Unicef Supply Division,
Pharmaceutical Importer · Denmark · Other Focus · $7.8M Total Trade · DGFT Verified
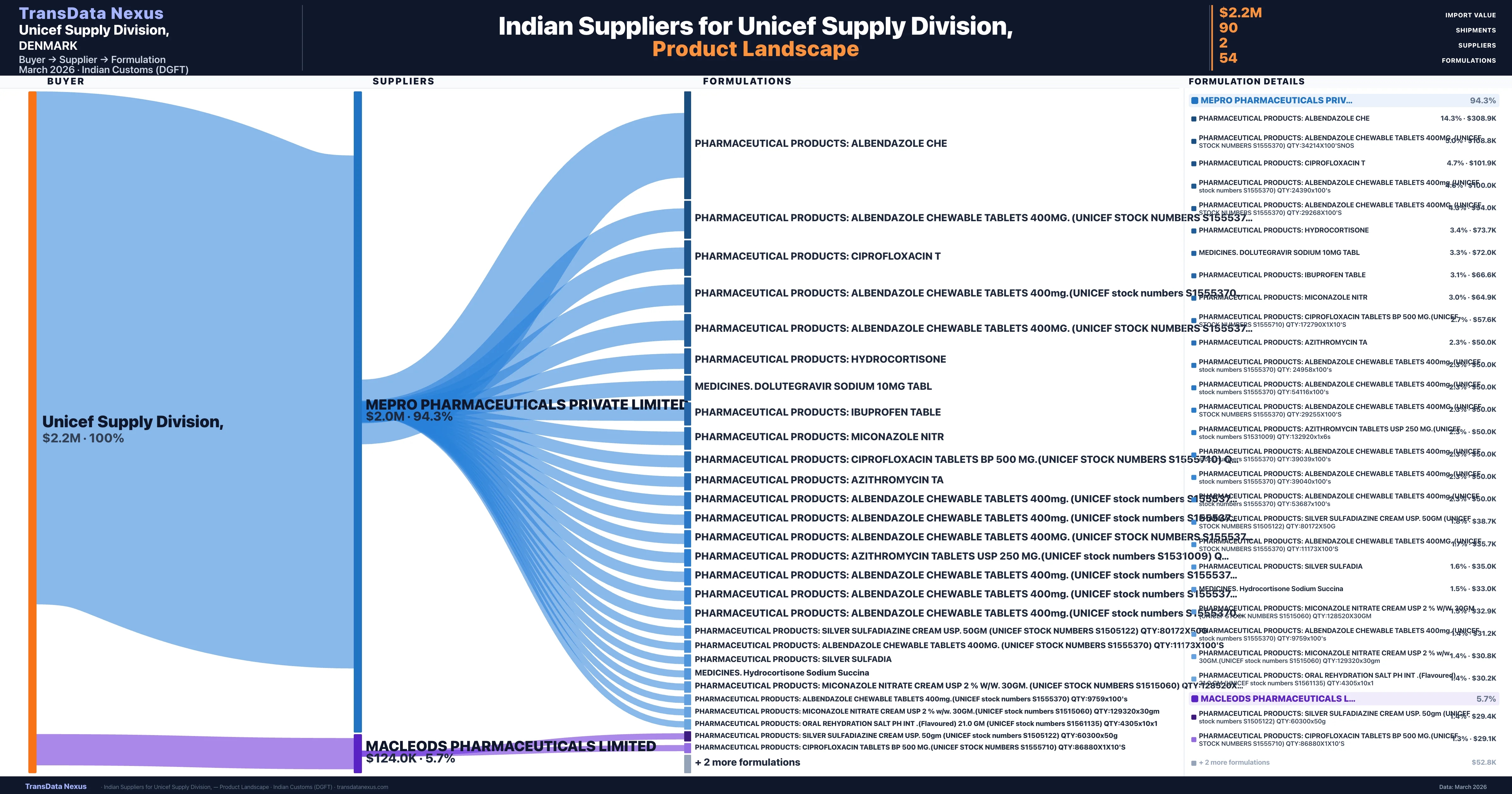
Unicef Supply Division, is a pharmaceutical importer based in Denmark with a total trade value of $7.8M across 6 products in 5 therapeutic categories. Based on 176 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Unicef Supply Division, sources from 2 verified Indian suppliers, with Mepro Pharmaceuticals Private Limited accounting for 94.3% of imports.
