Yusen Logistics Benelux Belgium
Pharmaceutical Importer · Belgium · Gastrointestinal Focus · $279.0M Total Trade · DGFT Verified
Yusen Logistics Benelux Belgium is a pharmaceutical importer based in Belgium with a total trade value of $279.0M across 42 products in 12 therapeutic categories. Based on 7,629 verified import shipments from Indian Customs (DGFT) records, Yusen Logistics Benelux Belgium is the #1 buyer in 9 products including Esomeprazole, Magnesium, Omeprazole. Yusen Logistics Benelux Belgium sources from 2 verified Indian suppliers, with Sun Pharmaceutical Industries Limited accounting for 96.6% of imports.
Yusen Logistics Benelux Belgium — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Yusen Logistics Benelux Belgium?
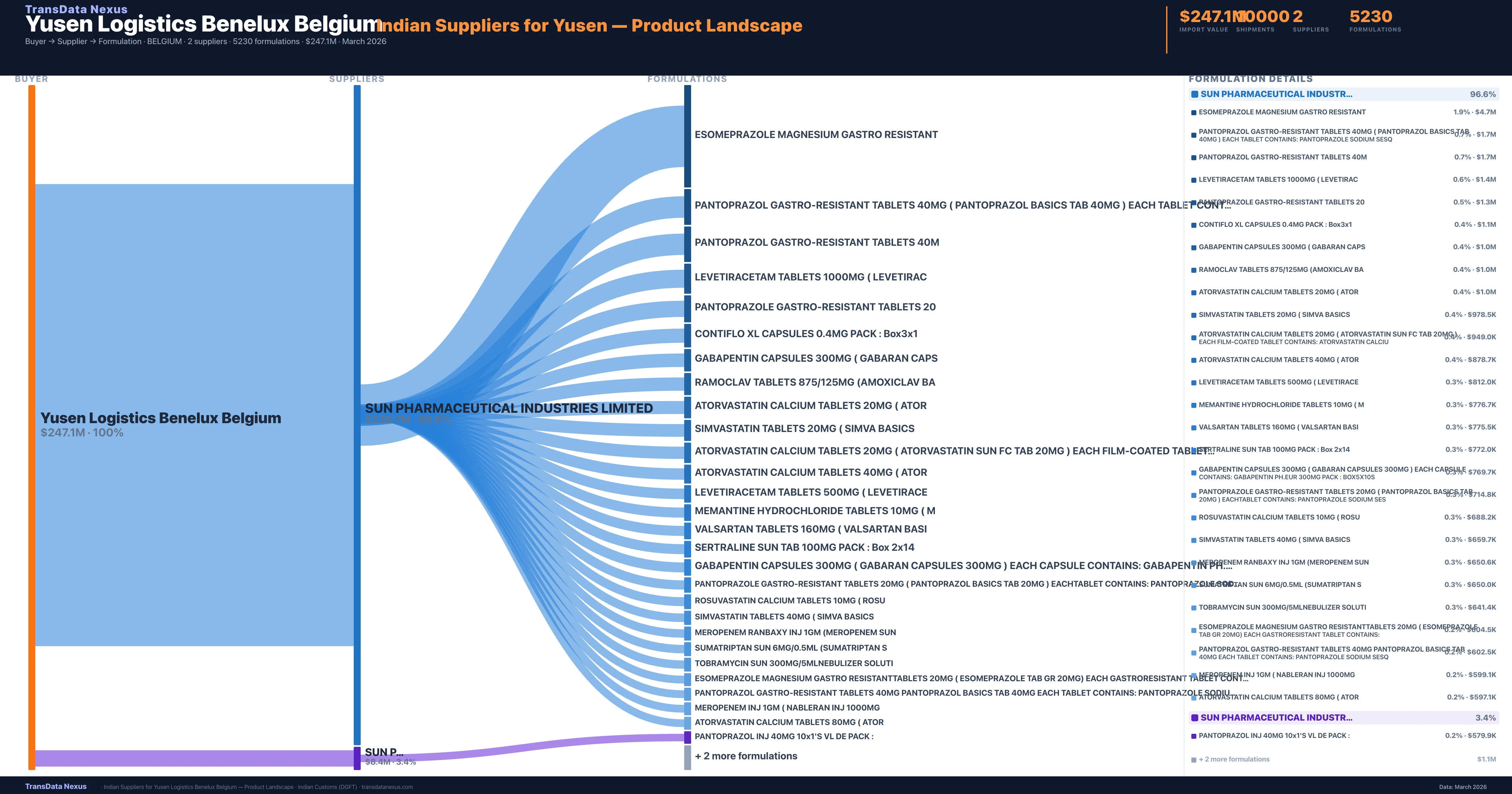
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Sun Pharmaceutical Industries Limited | $238.7M | 9,651 | 96.6% |
| Sun Pharmaceutical Industries Ltd | $8.4M | 349 | 3.4% |
Yusen Logistics Benelux Belgium sources from 2 verified Indian suppliers across 5,230 distinct formulations. The sourcing is highly concentrated — Sun Pharmaceutical Industries Limited accounts for 96.6% of total imports, indicating a strategic single-source relationship.
