State Trading Organization PLC
Pharmaceutical Importer · Maldives · Advanced Oncology Focus · $461.8K Total Trade · DGFT Verified
State Trading Organization PLC is a pharmaceutical importer based in Maldives with a total trade value of $461.8K across 5 products in 4 therapeutic categories. Based on 28 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. State Trading Organization PLC sources from 36 verified Indian suppliers, with Medley Pharmaceuticals Limited accounting for 23.2% of imports.
State Trading Organization PLC — Import Portfolio & Supplier Network
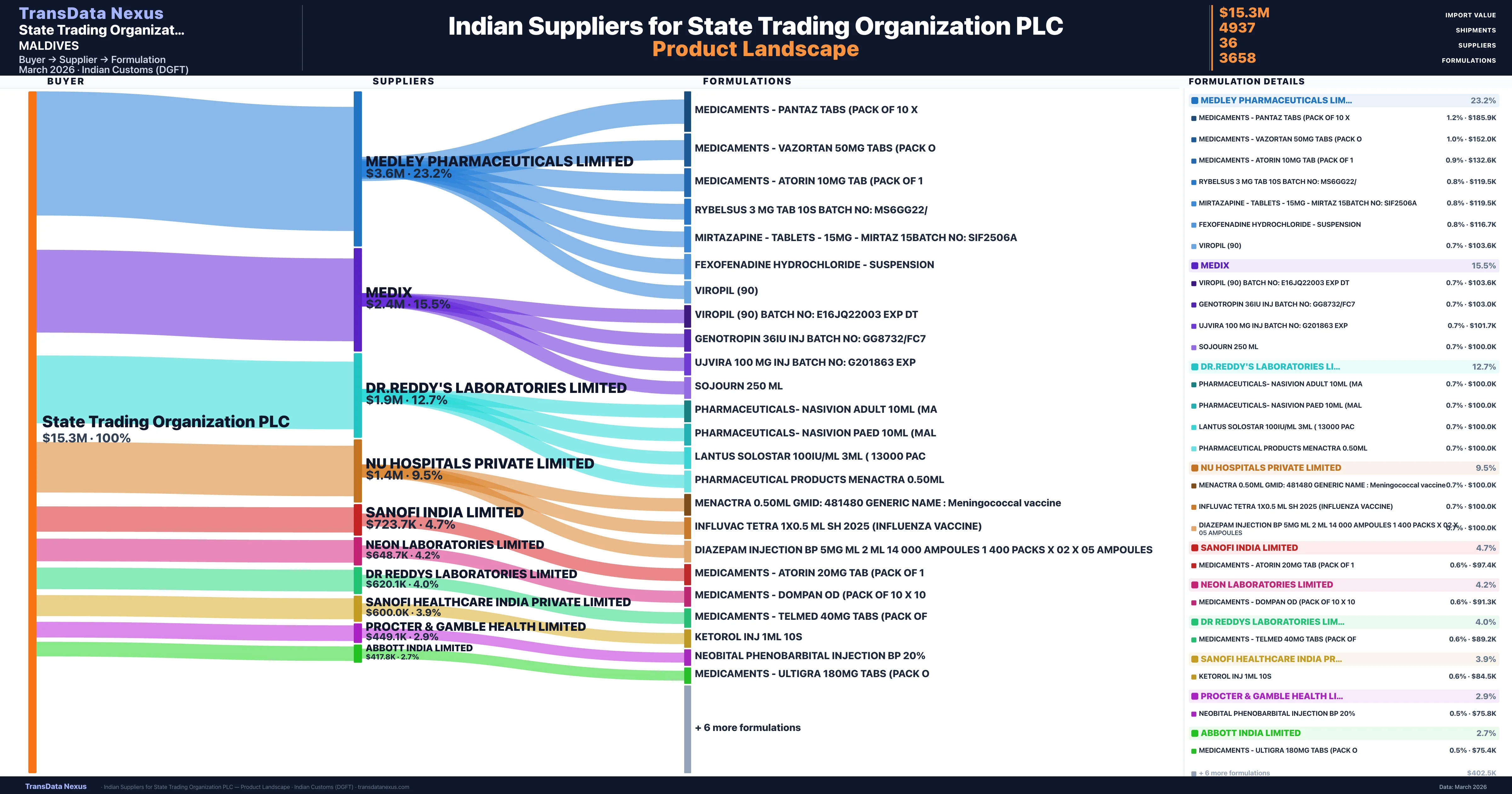
Who Are the Verified Indian Suppliers to State Trading Organization PLC?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Medley Pharmaceuticals Limited | $3.6M | 595 | 23.2% |
| Medix | $2.4M | 1,906 | 15.5% |
| Dr.reddy's Laboratories Limited | $1.9M | 127 | 12.7% |
| Nu Hospitals Private Limited | $1.4M | 545 | 9.5% |
| Sanofi India Limited | $723.7K | 66 | 4.7% |
| Neon Laboratories Limited | $648.7K | 26 | 4.2% |
| Dr Reddys Laboratories Limited | $620.1K | 35 | 4.0% |
| Sanofi Healthcare India Private Limited | $600.0K | 12 | 3.9% |
| Procter & Gamble Health Limited | $449.1K | 20 | 2.9% |
| Abbott India Limited | $417.8K | 49 | 2.7% |
| Paragon Diagnostics | $404.0K | 398 | 2.6% |
| Dr Reddy S Laboratories Limited | $337.1K | 17 | 2.2% |
| Darius Healthcare And Management Services Private | $284.3K | 197 | 1.9% |
| Fresenius Kabi India Private Limited | $264.8K | 29 | 1.7% |
| Rajagiri Health Care And Education Trust | $218.9K | 156 | 1.4% |
| Raman & Weil Private Limited | $155.4K | 6 | 1.0% |
| Miot Medical Supplies Private Limited | $146.3K | 114 | 1.0% |
| Citro Pharma Private Limited | $132.7K | 29 | 0.9% |
| Cipla Limited | $108.3K | 38 | 0.7% |
| Procter Gamble Health Limited | $99.7K | 3 | 0.7% |
| Mc Pharma Distributors | $69.2K | 338 | 0.5% |
| Rusan Pharma Limited | $66.9K | 13 | 0.4% |
| Medicom Superdrugs Pharmacy Private Limited | $58.1K | 35 | 0.4% |
| Medi Inova | $55.0K | 27 | 0.4% |
| Sanofi Consumer Healthcare India Limited | $50.6K | 2 | 0.3% |
| M C Exports | $17.8K | 81 | 0.1% |
| Universal Multi Traders | $17.5K | 8 | 0.1% |
| Kyodo International Private Limited | $15.3K | 3 | 0.1% |
| H Plus Pharma Llp | $13.5K | 14 | 0.1% |
