Antidandruff products are regulated by the U.S. Food and Drug Administration (FDA) as over-the-counter (OTC) drugs. According to 21 CFR § 358.750, these products must comply with specific labeling requirements, including statements of identity and indications for use. (law.cornell.edu)
The FDA's Orange Book lists approved drug products and their therapeutic equivalence evaluations. A search of the Orange Book reveals that there are several approved Abbreviated New Drug Applications (ANDAs) for antidandruff products, indicating the presence of generic alternatives in the market. However, specific details regarding the number of approved ANDAs and recent approvals are not readily available in the provided sources.
Given that antidandruff products are classified as OTC drugs, they are not typically subject to import alerts unless specific safety concerns arise. The regulatory pathway for these products involves compliance with the FDA's OTC Monograph system, which outlines acceptable active ingredients, dosages, formulations, and labeling requirements.
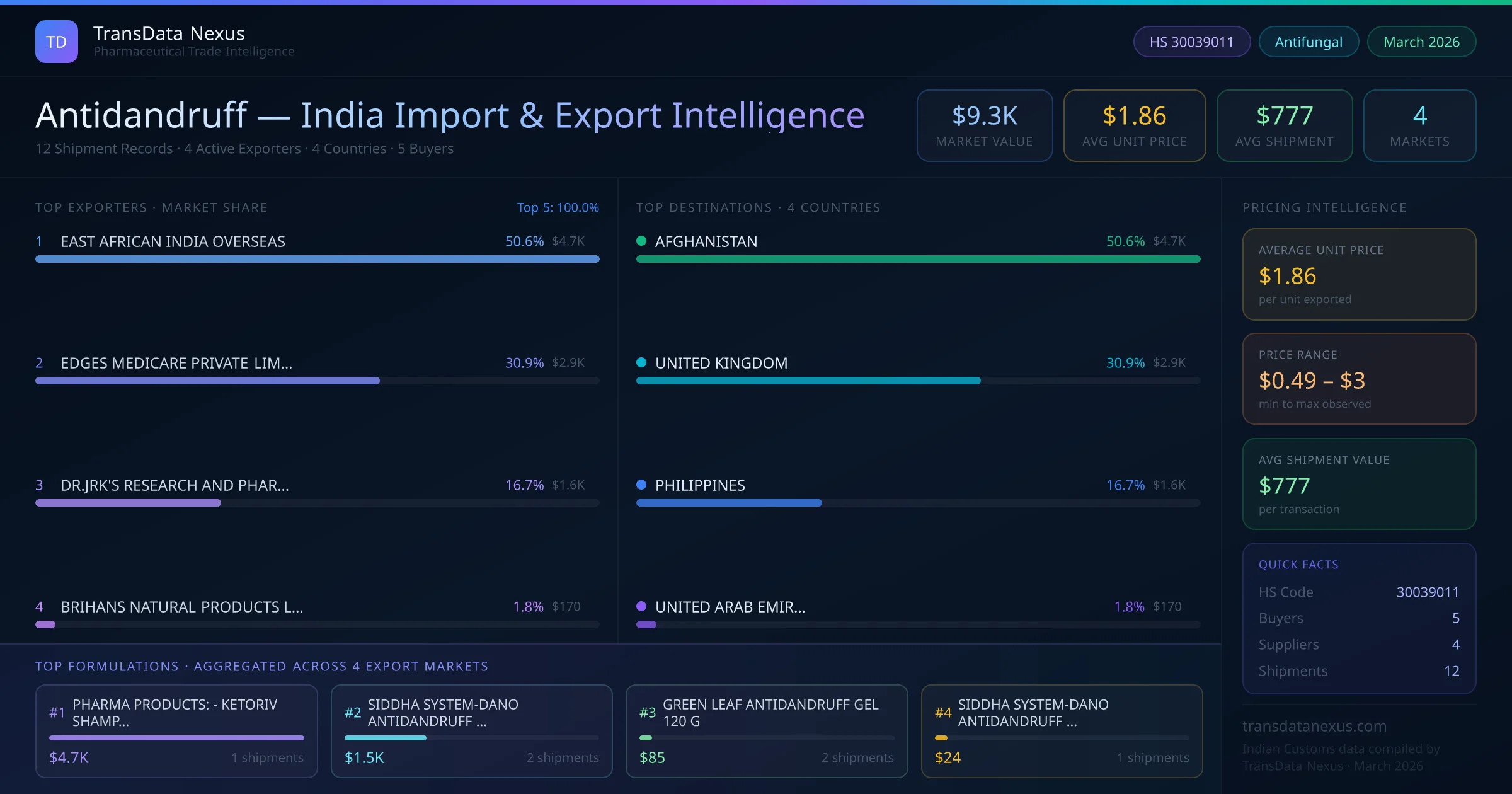
Considering the limited number of active Indian exporters (four) and the total export value of $0.0M USD from 2022 to 2026, it appears that Indian antidandruff products have a minimal presence in the U.S. market. This suggests potential opportunities for Indian manufacturers to expand their footprint by ensuring compliance with FDA regulations and exploring market entry strategies.