What Formulations Does Yusen Logistics Benelux Belgium Import?
| Formulation | Value | Ships |
|---|---|---|
| Esomeprazole magnesium gastro | $4.7M | 140 |
| Pantoprazol Gastro-Resistant tablets 40MG ( pantoprazol basics TAB 40MG ) Each tablet contains: pantoprazole sodium | $1.7M | 77 |
| Pantoprazol Gastro-Resistant tablets | $1.7M | 42 |
| Levetiracetam tablets 1000MG | $1.4M | 41 |
| Pantoprazole Gastro-Resistant tablets | $1.3M | 44 |
| Contiflo xl capsules 0.4MG Pack | $1.1M | 23 |
| Gabapentin capsules 300MG ( gabaran CAPS | $1.0M | 23 |
| Ramoclav tablets 875/125MG (amoxiclav | $1.0M | 22 |
| Atorvastatin calcium tablets 20MG | $1.0M | 48 |
| Simvastatin tablets 20MG ( simva | $978.5K | 32 |
| Atorvastatin calcium tablets 20MG ( atorvastatin sun fc TAB 20MG ) Each Film-Coated tablet contains: atorvastatin calciu | $949.0K | 77 |
| Atorvastatin calcium tablets 40MG | $878.7K | 35 |
| Levetiracetam tablets 500MG | $812.0K | 29 |
| Memantine hydrochloride tablets 10MG | $776.7K | 20 |
| Valsartan tablets 160MG ( valsartan | $775.5K | 20 |
Yusen Logistics Benelux Belgium imports 5,230 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Yusen Logistics Benelux Belgium Import?
Yusen Logistics Benelux Belgium Therapeutic Categories — 12 Specializations
Yusen Logistics Benelux Belgium imports across 12 therapeutic categories, with Gastrointestinal (24.3%), Vitamins & Supplements (19.0%), Cardiovascular (16.2%) representing the largest segments. Well-diversified — top 5 products represent 44% of imports.
Gastrointestinal
3 products · 24.3% · $67.8M
Vitamins & Supplements
2 products · 19.0% · $53.1M
Cardiovascular
7 products · 16.2% · $45.3M
CNS & Psychiatric
4 products · 11.2% · $31.2M
Medical Devices & Diagnostics
2 products · 7.6% · $21.1M
Advanced Oncology
11 products · 5.5% · $15.3M
Advanced Antibiotics
5 products · 4.7% · $13.2M
Antibiotics
2 products · 4.1% · $11.5M
Oncology
2 products · 2.8% · $7.8M
Import Portfolio — Top 30 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Esomeprazole | Gastrointestinal | $31.6M | 632 | 6.9% | 1 |
| 2 | Magnesium | Vitamins & Supplements | $29.6M | 591 | 8.2% | 1 |
| 3 | Calcium | Vitamins & Supplements | $23.5M | 939 | 2.6% | 7 |
| 4 | Omeprazole | Gastrointestinal | $20.4M | 407 | 3.2% | 1 |
| 5 | Strip | Medical Devices & Diagnostics | $16.4M | 328 | 8.3% | 3 |
| 6 | Pantoprazole | Gastrointestinal | $15.9M | 523 | 1.2% | 5 |
| 7 | Atorvastatin | Cardiovascular | $14.7M | 663 | 1.4% | 10 |
| 8 | Sertraline | CNS & Psychiatric | $14.1M | 281 | 2.5% | 2 |
| 9 | Rosuvastatin | Cardiovascular | $12.3M | 341 | 2.7% | 8 |
| 10 | Clarithromycin | Antibiotics | $9.2M | 186 | 9.3% | 2 |
| 11 | Citalopram | CNS & Psychiatric | $8.9M | 179 | 3.5% | 4 |
| 12 | Simvastatin | Cardiovascular | $8.9M | 368 | 7.1% | 2 |