Unicef Supply Division, — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Unicef Supply Division,?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Mepro Pharmaceuticals Private Limited | $2.0M | 82 | 94.3% |
| Macleods Pharmaceuticals Limited | $124.0K | 8 | 5.7% |
Unicef Supply Division, sources from 2 verified Indian suppliers across 54 distinct formulations. The sourcing is highly concentrated — Mepro Pharmaceuticals Private Limited accounts for 94.3% of total imports, indicating a strategic single-source relationship.
What Formulations Does Unicef Supply Division, Import?
| Formulation | Value | Ships |
|---|---|---|
| Pharmaceutical products: albendazole | $308.9K | 7 |
| Pharmaceutical products: albendazole chewable tablets 400MG. (unicef stock numbers s1555370) | $108.8K | 3 |
| Pharmaceutical products: ciprofloxacin | $101.9K | 5 |
| Pharmaceutical products: albendazole chewable tablets 400MG.(unicef stock numbers s1555370) | $100.0K | 2 |
| Pharmaceutical products: albendazole chewable tablets 400MG. (unicef stock numbers s1555370) | $94.0K | 3 |
| Pharmaceutical products | $73.7K | 3 |
| Medicines. dolutegravir sodium 10MG | $72.0K | 4 |
| Pharmaceutical products: ibuprofen | $66.6K | 2 |
| Pharmaceutical products: miconazole | $64.9K | 4 |
| Pharmaceutical products: ciprofloxacin tablets BP 500 MG.(unicef stock numbers s1555710) | $57.6K | 3 |
| Pharmaceutical products: azithromycin | $50.0K | 1 |
| Pharmaceutical products: albendazole chewable tablets 400MG. (unicef stock numbers s1555370) qty | $50.0K | 1 |
| Pharmaceutical products: albendazole chewable tablets 400MG. (unicef stock numbers s1555370) | $50.0K | 1 |
| Pharmaceutical products: albendazole chewable tablets 400MG. (unicef stock numbers s1555370) | $50.0K | 1 |
| Pharmaceutical products: azithromycin tablets USP 250 MG.(unicef stock numbers s1531009) qty:132920x1x6s | $50.0K | 1 |
Unicef Supply Division, imports 54 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Unicef Supply Division, Import?
Top Products by Import Value
Unicef Supply Division, Therapeutic Categories — 5 Specializations
Unicef Supply Division, imports across 5 therapeutic categories, with Other (63.2%), Antimalarial & Antiparasitic (22.3%), Vitamins & Supplements (7.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 99% of total imports.
Other
1 products · 63.2% · $5.0M
Antimalarial & Antiparasitic
1 products · 22.3% · $1.8M
Vitamins & Supplements
1 products · 7.0% · $550.0K
Antifungals
1 products · 4.9% · $383.5K
Antibiotics
2 products · 2.6% · $200.0K
Import Portfolio — Top 6 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Vaccine | Other | $5.0M | 99 | 0.5% | 2 |
| 2 | Albendazole | Antimalarial & Antiparasitic | $1.8M | 35 | 1.6% | 4 |
| 3 | Zinc | Vitamins & Supplements | $550.0K | 11 | 0.7% | 17 |
| 4 | Miconazole | Antifungals | $383.5K | 27 | 1.1% | 11 |
| 5 | Erythromycin | Antibiotics | $100.0K | 2 | 0.1% | 16 |
| 6 | Tetracycline | Antibiotics | $100.0K | 2 | 0.6% | 4 |
Unicef Supply Division, imports 6 pharmaceutical products across 5 categories into Denmark totaling $7.8M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Unicef Supply Division,.
Request DemoUnicef Supply Division, — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
The UNICEF Supply Division is a specialized branch of the United Nations Children's Fund (UNICEF), dedicated to procuring and delivering essential supplies to support UNICEF's global programs. Headquartered in Copenhagen, Denmark, the Supply Division plays a pivotal role in ensuring that UNICEF's humanitarian and development initiatives are equipped with the necessary resources, including pharmaceuticals, medical equipment, and educational materials. As a government procurement entity, it operates under the auspices of the United Nations, focusing on the efficient and ethical sourcing of products to meet the needs of children and communities worldwide.
In Denmark, the UNICEF Supply Division serves as a central hub for the organization's procurement activities, coordinating with various suppliers and stakeholders to maintain a steady supply chain. Its operations are integral to UNICEF's mission, emphasizing quality assurance, cost-effectiveness, and timely delivery of goods and services. The Division's activities are guided by international standards and best practices, ensuring that all procured items meet the stringent requirements necessary for their intended use in diverse and often challenging environments.