| Nila Pharmaceuticals | $5.2K | 11 | 0.0% |
State Trading Organization PLC sources from 36 verified Indian suppliers across 3,658 distinct formulations. The supply base is diversified across 36 suppliers, reducing single-source dependency risk.
What Formulations Does State Trading Organization PLC Import?
| Formulation | Value | Ships |
|---|---|---|
| Medicaments - pantaz tabs (Pack of 10 | $185.9K | 9 |
| Medicaments - vazortan 50MG tabs (Pack | $152.0K | 9 |
| Medicaments - atorin 10MG TAB (Pack of | $132.6K | 6 |
| Rybelsus 3 MG TAB 10s batch no | $119.5K | 7 |
| Mirtazapine - tablets - 15MG - mirtaz 15batch no | $119.5K | 3 |
| Fexofenadine hydrochloride - | $116.7K | 4 |
| Viropil (90) | $103.6K | 3 |
| Viropil (90) batch no: e16jq22003 exp | $103.6K | 3 |
| Genotropin 36iu INJ batch no | $103.0K | 3 |
| Ujvira 100 MG INJ batch no: g201863 | $101.7K | 3 |
| Sojourn 250 ML | $100.0K | 2 |
| Pharmaceuticals- nasivion adult 10ML | $100.0K | 2 |
| Pharmaceuticals- nasivion paed 10ML | $100.0K | 2 |
| Lantus solostar 100iu/ML 3ML ( 13000 | $100.0K | 2 |
| Pharmaceutical products menactra 0.50ML | $100.0K | 2 |
State Trading Organization PLC imports 3,658 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does State Trading Organization PLC Import?
Top Products by Import Value
State Trading Organization PLC Therapeutic Categories — 4 Specializations
State Trading Organization PLC imports across 4 therapeutic categories, with Advanced Oncology (54.5%), CNS & Psychiatric (37.5%), Other (7.8%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Oncology
2 products · 54.5% · $251.5K
CNS & Psychiatric
1 products · 37.5% · $173.0K
Other
1 products · 7.8% · $36.1K
Brand Names & OTC Products
1 products · 0.2% · $1.1K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Bevacizumab | Advanced Oncology | $219.2K | 10 | 0.5% | 13 |
| 2 | Diazepam | CNS & Psychiatric | $173.0K | 4 | 2.3% | 7 |
| 3 | Nicotine | Other | $36.1K | 5 | 0.2% | 12 |
| 4 | Nivolumab | Advanced Oncology | $32.3K | 6 | 1.4% | 4 |
| 5 | Soframycin | Brand Names & OTC Products | $1.1K | 3 | 0.2% | 7 |
State Trading Organization PLC imports 5 pharmaceutical products across 4 categories into Maldives totaling $461.8K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for State Trading Organization PLC.