| 13 | Meropenem | Advanced Antibiotics | $7.2M | 143 | 3.2% | 1 |
| 14 | Gabapentin | CNS & Psychiatric | $6.2M | 123 | 0.7% | 14 |
| 15 | Fluconazole | Antifungals | $5.1M | 123 | 5.5% | 2 |
| 16 | Gemcitabine | Oncology | $5.1M | 116 | 10.9% | 1 |
| 17 | Syringe | Medical Devices & Diagnostics | $4.8M | 95 | 2.6% | 2 |
| 18 | Temozolomide | Advanced Oncology | $4.7M | 184 | 19.9% | 1 |
| 19 | Valsartan | Cardiovascular | $4.5M | 187 | 2.3% | 8 |
| 20 | Fenofibrate | Lipid & Metabolism | $3.7M | 74 | 5.0% | 1 |
| 21 | Ezetimibe | Lipid & Metabolism | $3.4M | 99 | 0.7% | 7 |
| 22 | Cefuroxime | Advanced Antibiotics | $3.1M | 93 | 2.4% | 4 |
| 23 | Irinotecan | Oncology | $2.8M | 78 | 6.7% | 2 |
| 24 | Irbesartan | Cardiovascular | $2.6M | 144 | 2.4% | 8 |
| 25 | Lenalidomide | Advanced Oncology | $2.5M | 79 | 0.3% | 9 |
| 26 | Tobramycin | Antibiotics | $2.4M | 47 | 6.0% | 4 |
| 27 | Oxaliplatin | Advanced Oncology | $2.2M | 94 | 4.8% | 4 |
| 28 | Escitalopram | CNS & Psychiatric | $2.1M | 93 | 1.1% | 11 |
| 29 | Ramipril | Cardiovascular | $1.8M | 143 | 2.0% | 11 |
| 30 | Imipenem | Advanced Antibiotics | $1.6M | 31 | 4.6% | 2 |
Yusen Logistics Benelux Belgium imports 42 pharmaceutical products across 12 categories into Belgium totaling $279.0M. The company is the #1 buyer for 9 products: Esomeprazole, Magnesium, Omeprazole, Meropenem, Gemcitabine.
All 42 Products
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Yusen Logistics Benelux Belgium.
Request DemoYusen Logistics Benelux Belgium — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Yusen Logistics Benelux Belgium is a subsidiary of Yusen Logistics (Benelux) B.V., a logistics and supply chain management company headquartered in Moerdijk, Netherlands. (craft.co) The company specializes in providing comprehensive logistics services, including air and ocean freight forwarding, warehousing, distribution, and supply chain management. In Belgium, Yusen Logistics operates as a third-party logistics (3PL) provider, focusing on the pharmaceutical and healthcare sectors. The Belgian branch is located at Keetberglaan 2, 9120 Melsele, Belgium. (trendstop.knack.be)
Yusen Logistics Benelux is part of the global Yusen Logistics network, which is a subsidiary of Nippon Yusen Kabushiki Kaisha (NYK Line), one of the world's largest shipping companies. (craft.co) The company plays a significant role in Belgium's pharmaceutical supply chain by offering specialized logistics services tailored to the needs of pharmaceutical companies. This includes the importation and distribution of finished pharmaceutical formulations, as evidenced by TransData Nexus proprietary trade data indicating that Yusen Logistics Benelux Belgium imported $279.0 million USD worth of finished pharmaceutical products from India between 2022 and 2026.