2Distribution Network
The UNICEF Supply Division's distribution network is strategically designed to ensure the timely and efficient delivery of supplies to UNICEF's global programs. While specific warehouse locations and logistics capabilities are not publicly detailed, the Division operates a centralized warehouse in Copenhagen, Denmark, which serves as a primary distribution point. From this hub, supplies are dispatched to various UNICEF country offices and partners worldwide, facilitating rapid response to emergencies and ongoing development projects.
The Division's logistics operations are characterized by a commitment to quality assurance and compliance with international standards. This includes adherence to Good Distribution Practice (GDP) guidelines, ensuring that all products, particularly pharmaceuticals, are stored and transported under conditions that maintain their integrity and efficacy. The network's geographic coverage spans across all UNICEF program countries, enabling the organization to meet the diverse needs of children and communities in both developed and developing regions.
3Industry Role
Within Denmark's pharmaceutical supply chain, the UNICEF Supply Division functions primarily as a government procurement entity. It is not a wholesaler, distributor, hospital supplier, or logistics intermediary in the traditional commercial sense. Instead, its role is to source and procure pharmaceutical products and other essential supplies to support UNICEF's global programs. This includes engaging with various suppliers, including those in Denmark and internationally, to ensure the availability of high-quality products that meet UNICEF's stringent standards.
The Division's procurement activities are guided by principles of transparency, fairness, and efficiency, aligning with UNICEF's overall mission to uphold the rights of every child. By leveraging its position within the United Nations system, the Supply Division can negotiate favorable terms and conditions with suppliers, ensuring that resources are utilized effectively to maximize impact. Its operations are integral to the broader pharmaceutical supply chain in Denmark, contributing to the country's role in global humanitarian efforts.
Supplier Relationship Intelligence — Unicef Supply Division,
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
The UNICEF Supply Division's sourcing strategy from India reveals a high degree of concentration, with a total import value of $7.8 million USD over 176 shipments, encompassing six products across five therapeutic categories. The top five products imported from India include vaccines ($5.0 million, 0.5% share), albendazole ($1.8 million, 1.6% share), zinc ($550,000, 0.7% share), miconazole ($384,000, 1.1% share), and erythromycin ($100,000, 0.1% share). Notably, the top five products account for 98.7% of the total import value, indicating a highly concentrated product portfolio.
This concentration suggests a strategic sourcing approach, focusing on a select range of essential pharmaceutical products that are critical to UNICEF's health programs. The primary supplier, MEPRO Pharmaceuticals Private Limited, accounts for 94.3% of the shipments, indicating a strong and stable relationship. The secondary supplier, Macleods Pharmaceuticals Limited, contributes 5.7% of the shipments, suggesting a supplementary role in the supply chain. The limited number of suppliers and the high concentration of imports from India may pose risks related to supply chain disruptions or geopolitical factors affecting trade relations.
However, the strategic choice to concentrate sourcing from a select number of suppliers can also lead to benefits such as streamlined procurement processes, better negotiation leverage, and consistency in product quality. The stability of the relationship with MEPRO Pharmaceuticals Private Limited, as evidenced by the volume of shipments, indicates a reliable supply chain. Nonetheless, it is advisable for the UNICEF Supply Division to periodically assess and diversify its supplier base to mitigate potential risks associated with over-reliance on a limited number of suppliers.
2Supply Chain Resilience
The resilience of the UNICEF Supply Division's Indian supply chain is a critical factor in ensuring the uninterrupted delivery of essential pharmaceutical products. The Division's sourcing strategy from India is characterized by a high concentration of imports from a limited number of suppliers, with MEPRO Pharmaceuticals Private Limited accounting for 94.3% of the shipments. This concentration can enhance supply chain efficiency and consistency but also introduces potential vulnerabilities.
To bolster supply chain resilience, it is essential for the UNICEF Supply Division to establish contingency plans, including identifying alternative suppliers and maintaining strategic stockpiles of critical products. Diversifying the supplier base can mitigate risks associated with geopolitical tensions, natural disasters, or other disruptions that may affect the primary supplier. Additionally, ensuring that all suppliers adhere to international quality standards and regulatory requirements is crucial for maintaining the integrity and safety of the pharmaceutical products.
Regular audits and assessments of the supply chain, along with fostering strong relationships with multiple suppliers, can further enhance resilience. By proactively addressing potential risks and implementing robust supply chain management practices, the UNICEF Supply Division can ensure a steady and reliable supply of essential pharmaceuticals to support its global health initiatives.
