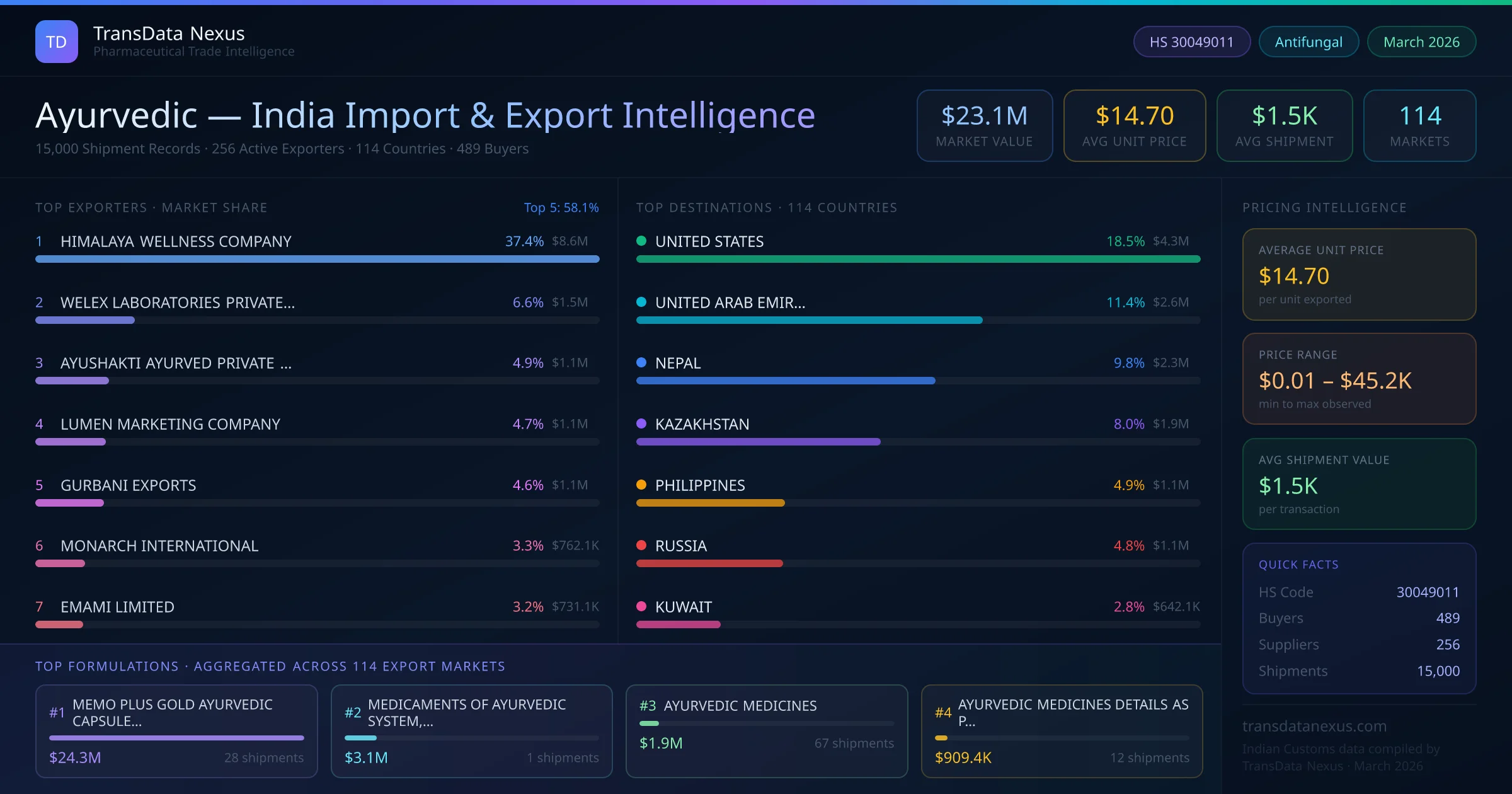
In June 2025, the Indian Ministry of AYUSH introduced new guidelines aimed at enhancing the quality control of Ayurvedic products intended for export. These guidelines emphasize stringent testing for heavy metals and microbial contamination, aligning with international safety standards.
In September 2025, the European Medicines Agency (EMA) updated its list of herbal monographs to include several Ayurvedic herbs, providing clearer guidance for companies seeking traditional use registrations within the EU.
In December 2025, the United States Pharmacopeia (USP) announced the inclusion of monographs for select Ayurvedic ingredients, facilitating standardization and quality assurance for products entering the U.S. market.
In February 2026, the World Health Organization (WHO) published a comprehensive report on the safety and efficacy of traditional medicines, including Ayurveda, encouraging member states to integrate these practices into their national health systems.
In March 2026, the Indian government launched an initiative to support small and medium-sized enterprises (SMEs) in the Ayurvedic sector, providing financial assistance and training to enhance export capabilities and compliance with international regulations.
These developments reflect a global trend towards the recognition and integration of Ayurvedic medicine into mainstream healthcare, provided that products meet established quality and safety standards.