2Distribution Network
Yusen Logistics Benelux operates multiple warehouse facilities in Belgium to support its logistics operations. Notably, the company has a facility in Melsele, near Antwerp, which was expanded in July 2020 to include a new warehouse offering space for up to 15,000 pallets over nearly 5,000 square meters at a height of 17 meters. Additionally, in June 2024, Yusen Logistics, in collaboration with MG Real Estate and GSK, inaugurated a new 40,000 square meter pharmaceutical warehouse in Gembloux, Wallonia. This facility is the largest biopharmaceutical logistics center in Wallonia and features advanced technologies such as automated mobile robots and automated packaging systems. (mgrealestate.eu)
Yusen Logistics Benelux Belgium serves as a critical logistics intermediary within Belgium's pharmaceutical supply chain. The company's role encompasses the importation, storage, and distribution of finished pharmaceutical products, ensuring that these products are efficiently and safely delivered to various stakeholders, including wholesalers, hospitals, and pharmacies. The company's significant import activities, particularly from Indian pharmaceutical manufacturers, highlight its pivotal position in facilitating the availability of a diverse range of pharmaceutical products in the Belgian market.
Supplier Relationship Intelligence — Yusen Logistics Benelux Belgium
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Yusen Logistics Benelux Belgium's import data from India between 2022 and 2026 indicates a significant reliance on a single supplier. Out of a total import value of $279.0 million USD across 7,629 shipments, a substantial $238.7 million (approximately 85.6%) was sourced from SUN PHARMACEUTICAL INDUSTRIES LIMITED, accounting for 9,651 shipments. An additional $8.4 million (3.0%) came from SUN PHARMACEUTICAL INDUSTRIES LTD, with 349 shipments. This results in a combined supplier concentration of 88.6% from these two entities, leaving only 11.4% of imports diversified among other suppliers.
Such a high level of supplier concentration poses potential risks. While a strong relationship with a primary supplier can ensure consistent quality and streamlined operations, over-reliance on a single source may expose the company to supply disruptions due to unforeseen events affecting the supplier. Additionally, this dependency can diminish Yusen Logistics' negotiating leverage, potentially leading to less favorable pricing and terms. Industry benchmarks suggest that for non-specialized categories, a Herfindahl-Hirschman Index (HHI) below 1,500 indicates low concentration, while above 2,500 signifies high concentration. In this context, Yusen's supplier concentration would likely fall into the high category, indicating a need for diversification to mitigate associated risks.
2Supply Chain Resilience
The resilience of Yusen Logistics Benelux Belgium's supply chain is currently compromised due to its heavy dependence on a single supplier. The absence of a robust backup supplier strategy means that any disruption affecting SUN PHARMACEUTICAL INDUSTRIES LIMITED could significantly impact Yusen's ability to meet market demands. While the company imports a diverse range of 42 products across 12 therapeutic categories, the lack of supplier diversification undermines the overall resilience of the supply chain.
To enhance supply chain resilience, it is advisable for Yusen Logistics to establish relationships with additional suppliers, particularly those in different geographic regions. This geographic diversification can mitigate risks associated with regional disruptions. Furthermore, ensuring that all suppliers comply with regulatory standards, such as those set by the European Medicines Agency (EMA), is crucial to maintain product quality and regulatory compliance.
3Strategic Implications
The current sourcing pattern places Yusen Logistics Benelux Belgium in a vulnerable position regarding supply security and negotiating power. The heavy reliance on a single supplier limits the company's ability to negotiate favorable terms and exposes it to potential supply chain disruptions. Diversifying the supplier base would not only mitigate these risks but also enhance the company's competitive position by ensuring a more stable and flexible supply chain.
For Indian exporters seeking to become alternative suppliers, this presents an opportunity to engage with Yusen Logistics. By offering competitive pricing, ensuring regulatory compliance, and demonstrating reliability, these exporters can position themselves as viable partners. This diversification would benefit both parties by reducing dependency risks and fostering a more resilient supply chain.
Importing Pharmaceuticals into Belgium — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Belgium
1Regulatory Authority & Framework
In Belgium, the Federal Agency for Medicines and Health Products (FAMHP) serves as the national regulatory authority overseeing the importation and distribution of pharmaceutical products. The primary legislation governing these activities includes the Royal Decree of 14 December 2006, which regulates medicinal products for human use.
For Indian generic pharmaceuticals to be marketed in Belgium, they must obtain a marketing authorization. This process involves submitting a comprehensive dossier to the FAMHP, detailing the product's quality, safety, and efficacy. The authorization can be pursued through national procedures or via the European Medicines Agency (EMA) for centralized approval, depending on the product's classification and intended market reach.