3Strategic Implications
The UNICEF Supply Division's concentrated sourcing pattern from India, particularly its reliance on MEPRO Pharmaceuticals Private Limited, has significant strategic implications. This approach allows for streamlined procurement processes, potential cost efficiencies, and consistency in product quality, which are advantageous for UNICEF's operations. However, the high dependency on a limited number of suppliers also exposes the organization to risks related to supply chain disruptions, geopolitical factors, and changes in trade policies.
For Indian exporters seeking to become alternative suppliers to the UNICEF Supply Division, understanding the organization's procurement criteria and quality standards is essential. Demonstrating compliance with international regulations, such as Good Manufacturing Practice (GMP), and ensuring the ability to meet UNICEF's specific product requirements can enhance the prospects of establishing a partnership. Additionally, offering competitive pricing, reliable delivery schedules, and a commitment to ethical business practices are critical factors that can influence the UNICEF Supply Division's sourcing decisions.
Importing Pharmaceuticals into Denmark — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Denmark
1Regulatory Authority & Framework
In Denmark, the primary regulatory authority overseeing the import and distribution of pharmaceutical products is the Danish Medicines Agency (Lægemiddelstyrelsen). This agency is responsible for ensuring that medicinal products available in Denmark are safe, effective, and of high quality. The Danish Medicines Agency operates under the Danish Medicines Act, which outlines the legal framework for the manufacture, import, export, storage, and distribution of medicinal products. According to Section 39 of the Danish Medicines Act, any company involved in these activities must obtain authorization from the Danish Medicines Agency. (wipolex-res.wipo.int)
The marketing authorization pathway for pharmaceutical products in Denmark involves several key steps. Initially, a company must submit an application to the Danish Medicines Agency, providing comprehensive information about the medicinal product, including its composition, manufacturing process, and intended use. The agency evaluates the application to ensure that the product meets the necessary safety, efficacy, and quality standards. If the product complies with these requirements, the Danish Medicines Agency grants a marketing authorization, allowing the product to be legally marketed and distributed within Denmark. This process is aligned with European Union regulations, ensuring consistency and harmonization across member states. (wipolex-res.wipo.int)
2Import Licensing & GMP
To import pharmaceutical products into Denmark, companies must obtain a Manufacturing and Importation Authorization (MIA) from the Danish Medicines Agency. This authorization is granted under Section 39 of the Danish Medicines Act and is mandatory for any company involved in the import, manufacture, or distribution of medicinal products. The application process for an MIA requires detailed information about the company's operations, including premises, technical equipment, and the specific medicinal products intended for import. Additionally, companies must demonstrate compliance with Good Manufacturing Practice (GMP) standards, which are essential for ensuring the quality and safety of pharmaceutical products. The Danish Medicines Agency conducts inspections to verify adherence to GMP guidelines, and mutual recognition agreements with other countries facilitate the acceptance of GMP certifications from recognized authorities. (laegemiddelstyrelsen.dk)
3Quality & Labeling
Pharmaceutical products imported into Denmark must undergo batch testing to verify their quality, safety, and efficacy. This testing ensures that each batch meets the required standards before it is released for distribution. Stability requirements are also critical, as they determine the shelf-life of the product and ensure that it remains effective throughout its intended use period. Labeling requirements stipulate that all information on the product's packaging must be accurate, clear, and in compliance with Danish regulations.
Frequently Asked Questions — Unicef Supply Division,
What products does Unicef Supply Division, import from India?
Unicef Supply Division, imports 6 pharmaceutical products across 5 categories. Top imports: Vaccine ($5.0M), Albendazole ($1.8M), Zinc ($550.0K), Miconazole ($383.5K), Erythromycin ($100.0K).
Who supplies pharmaceuticals to Unicef Supply Division, from India?
Unicef Supply Division, sources from 2 verified Indian suppliers. The primary supplier is Mepro Pharmaceuticals Private Limited (94.3% of imports, $2.0M).
What is Unicef Supply Division,'s total pharmaceutical import value?
Unicef Supply Division,'s total pharmaceutical import value from India is $7.8M, based on 176 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Unicef Supply Division, focus on?
Unicef Supply Division, imports across 5 categories. The largest: Other (63.2%), Antimalarial & Antiparasitic (22.3%), Vitamins & Supplements (7.0%).
Get Full Unicef Supply Division, Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Unicef Supply Division, identified across shipments using consignee name normalization, aggregating 3 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Unicef Supply Division,'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 176 individual customs records matching Unicef Supply Division,.
- 5.Supplier Verification: Unicef Supply Division, sources from 2 verified Indian suppliers across 54 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
6 Products Tracked
5 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 3 company name variants from customs records. For current shipment-level data, contact TransData Nexus.