $70 | 1.0% |
| 12 | Metformin | $60 | 0.8% |
| 13 | Trimethoprim | $49 | 0.7% |
| 14 | Fluoxetine | $47 | 0.7% |
| 15 | Citalopram | $46 | 0.6% |
India imports 20+ pharmaceutical formulations from Bangladesh with a combined trade value of $7.2K. Key products include Famotidine ($3K), Salbutamol ($1K), Plasma ($649), Empagliflozin ($380), Linagliptin ($364). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 89 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — Bangladesh to India Trade Routes
These are the top pharmaceutical products exported from India to Bangladesh, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Bangladesh buyers, regulatory requirements, and logistics for that specific product corridor. Products include Famotidine ($3K), Salbutamol ($1K), Plasma ($649), Empagliflozin ($380), Linagliptin ($364) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Bangladesh Suppliers to India
23 Indian pharmaceutical companies export finished formulations to Bangladesh. Leading exporters include Square Pharmaceuticals Ltd., Square Pharmaceutical Ltd, Beximco Pharmaceuticals Limited, Beximco Pharmaceuticals Ltd.. The top exporter accounts for 20.8% of total India–Bangladesh pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from Bangladesh
19 companies in Bangladesh import pharmaceutical formulations from India. Top buyers include Cliantha Research Limited, Axis Clinicals Limited, Serum Institute Of India Private Limited, Syngene International Limited. The largest buyer accounts for 60.1% of India–Bangladesh pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
India–Bangladesh Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade corridor between Bangladesh and India has experienced notable growth and diversification over recent years. In 2024, India imported pharmaceutical products from Bangladesh valued at approximately $6.75 million, marking a significant increase from previous years. This surge reflects the strengthening of bilateral trade relations and Bangladesh's growing capacity to meet India's pharmaceutical demands. The trade encompasses a wide range of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, indicating a shift towards more complex and varied product offerings.
The evolution of this trade corridor is also characterized by the increasing involvement of multiple suppliers and buyers. In 2024, 23 foreign suppliers from Bangladesh and 19 Indian buyers participated in this trade, highlighting the expanding network of stakeholders. The average shipment value stood at $81, suggesting a focus on smaller, more frequent transactions. This trend underscores the growing integration of Bangladesh's pharmaceutical industry into India's supply chain, driven by competitive pricing and the ability to meet specific market needs.
2Bangladesh's Role in India's Pharma Imports
Bangladesh's contribution to India's pharmaceutical imports, while modest in absolute terms, is significant in certain therapeutic areas. The top ten products imported from Bangladesh in 2024 include Famotidine, Salbutamol, Plasma, Empagliflozin, Linagliptin, Fluticasone, Vaccines, Erythromycin, Sodium, and Montelukast. These products cater to a diverse range of medical conditions, from gastrointestinal disorders to respiratory issues and metabolic diseases. The presence of vaccines in this list indicates Bangladesh's growing capabilities in producing biologics, a sector that is increasingly important in global healthcare.
Despite the relatively small total import value, the diversity and specificity of the products suggest that Bangladesh plays a crucial role in supplying niche pharmaceutical products to India. This role is particularly important in areas where India may face supply constraints or where imported products offer advantages over domestic alternatives.
3Recent Developments
Between 2024 and 2026, several developments have influenced the pharmaceutical trade between Bangladesh and India. The Central Drugs Standard Control Organisation (CDSCO) in India has implemented policy changes aimed at streamlining the registration process for foreign pharmaceutical products, including those from Bangladesh. These changes are designed to reduce approval times and enhance market access for foreign suppliers. Additionally, trade agreements between India and Bangladesh have been revisited to address tariff structures and non-tariff barriers, fostering a more conducive environment for pharmaceutical trade. Regulatory harmonization efforts have also been undertaken to align standards and facilitate smoother cross-border transactions.
Bangladesh Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1CDSCO Registration Process
For Bangladeshi pharmaceutical companies aiming to export finished formulations to India, obtaining approval from the Central Drugs Standard Control Organisation (CDSCO) is mandatory. The registration process involves several key steps:
1. Application Submission: Companies must submit an application through the CDSCO's online portal, providing detailed information about the product, including its composition, manufacturing process, and intended use.
2. Form 40/41: Depending on the product category, applicants may need to submit Form 40 (for new drugs) or Form 41 (for clinical trial applications). These forms require comprehensive data to demonstrate the product's safety and efficacy.
3. Clinical Trial Waivers: In certain cases, CDSCO may grant waivers for clinical trials if the product has been approved in other countries with stringent regulatory standards.
4. Approval Timeline: The approval process can vary in duration, typically ranging from several months to over a year, depending on the complexity of the product and the completeness of the application.
It's crucial for Bangladeshi companies to ensure that all documentation is accurate and complete to avoid delays. Engaging with regulatory consultants familiar with CDSCO requirements can facilitate a smoother approval process.
2GMP & Manufacturing Standards
India requires that pharmaceutical products imported from Bangladesh adhere to Good Manufacturing Practice (GMP) standards. Manufacturing facilities must be certified by recognized authorities, such as the World Health Organization (WHO), to ensure compliance with quality and safety standards. The CDSCO conducts inspections to verify adherence to these standards, assessing aspects like facility conditions, equipment, personnel qualifications, and quality control processes. Non-compliance can result in delays or rejections of import applications.
3Import Documentation
To import pharmaceutical products into India, Bangladeshi companies must provide several key documents:
1. Import License (Form 10): This license authorizes the importation of drugs into India and is issued by the CDSCO.
2. Registration Certificate: Proof that the product has been registered with the CDSCO, indicating that it meets Indian regulatory standards.