2Import Licensing & GMP
Importing pharmaceutical products into Belgium requires a Manufacturing and Import Authorization (MIA), which mandates compliance with Good Manufacturing Practice (GMP) standards. The EU recognizes GMP certificates from countries with Mutual Recognition Agreements (MRAs); however, India is not currently among these countries. Consequently, Indian manufacturing sites must undergo inspections by EU authorities to ensure GMP compliance. Additionally, entities involved in the wholesale distribution of pharmaceuticals must obtain a Wholesale Distribution Authorization (WDA) from the FAMHP, ensuring adherence to Good Distribution Practice (GDP) guidelines.
3Quality & Labeling
Each batch of imported pharmaceuticals must be certified by a Qualified Person (QP) within the EU before release to the market. This certification includes a full qualitative analysis, quantitative analysis of all active ingredients, and other necessary tests to confirm compliance with the marketing authorization. Labeling requirements in Belgium stipulate that information must be presented in the country's three official languages: Dutch, French, and German. Labels must include essential details such as the product name, dosage, method of administration, and any necessary warnings or precautions.
4Recent Regulatory Changes
Several regulatory changes between 2024 and 2026 have impacted the importation of Indian pharmaceuticals into Belgium:
- September 2024: Belgium introduced a Royal Decree on 12 September 2024, effective from 1 November 2024, establishing import regimes for unlicensed medicines to address critical shortages. This decree allows for the importation of unlicensed medicines under specific conditions, potentially affecting the availability and distribution of certain pharmaceuticals.
- December 2025: The European Union reached a political agreement to modernize its pharmaceutical legislation, marking the most significant overhaul in over two decades. The new legislation aims to enhance access to medicines, streamline procedures, and foster innovation. The adopted acts are expected to enter into force in 2026, with a transition period until 2028 for member states to align their national laws. (ema.europa.eu)
- January 2026: Belgium updated its web portal 'Co-responsibility' on 14 January 2026 to enhance safety, harmonize GMP and GDP processes, and improve the traceability of inspection document archiving. This update aims to streamline regulatory processes and ensure compliance with GMP and GDP standards.
These regulatory developments underscore Belgium's commitment to ensuring the quality and safety of pharmaceutical imports, including those from India. Companies like Yusen Logistics Benelux Belgium, which imported finished pharmaceutical formulations valued at $279.0 million USD from India between 2022 and 2026, must stay abreast of these changes to maintain compliance and ensure uninterrupted market access.
Yusen Logistics Benelux Belgium — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Yusen Logistics Benelux Belgium's import data from India between 2022 and 2026 reveals a strategic focus on specific therapeutic areas. The top three categories—Gastrointestinal (24.3%), Vitamins & Supplements (19.0%), and Cardiovascular (16.2%)—account for nearly 60% of their imports. This concentration suggests a deliberate alignment with prevalent health concerns in Belgium, such as digestive disorders, nutritional deficiencies, and heart-related conditions. The emphasis on these categories indicates a response to substantial market demand, aiming to address common health issues within the population.
The prominence of products like Esomeprazole ($31.6M), Magnesium ($29.6M), and Omeprazole ($20.4M) further underscores this strategy. Esomeprazole and Omeprazole are widely used for treating acid reflux and related gastrointestinal conditions, while Magnesium supplements cater to nutritional needs. By prioritizing these products, Yusen Logistics demonstrates a commitment to supplying medications that are essential for managing prevalent health concerns in Belgium.
2Sourcing Profile
Yusen Logistics Benelux Belgium's procurement strategy heavily favors generic pharmaceuticals, as evidenced by their substantial imports from Indian suppliers. Notably, 96.6% of their imports, amounting to $238.7M, were sourced from SUN PHARMACEUTICAL INDUSTRIES LIMITED, with an additional $8.4M (3.4%) from SUN PHARMACEUTICAL INDUSTRIES LTD. This reliance on a single supplier for the majority of their imports indicates a preference for cost-effective generic medications, which are integral to their supply chain.
The importation of 5,230 unique finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, reflects a diverse formulation preference. This variety ensures the availability of multiple administration routes, catering to different patient needs and preferences. India's role as a leading producer of high-quality, affordable generic drugs aligns with Yusen Logistics' strategy to provide a broad spectrum of medications while maintaining cost efficiency.
3Market Positioning
The product mix and sourcing patterns of Yusen Logistics Benelux Belgium suggest a strategic positioning within the Belgian pharmaceutical market. The focus on essential medications across key therapeutic areas, combined with the importation of finished pharmaceutical formulations, indicates a primary engagement with retail pharmacies and hospital networks. By supplying a wide range of generic medications, Yusen Logistics supports healthcare providers in offering cost-effective treatment options to patients.
Additionally, the scale and diversity of their imports position Yusen Logistics as a significant player in the wholesale distribution sector. Their ability to supply large volumes of essential medications aligns with the needs of government tenders and large healthcare institutions seeking reliable and affordable pharmaceutical products. This comprehensive approach enables Yusen Logistics to effectively serve various segments of the Belgian healthcare market.
Seller's Guide — How to Become a Supplier to Yusen Logistics Benelux Belgium
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
Given the current supplier concentration, opportunities for new Indian pharmaceutical exporters to collaborate with Yusen Logistics Benelux Belgium appear limited. Sun Pharmaceutical Industries Limited's substantial market share indicates a strong, established relationship, potentially leaving minimal room for additional suppliers. However, potential gaps may exist in therapeutic categories beyond the top three, or in niche products not currently supplied by existing partners. Identifying these specific areas could present entry points for new suppliers.
2Requirements & Qualifications
To engage with Yusen Logistics Benelux Belgium and the broader Belgian market, Indian pharmaceutical exporters must adhere to stringent quality and regulatory standards. Essential certifications include:
- Good Manufacturing Practice (GMP): Compliance with GMP standards, as recognized by the European Medicines Agency (EMA), is mandatory to ensure product quality and safety.
- European Union Good Distribution Practice (GDP): Adherence to GDP guidelines is crucial for maintaining product integrity throughout the supply chain.
- Product Registration: Medicinal products must be registered with the Federal Agency for Medicines and Health Products (FAMHP) in Belgium, necessitating comprehensive documentation and compliance with local regulations.
Additionally, obtaining a Certificate of Suitability (CEP) from the European Directorate for the Quality of Medicines (EDQM) can facilitate market entry by demonstrating compliance with European Pharmacopoeia standards.
3How to Approach
Establishing a partnership with Yusen Logistics Benelux Belgium requires a strategic and methodical approach:
1. Market Analysis: Conduct thorough research to identify unmet needs within Yusen's product portfolio, focusing on therapeutic areas or products not currently supplied by existing partners.
2. Regulatory Compliance: Ensure all products meet EU regulatory requirements, including GMP and GDP certifications, and complete the necessary product registrations with FAMHP.
3. Direct Engagement: Initiate contact with Yusen Logistics through formal channels, presenting a well-prepared proposal that highlights your company's capabilities, compliance credentials, and the specific products you offer that align with their potential needs.
4. Tender Participation: Monitor for any tender opportunities issued by Yusen Logistics or associated pharmaceutical companies. Participating in these tenders can provide a structured pathway to demonstrate your offerings and establish credibility.
5. Distributor Partnerships: Consider collaborating with established distributors in Belgium who have existing relationships with Yusen Logistics. This can serve as an indirect route to supply products while navigating market entry challenges.
Building a relationship with Yusen Logistics Benelux Belgium is a long-term endeavor, requiring consistent engagement, adherence to regulatory standards, and a clear demonstration of value addition to their supply chain.
Frequently Asked Questions — Yusen Logistics Benelux Belgium
Who supplies pharmaceutical formulations to Yusen Logistics Benelux Belgium from India?
Who supplies pharmaceutical formulations to Yusen Logistics Benelux Belgium from India? Yusen Logistics Benelux Belgium primarily sources its pharmaceutical formulations from two verified Indian suppliers: SUN PHARMACEUTICAL INDUSTRIES LIMITED, accounting for $238.7 million USD (96.6% of total imports), and SUN PHARMACEUTICAL INDUSTRIES LTD, contributing $8.4 million USD (3.4%). These two suppliers collectively fulfill the company's import requirements from India.
How much does Yusen Logistics Benelux Belgium import in pharmaceutical formulations from India?
How much does Yusen Logistics Benelux Belgium import in pharmaceutical formulations from India? Between 2022 and 2026, Yusen Logistics Benelux Belgium imported pharmaceutical formulations from India totaling $279.0 million USD, distributed over 7,629 shipments. This substantial import activity underscores the company's significant role in the pharmaceutical supply chain between India and Belgium.
What pharmaceutical products does Yusen Logistics Benelux Belgium import?
What pharmaceutical products does Yusen Logistics Benelux Belgium import? The company imports 42 distinct pharmaceutical products across 12 therapeutic categories. The top five products by import value are Esomeprazole ($31.6 million USD), Magnesium ($29.6 million USD), Calcium ($23.5 million USD), Omeprazole ($20.4 million USD), and Strip ($16.4 million USD). These products collectively represent 43.5% of the company's total import portfolio.
Which Indian pharmaceutical companies are verified suppliers to Yusen Logistics Benelux Belgium?
Which Indian pharmaceutical companies are verified suppliers to Yusen Logistics Benelux Belgium? Yusen Logistics Benelux Belgium's verified Indian suppliers are SUN PHARMACEUTICAL INDUSTRIES LIMITED and SUN PHARMACEUTICAL INDUSTRIES LTD. SUN PHARMACEUTICAL INDUSTRIES LIMITED is the predominant supplier, accounting for 96.6% of the total import value, while SUN PHARMACEUTICAL INDUSTRIES LTD contributes 3.4%.
What therapeutic categories does Yusen Logistics Benelux Belgium focus on?
What therapeutic categories does Yusen Logistics Benelux Belgium focus on? The company's imports are concentrated in three primary therapeutic categories: gastrointestinal (24.3% of total imports), vitamins & supplements (19.0%), and cardiovascular (16.2%). This focus reflects the company's strategic emphasis on these key areas within the pharmaceutical market.
How concentrated is Yusen Logistics Benelux Belgium's supplier base?
How concentrated is Yusen Logistics Benelux Belgium's supplier base? Yusen Logistics Benelux Belgium's supplier base is highly concentrated, with only two verified Indian suppliers. SUN PHARMACEUTICAL INDUSTRIES LIMITED alone accounts for 96.6% of the total import value, indicating a strong reliance on this single supplier for their pharmaceutical formulations.
What formulation types does Yusen Logistics Benelux Belgium import from India?
What formulation types does Yusen Logistics Benelux Belgium import from India? The company imports a diverse range of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. These products are fully manufactured and ready for distribution, highlighting the company's role in supplying ready-to-use medications to the Belgian market.
How can an Indian pharmaceutical company become a supplier to Yusen Logistics Benelux Belgium?
How can an Indian pharmaceutical company become a supplier to Yusen Logistics Benelux Belgium? To become a supplier, an Indian pharmaceutical company should demonstrate compliance with international quality standards, such as Good Manufacturing Practices (GMP) and Good Distribution Practices (GDP). Establishing a track record of regulatory approvals from authorities like the European Medicines Agency (EMA) and the Belgian Federal Agency for Medicines and Health Products (FAMHP) is also crucial. Building relationships through industry events and direct engagement with Yusen Logistics' procurement team can further facilitate supplier opportunities.
What quality certifications does Yusen Logistics Benelux Belgium require from Indian suppliers?
What quality certifications does Yusen Logistics Benelux Belgium require from Indian suppliers? Yusen Logistics Benelux Belgium requires Indian suppliers to adhere to stringent quality certifications, including compliance with GMP and GDP standards. Additionally, suppliers must obtain approvals from regulatory bodies such as the EMA and FAMHP to ensure the safety and efficacy of the pharmaceutical products supplied.
What is Yusen Logistics Benelux Belgium's importing pattern and order frequency?
What is Yusen Logistics Benelux Belgium's importing pattern and order frequency? Between 2022 and 2026, Yusen Logistics Benelux Belgium imported 7,629 shipments from India, averaging approximately 1,526 shipments per year. This consistent import pattern indicates a steady demand for pharmaceutical formulations, reflecting the company's ongoing commitment to maintaining a robust supply chain.
Get Full Yusen Logistics Benelux Belgium Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Yusen Logistics Benelux Belgium identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Yusen Logistics Benelux Belgium's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 7,629 individual customs records matching Yusen Logistics Benelux Belgium.
- 5.Supplier Verification: Yusen Logistics Benelux Belgium sources from 2 verified Indian suppliers across 5,230 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
42 Products Tracked
12 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.