Request DemoState Trading Organization PLC — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
State Trading Organization PLC (STO) is a state-owned enterprise established in 1964 by the Maldivian government to strengthen national food security. Over the decades, STO has diversified its operations, becoming a leading importer and distributor of essential goods, including pharmaceuticals, medical supplies, fuel, construction materials, and consumer electronics. Headquartered in Malé, the capital of the Maldives, STO plays a pivotal role in the nation's supply chain, ensuring the availability of critical products across the archipelago. (sto.mv)
In the pharmaceutical sector, STO has been instrumental in importing and distributing a wide range of medicines to meet the healthcare needs of the Maldivian population. However, in September 2025, the Maldivian government established the State Pharmaceutical and Medical Supply Corporation Limited (SPMSCL) to exclusively handle the import and distribution of pharmaceuticals, medical consumables, and medical equipment. This strategic move aims to address longstanding challenges related to access to medicines and streamline the supply chain. Consequently, STO's pharmacy operations, encompassing over 190 outlets, were transferred to SPMSCL in early 2026.
2Distribution Network
STO's distribution network is extensive, with operations spanning the entire Maldives. Prior to the transfer of its pharmacy operations to SPMSCL, STO managed over 190 pharmacies nationwide, ensuring widespread access to essential medicines and consumer goods. The company's logistics capabilities include a fleet of vehicles and partnerships with shipping companies to facilitate the timely delivery of products to various islands. While STO's primary focus has been on domestic distribution, it has also engaged in international trade, with operations in Singapore, to source products and manage supply chains effectively. (sto.mv)
3Industry Role
Before the establishment of SPMSCL, STO served as a primary wholesaler and distributor in the Maldivian pharmaceutical supply chain. The company was responsible for importing a diverse range of medicines and medical supplies, which were then distributed through its network of pharmacies and other outlets. This role was crucial in ensuring the availability of essential healthcare products across the Maldives. With the formation of SPMSCL, STO's direct involvement in pharmaceutical distribution has been reduced, allowing SPMSCL to focus exclusively on the importation and distribution of medical supplies.
Supplier Relationship Intelligence — State Trading Organization PLC
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
STO's pharmaceutical imports from India have been substantial, with a total import value of $462,000 USD across 28 shipments from 2022 to 2026. The top five products imported include Bevacizumab ($219,000), Diazepam ($173,000), Nicotine ($36,000), Nivolumab ($32,000), and Soframycin ($1,000). These products span therapeutic categories such as Advanced Oncology, CNS & Psychiatric, and Other. The concentration of imports in these specific products indicates a strategic focus on high-demand medications within these therapeutic areas. The absence of a single dominant supplier suggests a diversified sourcing strategy, which can mitigate risks associated with supplier dependency. However, the high concentration in a few products also means that any disruptions in the supply of these items could significantly impact STO's operations.
2Supply Chain Resilience
STO's supply chain resilience is supported by its diversified supplier base, with 36 unique suppliers from India. This broad network helps mitigate risks associated with over-reliance on a single supplier. The company's focus on finished pharmaceutical formulations, rather than raw APIs or bulk drugs, indicates a preference for ready-to-use products, which can streamline distribution and reduce lead times. While the transfer of pharmacy operations to SPMSCL may affect STO's direct involvement in pharmaceutical distribution, the company's established relationships with suppliers and its experience in logistics provide a foundation for continued resilience in the supply chain.
3Strategic Implications
The establishment of SPMSCL and the transfer of STO's pharmacy operations represent a strategic shift in the Maldivian pharmaceutical supply chain. For STO, this transition allows the company to refocus on its core competencies in other sectors, such as fuel, construction materials, and consumer goods. For Indian exporters, the change presents an opportunity to engage directly with SPMSCL, which now holds the responsibility for pharmaceutical imports. Building relationships with SPMSCL could lead to new partnerships and expanded market access in the Maldives.
Importing Pharmaceuticals into Maldives — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Maldives
1Regulatory Authority & Framework
The Maldives' pharmaceutical sector is regulated by the Ministry of Health, which oversees the importation, distribution, and sale of medical products. The establishment of SPMSCL in September 2025 centralizes the import and distribution of pharmaceuticals, aiming to streamline operations and improve access to medicines. This move aligns with global best practices, where dedicated entities manage pharmaceutical supply chains to ensure efficiency and accountability.