3. No Objection Certificate (NOC): A certificate from the manufacturer stating that they have no objection to the importation of the product into India.
4. Test License: A license that permits the testing of the imported product in India to ensure it meets quality standards.
5. Customs Procedures: Compliance with Indian customs regulations is essential, including proper labeling, packaging, and documentation to facilitate smooth clearance.
Ensuring that all documentation is accurate and up-to-date is vital to prevent delays or issues during the import process.
Product Categories & Therapeutic Trends — India to Bangladesh
Dominant categories, emerging opportunities, and demand drivers
1Dominant Import Categories
India's pharmaceutical imports from Bangladesh are predominantly composed of finished pharmaceutical formulations. The top ten products imported in 2024 include:
1. Famotidine ($3K, 46.1%)
2. Salbutamol ($1K, 16.6%)
3. Plasma ($649, 9.1%)
4. Empagliflozin ($380, 5.3%)
5. Linagliptin ($364, 5.1%)
6. Fluticasone ($268, 3.7%)
7. Vaccine ($208, 2.9%)
8. Erythromycin ($180, 2.5%)
9. Sodium ($101, 1.4%)
10. Montelukast ($72, 1.0%)
These products cater to a wide range of therapeutic areas, including gastrointestinal disorders, respiratory conditions, metabolic diseases, and infectious diseases. The presence of vaccines and plasma products indicates Bangladesh's growing capabilities in producing biologics and complex formulations, sectors that are increasingly important in global healthcare.
2Innovation & Specialty Imports
Bangladesh's pharmaceutical industry has been advancing into complex formulations, including insulin, hormones, anti-cancer drugs, metered-dose inhalers (MDIs), dry powder inhalers (DPIs), and lyophilized injectables. This progression is driven by the increasing demand for specialized treatments targeting conditions such as cancer, cardiac diseases, renal disorders, and diabetes. The export of these advanced therapies to India reflects Bangladesh's growing capacity to meet the needs of the Indian market for innovative and specialized pharmaceutical products.
3Import Demand Drivers
India's importation of pharmaceutical products from Bangladesh is influenced by several factors:
1. Patent Protection: Bangladesh's pharmaceutical industry benefits from the TRIPS waiver, allowing it to produce and export patented drugs without infringement, making these products more accessible and affordable in India.
2. Technology Gaps: Bangladesh's advancements in manufacturing complex formulations and biologics fill technology gaps in India's domestic production capabilities, particularly in specialized treatments.
3. Quality Requirements: Bangladesh's adherence to international quality standards, including WHO-GMP certification, ensures that its pharmaceutical products meet the stringent quality requirements of the Indian market.
4. Disease Burden: The high prevalence of diseases such as diabetes, cardiovascular conditions, and respiratory disorders in India creates a sustained demand for specific pharmaceutical products that Bangladesh can supply.
These factors collectively drive the demand for Bangladeshi pharmaceutical imports into India, contributing to the strengthening of bilateral trade relations.
Trade Policy & Tariff Intelligence — India and Bangladesh
Tariff structure, trade agreements, IP and patent landscape
1India's Import Tariff Structure
India's import tariff structure for pharmaceutical formulations includes several components:
1. Most Favored Nation (MFN) Tariff Rates: India applies MFN tariff rates to imports from Bangladesh, as both countries are members of the South Asian Free Trade Area (SAFTA). These rates are designed to promote trade by reducing tariffs among member countries.
2. Basic Customs Duty: This is the primary duty levied on imported goods, including pharmaceutical products. The rate can vary depending on the product category and is subject to change based on government policies.
3. Integrated Goods and Services Tax (IGST): IGST is applicable on imported goods
FAQ — India to Bangladesh Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Bangladesh?
India exported pharmaceuticals worth $7.2K to Bangladesh across 89 verified shipments.
Who are the top Indian pharmaceutical exporters to Bangladesh?
1. SQUARE PHARMACEUTICALS LTD. — $1.5K. 2. SQUARE PHARMACEUTICAL LTD — $1.5K. 3. BEXIMCO PHARMACEUTICALS LIMITED — $1.2K. Total: 23 suppliers.
Which companies in Bangladesh import pharmaceuticals from India?
1. CLIANTHA RESEARCH LIMITED — $4.3K. 2. AXIS CLINICALS LIMITED — $1.1K. 3. SERUM INSTITUTE OF INDIA PRIVATE LIMITED — $648. 19 buyers total.
What pharmaceutical products does India export most to Bangladesh?
1. Famotidine ($3.3K, 46.1%); 2. Salbutamol ($1.2K, 16.6%); 3. Plasma ($649, 9.1%); 4. Empagliflozin ($380, 5.3%); 5. Linagliptin ($364, 5.1%)
Which ports handle pharmaceutical shipments from India to Bangladesh?
Export: . Import: .
Why does Bangladesh import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $7.2K corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Bangladesh?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Bangladesh pharmaceutical trade?
$81 per consignment across 89 shipments.
How many Indian pharmaceutical companies export to Bangladesh?
23 Indian companies. Largest: SQUARE PHARMACEUTICALS LTD. with $1.5K.
How can I find verified Indian pharmaceutical suppliers for Bangladesh?
TransData Nexus covers 23 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Bangladesh Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 89 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Bangladesh identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 89 verified shipments from 23 Indian exporters to 19 Bangladesh buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
23 Exporters
19 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists