SUN Pharmaceutical Industries, Inc
Pharmaceutical Importer · United States · Respiratory Focus · $33.2M Total Trade · DGFT Verified
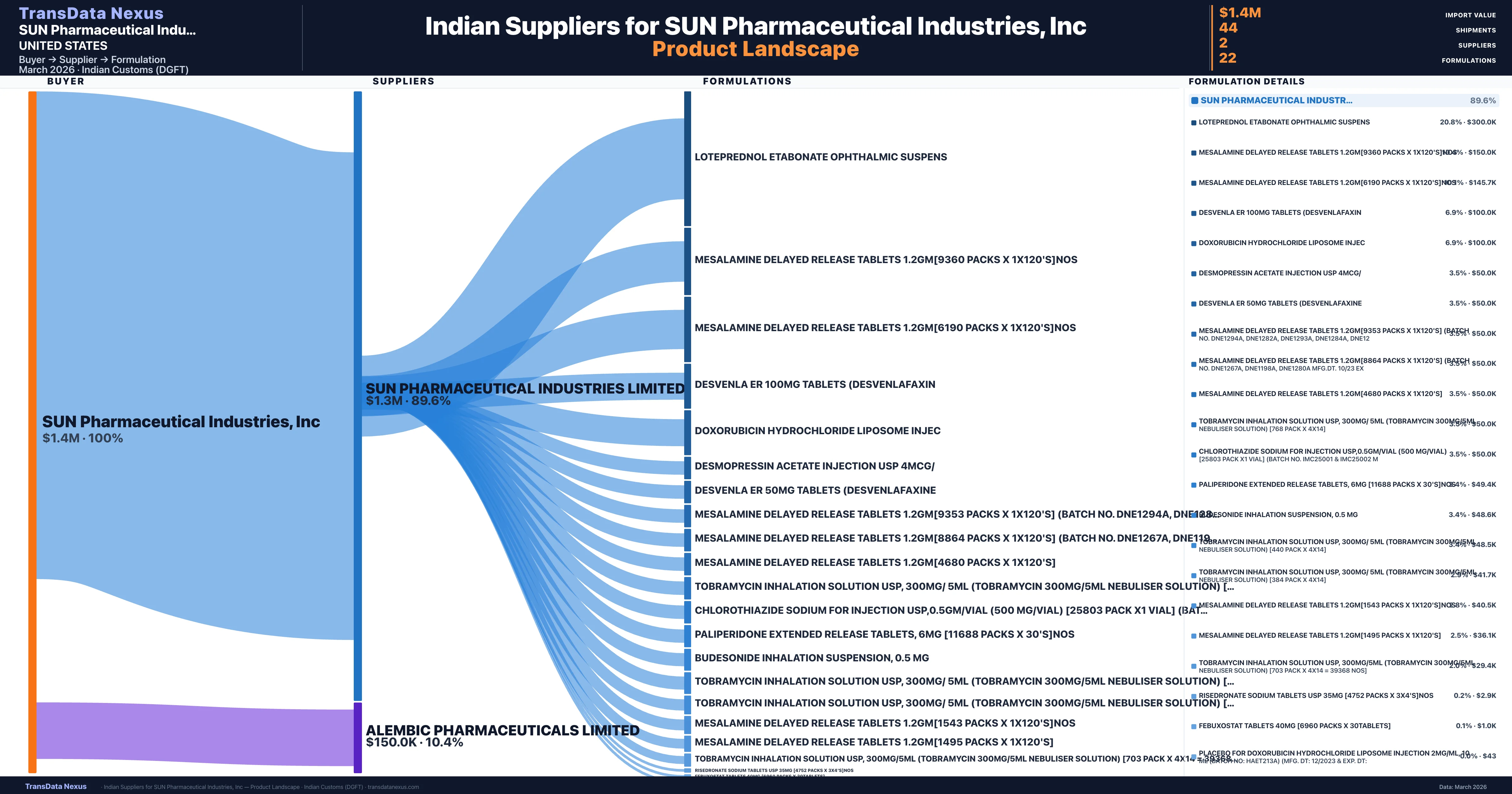
SUN Pharmaceutical Industries, Inc is a pharmaceutical importer based in United States with a total trade value of $33.2M across 19 products in 10 therapeutic categories. Based on 866 verified import shipments from Indian Customs (DGFT) records, SUN Pharmaceutical Industries, Inc is the #1 buyer in 1 product including Lenalidomide. SUN Pharmaceutical Industries, Inc sources from 2 verified Indian suppliers, with Sun Pharmaceutical Industries Limited accounting for 89.6% of imports.
SUN Pharmaceutical Industries, Inc — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to SUN Pharmaceutical Industries, Inc?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Sun Pharmaceutical Industries Limited | $1.3M | 41 | 89.6% |
| Alembic Pharmaceuticals Limited | $150.0K | 3 | 10.4% |
SUN Pharmaceutical Industries, Inc sources from 2 verified Indian suppliers across 22 distinct formulations. The sourcing is highly concentrated — Sun Pharmaceutical Industries Limited accounts for 89.6% of total imports, indicating a strategic single-source relationship.
What Formulations Does SUN Pharmaceutical Industries, Inc Import?
| Formulation | Value | Ships |
|---|---|---|
| Loteprednol etabonate ophthalmic | $300.0K | 6 |
| Mesalamine delayed release tablets 1.2GM[9360 packs x | $150.0K | 3 |
| Mesalamine delayed release tablets 1.2GM[6190 packs x | $145.7K | 3 |
| Desvenla er 100MG tablets | $100.0K | 2 |
| Doxorubicin hydrochloride liposome | $100.0K | 2 |
| Desmopressin acetate injection USP | $50.0K | 1 |
| Desvenla er 50MG tablets | $50.0K | 1 |
| Mesalamine delayed release tablets 1.2GM[9353 packs x 1x120's] (batch no. dne1294a, dne1282a, dne1293a, dne1284a | $50.0K | 1 |
| Mesalamine delayed release tablets 1.2GM[8864 packs x 1x120's] (batch no. dne1267a, dne1198a, dne1280a mfg.dt. 10/23 | $50.0K | 1 |
| Mesalamine delayed release tablets 1.2GM[4680 packs x | $50.0K | 1 |
| Tobramycin inhalation solution USP, 300MG/ 5ML (tobramycin 300MG/5ML nebuliser solution) [768 Pack x | $50.0K | 1 |
| Chlorothiazide sodium for injection USP,0.5GM/vial (500 MG/vial) [25803 Pack x1 vial] (batch no. imc25001 & imc25002 | $50.0K | 1 |
| Paliperidone extended release tablets, 6MG [11688 packs x | $49.4K | 3 |
| Budesonide inhalation suspension, 0.5 MG | $48.6K | 2 |
| Tobramycin inhalation solution USP, 300MG/ 5ML (tobramycin 300MG/5ML nebuliser solution) [440 Pack x | $48.5K | 1 |
SUN Pharmaceutical Industries, Inc imports 22 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does SUN Pharmaceutical Industries, Inc Import?
SUN Pharmaceutical Industries, Inc Therapeutic Categories — 10 Specializations
SUN Pharmaceutical Industries, Inc imports across 10 therapeutic categories, with Respiratory (26.6%), Gastrointestinal (19.3%), Advanced Oncology (13.9%) representing the largest segments. The portfolio is concentrated — top 5 products = 56% of total imports.
Respiratory
3 products · 26.6% · $8.8M
Gastrointestinal
1 products · 19.3% · $6.4M
Advanced Oncology
4 products · 13.9% · $4.6M
Antibiotics
2 products · 8.7% · $2.9M
Oncology
1 products · 8.0% · $2.6M
Antifungals
1 products · 7.2% · $2.4M
Corticosteroids
1 products · 6.3% · $2.1M
Advanced Antibiotics
3 products · 5.5% · $1.8M
Cardiovascular
2 products · 2.4% · $796.3K
Import Portfolio — Top 19 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Mesalamine | Gastrointestinal | $6.4M | 128 | 0.2% | 6 |
| 2 | Budesonide | Respiratory | $4.4M | 101 | 1.1% | 6 |
| 3 | Doxorubicin | Oncology | $2.6M | 53 | 1.3% | 3 |
| 4 | Ipratropium | Respiratory | $2.5M | 163 | 3.0% | 3 |
| 5 | Amphotericin | Antifungals | $2.4M | 48 | 1.0% | 2 |
| 6 | Ciprofloxacin | Antibiotics | $2.1M | 42 | 0.4% | 6 |
| 7 | Dexamethasone | Corticosteroids | $2.1M | 42 | 0.7% | 4 |
| 8 | Sunitinib | Advanced Oncology | $2.1M | 42 | 2.3% | 2 |
| 9 | Formoterol | Respiratory | $1.9M | 37 | 1.9% | 3 |
| 10 | Lenalidomide | Advanced Oncology | $1.2M | 24 | 0.0% | 1 |
| 11 | Ofloxacin | Advanced Antibiotics | $950.0K | 19 | 0.1% | 11 |
| 12 | Imatinib | Advanced Oncology | $871.0K | 26 | 1.6% | 15 |
| 13 | Tobramycin | Antibiotics | $800.0K | 16 | 1.4% | 6 |
| 14 | Niacin | Lipid & Metabolism | $663.1K | 47 | 8.6% | 4 |
| 15 | Nitrofurantoin | Advanced Antibiotics | $646.8K | 14 | 1.9% | 9 |
| 16 | Pazopanib | Advanced Oncology | $450.0K | 9 | 3.6% | 2 |
| 17 | Digoxin | Cardiovascular | $438.4K | 39 | 11.9% | 2 |
| 18 | Diltiazem | Cardiovascular | $357.9K | 11 | 2.0% | 15 |
| 19 | Minocycline | Advanced Antibiotics | $234.9K | 5 | 1.0% | 16 |
SUN Pharmaceutical Industries, Inc imports 19 pharmaceutical products across 10 categories into United States totaling $33.2M. The company is the #1 buyer for 1 product: Lenalidomide.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for SUN Pharmaceutical Industries, Inc.
Request DemoSUN Pharmaceutical Industries, Inc — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Sun Pharmaceutical Industries, Inc. is a prominent pharmaceutical company operating in the United States, specializing in the importation and distribution of finished pharmaceutical formulations. Headquartered in Cranbury, New Jersey, the company serves as a key player in the U.S. pharmaceutical supply chain, focusing on delivering a diverse range of high-quality medicines to meet various therapeutic needs. As a subsidiary of Sun Pharmaceutical Industries Limited, headquartered in Mumbai, India, Sun Pharmaceutical Industries, Inc. leverages its parent company's extensive global network and manufacturing capabilities to source and supply pharmaceutical products to the U.S. market. The company's role in the U.S. pharmaceutical distribution landscape is significant, as it collaborates with healthcare professionals, wholesalers, and distributors to ensure the availability of essential medications across the country.
2Distribution Network
Sun Pharmaceutical Industries, Inc. maintains a robust distribution network within the United States, with facilities strategically located to optimize logistics and ensure timely delivery of pharmaceutical products. The company's distribution center in Cranbury, New Jersey, serves as a central hub, facilitating efficient warehousing and distribution operations. Additionally, Sun Pharma has established distribution and customer service teams at multiple locations across the country, enhancing its ability to serve a wide geographic area effectively. This extensive network enables the company to reach various regions, ensuring that healthcare providers and patients have access to the necessary medications. Beyond the U.S., Sun Pharma's global presence spans over 100 countries, supported by 41 manufacturing facilities across five continents, reflecting its commitment to delivering high-quality, affordable medicines worldwide.
3Industry Role
In the United States pharmaceutical supply chain, Sun Pharmaceutical Industries, Inc. functions primarily as a pharmaceutical importer and distributor. The company sources finished pharmaceutical formulations from its parent company, Sun Pharmaceutical Industries Limited, and other suppliers, importing these products into the U.S. market. Once imported, Sun Pharma distributes these medications to various stakeholders, including wholesalers, healthcare providers, and pharmacies, ensuring that a diverse range of pharmaceutical products is accessible to meet the needs of patients and healthcare professionals. This role is crucial in bridging the gap between international pharmaceutical manufacturing and the U.S. healthcare system, facilitating the availability of both generic and specialty medications.
Supplier Relationship Intelligence — SUN Pharmaceutical Industries, Inc
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Sun Pharmaceutical Industries, Inc. demonstrates a notable concentration in its sourcing strategy, primarily importing finished pharmaceutical formulations from two Indian suppliers: Sun Pharmaceutical Industries Limited and Alembic Pharmaceuticals Limited. The majority of shipments, accounting for 89.6% of the total, are sourced from Sun Pharmaceutical Industries Limited, amounting to $1.3 million across 41 shipments. Alembic Pharmaceuticals Limited contributes a smaller portion, with 3 shipments totaling $150,000, representing 10.4% of the total shipments. This sourcing pattern indicates a strong reliance on Sun Pharmaceutical Industries Limited, which may present both strategic advantages and potential risks.
The high dependency on a single supplier could lead to vulnerabilities in the supply chain, especially if disruptions occur within Sun Pharmaceutical Industries Limited. However, the established relationship and consistent supply from this supplier suggest a stable and reliable partnership. The limited engagement with Alembic Pharmaceuticals Limited may reflect strategic decisions to maintain quality control, negotiate favorable terms, or leverage existing relationships. Overall, while the sourcing concentration offers operational efficiencies, it also necessitates contingency planning to mitigate potential risks associated with supplier dependency.
2Supply Chain Resilience
The resilience of Sun Pharmaceutical Industries, Inc.'s supply chain is closely tied to the stability and reliability of its primary supplier, Sun Pharmaceutical Industries Limited. Given that 89.6% of shipments originate from this supplier, any disruptions in their operations could significantly impact Sun Pharma's ability to meet market demands. The limited number of suppliers, with only two verified sources, suggests a lack of diversification, which could pose challenges in maintaining a continuous supply of products.
To enhance supply chain resilience, it would be prudent for Sun Pharmaceutical Industries, Inc. to consider diversifying its supplier base and establishing relationships with additional manufacturers. This strategy could mitigate risks associated with over-reliance on a single supplier and provide alternative sources in case of unforeseen disruptions. Additionally, evaluating the regulatory compliance and manufacturing capabilities of potential suppliers is essential to ensure that they meet the stringent standards required for pharmaceutical products in the U.S. market.
3Strategic Implications
The current sourcing pattern of Sun Pharmaceutical Industries, Inc., characterized by a heavy reliance on Sun Pharmaceutical Industries Limited, has several strategic implications. For Sun Pharma, this concentration may lead to favorable terms and streamlined operations due to the established relationship with its parent company. However, it also exposes the company to risks associated with supply chain disruptions, regulatory changes, or operational challenges within the primary supplier.
For Indian exporters seeking to become alternative suppliers to Sun Pharmaceutical Industries, Inc., this presents an opportunity to diversify the company's supply chain and reduce dependency on a single source. By offering high-quality products that meet U.S. regulatory standards, Indian exporters can position themselves as viable partners. Building relationships with Sun Pharmaceutical Industries, Inc. could lead to increased market access and a more balanced distribution of supply sources, benefiting both parties through enhanced stability and flexibility in the supply chain.
Importing Pharmaceuticals into United States — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United States
1Regulatory Authority & Framework
In the United States, the Food and Drug Administration (FDA) serves as the primary regulatory authority overseeing the importation and distribution of pharmaceutical products. The FDA enforces the Federal Food, Drug, and Cosmetic Act (FD&C Act), which sets forth the legal framework for the safety, efficacy, and quality of drugs entering the U.S. market. This legislation outlines requirements for drug approval, manufacturing practices, labeling, and importation procedures. For Indian pharmaceutical companies exporting to the U.S., understanding and complying with the FDA's regulations is essential to ensure market access and avoid potential legal issues.
The FDA's regulatory framework includes specific pathways for the importation of foreign-manufactured drugs. Drugs imported into the U.S. must meet FDA standards for quality, safety, and effectiveness. The FDA reviews shipments of imported drugs to determine their admissibility, and a drug may be refused entry if it appears to be adulterated, misbranded, or unapproved. Additionally, the FDA examines and analyzes samples of imported drugs to ensure they comply with applicable standards and labeling requirements.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products entering the United States are governed by the FDA. All foreign drug establishments whose products are imported into the U.S. are required to register their establishment with the FDA and list all of their drug products in commercial distribution in the United States. This registration process ensures that the FDA is notified of all regulated products imported into the U.S. and facilitates the agency's oversight of these products.
Regarding Good Manufacturing Practice (GMP) certification, the FDA recognizes GMP standards from various international organizations, including the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Indian pharmaceutical manufacturers seeking to export to the U.S. should ensure that their facilities comply with these recognized GMP standards to meet FDA requirements. Additionally, obtaining wholesale distribution authorization is necessary for companies involved in the distribution of pharmaceutical products within the U.S. market.
3Quality & Labeling
Imported pharmaceutical products must adhere to FDA requirements for quality and labeling. Batch testing is essential to verify that each batch of a drug product meets the required specifications for identity, strength, quality, and purity. Stability studies are also necessary to determine the shelf life of the product and ensure its efficacy and safety over time. Labeling requirements include providing accurate and truthful information about the drug's ingredients, usage instructions, warnings, and storage conditions. All labeling must be in English and comply with FDA regulations to ensure that healthcare professionals and patients can make informed decisions regarding the use of the product.
Serialization mandates require that pharmaceutical products have unique identifiers to track and trace the product throughout the supply chain. This system enhances the ability to detect and prevent counterfeit drugs, ensuring the integrity of the pharmaceutical supply chain. Compliance with these quality and labeling requirements is crucial for the admissibility of imported pharmaceutical products into the U.S. market.
4Recent Regulatory Changes
Between 2024 and 2026, several policy changes have impacted the importation of Indian pharmaceutical products into the United States. The FDA has continued to enforce stringent regulations to ensure the safety and efficacy of imported drugs. This includes enhanced scrutiny of manufacturing practices, labeling accuracy, and compliance with GMP standards. Additionally, the FDA has implemented more rigorous inspection procedures and increased collaboration with international regulatory bodies to monitor and enforce compliance among foreign manufacturers. These measures aim to protect public health by ensuring that all pharmaceutical products entering the U.S. market meet the highest standards of quality and safety.
SUN Pharmaceutical Industries, Inc — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Sun Pharmaceutical Industries, Inc. focuses its product strategy on importing a diverse range of finished pharmaceutical formulations across multiple therapeutic areas. The company's top five imported products include Mesalamine, Budeson
Frequently Asked Questions — SUN Pharmaceutical Industries, Inc
What products does SUN Pharmaceutical Industries, Inc import from India?
SUN Pharmaceutical Industries, Inc imports 19 pharmaceutical products across 10 categories. Top imports: Mesalamine ($6.4M), Budesonide ($4.4M), Doxorubicin ($2.6M), Ipratropium ($2.5M), Amphotericin ($2.4M).
Who supplies pharmaceuticals to SUN Pharmaceutical Industries, Inc from India?
SUN Pharmaceutical Industries, Inc sources from 2 verified Indian suppliers. The primary supplier is Sun Pharmaceutical Industries Limited (89.6% of imports, $1.3M).
What is SUN Pharmaceutical Industries, Inc's total pharmaceutical import value?
SUN Pharmaceutical Industries, Inc's total pharmaceutical import value from India is $33.2M, based on 866 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does SUN Pharmaceutical Industries, Inc focus on?
SUN Pharmaceutical Industries, Inc imports across 10 categories. The largest: Respiratory (26.6%), Gastrointestinal (19.3%), Advanced Oncology (13.9%).
Get Full SUN Pharmaceutical Industries, Inc Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: SUN Pharmaceutical Industries, Inc identified across shipments using consignee name normalization, aggregating 9 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as SUN Pharmaceutical Industries, Inc's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 866 individual customs records matching SUN Pharmaceutical Industries, Inc.
- 5.Supplier Verification: SUN Pharmaceutical Industries, Inc sources from 2 verified Indian suppliers across 22 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
19 Products Tracked
10 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 9 company name variants from customs records. For current shipment-level data, contact TransData Nexus.