2Import Licensing & GMP
Import licensing requirements for pharmaceuticals in the Maldives are managed by the Ministry of Health. Suppliers must obtain the necessary approvals to import medical products into the country. The Maldives recognizes Good Manufacturing Practice (GMP) certifications from reputable international bodies, including the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). These certifications ensure that imported medicines meet quality standards and are safe for use.
3Quality & Labeling
Imported pharmaceuticals must undergo batch testing to confirm their safety and efficacy. Stability requirements ensure that medicines maintain their effectiveness throughout their shelf life. Labeling must be in the official language of the Maldives, Dhivehi, and include essential information such as dosage instructions, expiration dates, and storage conditions. Serialization mandates may be implemented to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
In September 2025, the Maldivian government established the State Pharmaceutical and Medical Supply Corporation Limited (SPMSCL) to exclusively handle the import and distribution of pharmaceuticals. This restructuring aims to address challenges related to access to medicines and improve the efficiency of the supply chain. The transfer of STO's pharmacy operations to SPMSCL was completed by February 2026, marking a significant shift in the pharmaceutical distribution landscape in the Maldives.
State Trading Organization PLC — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
STO's focus on importing high-value pharmaceuticals such as Bevacizumab and Diazepam indicates a strategic emphasis on critical and high-demand medications. The concentration in therapeutic areas like Advanced Oncology and CNS & Psychiatric suggests a targeted approach to address prevalent health issues in the Maldives. This strategy aligns with the government's efforts to diversify the range of medicines available, expanding from around 200 to between 800 and 900 items, to strengthen the national healthcare system. (plus.mv)
2Sourcing Profile
STO's sourcing strategy involved importing finished pharmaceutical formulations from a diverse set of suppliers in India. This approach ensured a steady supply of ready-to-use medicines, facilitating efficient distribution. The company's previous role in managing a significant portion of the pharmaceutical supply chain in the Maldives underscores the importance of strategic sourcing in maintaining the availability of essential medicines.
3Market Positioning
Prior to the establishment of SPMSCL, STO served a broad segment of the Maldivian market, including retail pharmacies, hospitals, and government tenders. The company's extensive distribution network and diverse product portfolio enabled it to meet the needs of various healthcare providers and consumers across the nation.
Seller's Guide — How to Become a Supplier to State Trading Organization PLC
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
With the formation of SPMSCL, there is a clear opportunity for new Indian suppliers to engage directly with the government entity responsible for pharmaceutical imports. This shift presents a chance to establish partnerships and supply essential medicines to the Maldivian market. Identifying gaps in SPMSCL's current sourcing and offering competitive, high-quality products can position new suppliers favorably.
2Requirements & Qualifications
Indian exporters aiming to supply pharmaceuticals to the Maldives must ensure their products are registered with the Ministry of Health and comply with the country's import licensing requirements. Obtaining GMP certifications from recognized international bodies such as WHO or PIC/S is essential to meet quality standards. Additionally, adherence to labeling and packaging regulations, including language requirements and serialization mandates, is necessary for market entry.
3How to Approach
To establish a relationship with SPMSCL, Indian exporters should initiate contact through official channels, providing detailed product information and demonstrating compliance with Maldivian regulatory standards. Participating in government tenders and engaging in direct negotiations can facilitate partnerships. Understanding the regulatory filing process and preparing for potential audits or inspections will be crucial. Given the recent restructuring, timelines for approval and distribution may vary, so maintaining open communication with SPMSCL is advisable.
Frequently Asked Questions — State Trading Organization PLC
What products does State Trading Organization PLC import from India?
State Trading Organization PLC imports 5 pharmaceutical products across 4 categories. Top imports: Bevacizumab ($219.2K), Diazepam ($173.0K), Nicotine ($36.1K), Nivolumab ($32.3K), Soframycin ($1.1K).
Who supplies pharmaceuticals to State Trading Organization PLC from India?
State Trading Organization PLC sources from 36 verified Indian suppliers. The primary supplier is Medley Pharmaceuticals Limited (23.2% of imports, $3.6M).
What is State Trading Organization PLC's total pharmaceutical import value?
State Trading Organization PLC's total pharmaceutical import value from India is $461.8K, based on 28 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does State Trading Organization PLC focus on?
State Trading Organization PLC imports across 4 categories. The largest: Advanced Oncology (54.5%), CNS & Psychiatric (37.5%), Other (7.8%).
Get Full State Trading Organization PLC Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: State Trading Organization PLC identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as State Trading Organization PLC's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 28 individual customs records matching State Trading Organization PLC.
- 5.Supplier Verification: State Trading Organization PLC sources from 36 verified Indian suppliers across 3,658 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.