Sekretariat Ditjen Pencegahan DAN
Pharmaceutical Importer · Indonesia · Tuberculosis Medications Focus · $1.3M Total Trade · DGFT Verified
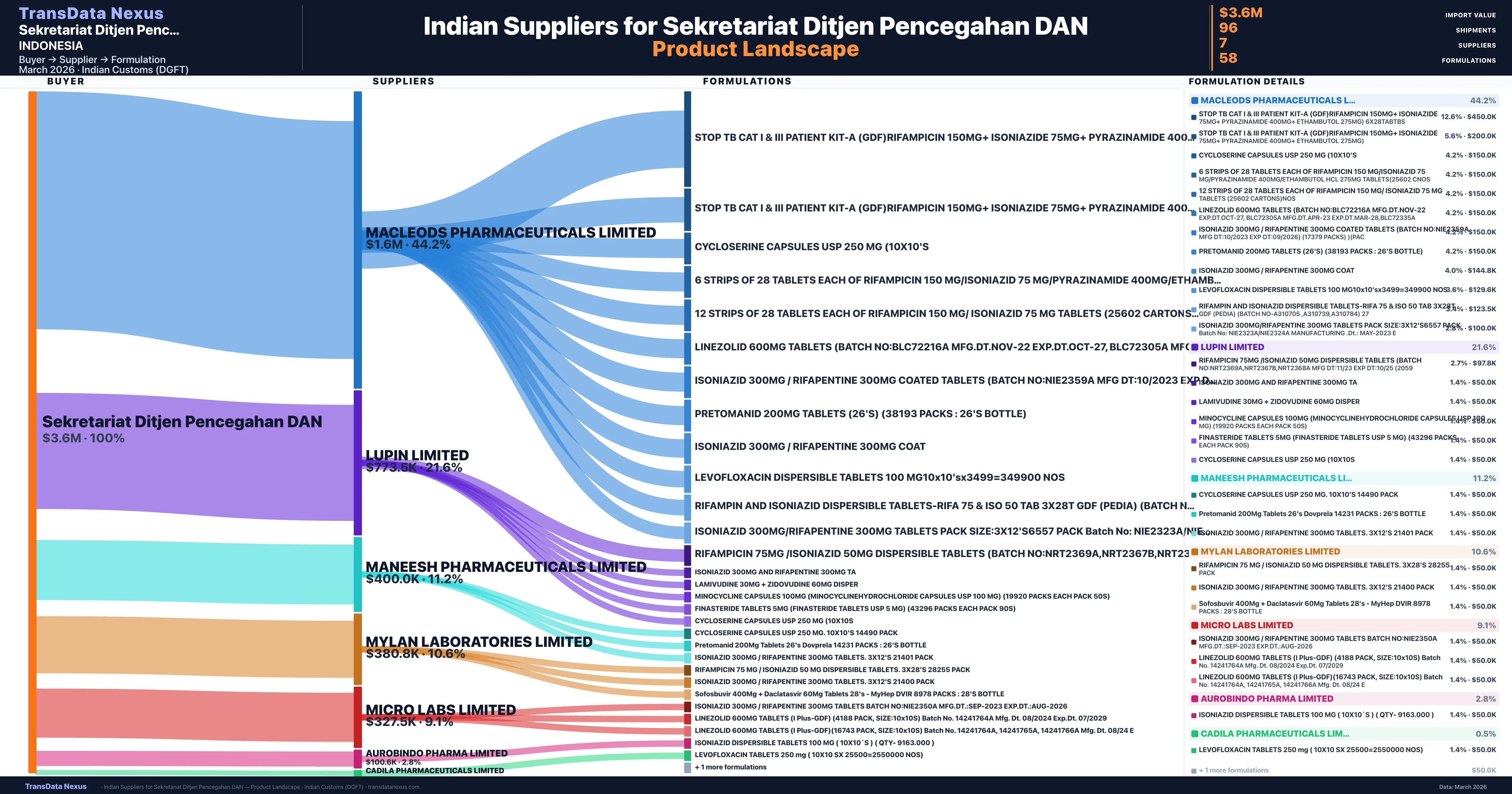
Sekretariat Ditjen Pencegahan DAN is a pharmaceutical importer based in Indonesia with a total trade value of $1.3M across 2 products in 2 therapeutic categories. Based on 25 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Sekretariat Ditjen Pencegahan DAN sources from 7 verified Indian suppliers, with Macleods Pharmaceuticals Limited accounting for 44.2% of imports.
Sekretariat Ditjen Pencegahan DAN — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Sekretariat Ditjen Pencegahan DAN?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Macleods Pharmaceuticals Limited | $1.6M | 45 | 44.2% |
| Lupin Limited | $773.5K | 18 | 21.6% |
| Maneesh Pharmaceuticals Limited | $400.0K | 10 | 11.2% |
| Mylan Laboratories Limited | $380.8K | 10 | 10.6% |
| Micro Labs Limited | $327.5K | 8 | 9.1% |
| Aurobindo Pharma Limited | $100.6K | 4 | 2.8% |
| Cadila Pharmaceuticals Limited | $17.0K | 1 | 0.5% |
Sekretariat Ditjen Pencegahan DAN sources from 7 verified Indian suppliers across 58 distinct formulations. The supply base is diversified across 7 suppliers, reducing single-source dependency risk.
What Formulations Does Sekretariat Ditjen Pencegahan DAN Import?
| Formulation | Value | Ships |
|---|---|---|
| Stop tb cat i & iii patient kit-a (gdf)rifampicin 150MG+ isoniazide 75MG+ pyrazinamide 400MG+ ethambutol 275MG) | $450.0K | 9 |
| Stop tb cat i & iii patient kit-a (gdf)rifampicin 150MG+ isoniazide 75MG+ pyrazinamide 400MG+ ethambutol 275MG) | $200.0K | 4 |
| Cycloserine capsules USP 250 MG | $150.0K | 3 |
| 6 strips of 28 tablets Each of rifampicin 150 MG/isoniazid 75 MG/pyrazinamide 400MG/ethambutol hcl 275MG tablets(25602 | $150.0K | 3 |
| 12 strips of 28 tablets Each of rifampicin 150 MG/ isoniazid 75 MG tablets (25602 cartons | $150.0K | 3 |
| Linezolid 600MG tablets (batch no:blc72216a mfg.dt.nov-22 exp.dt.oct-27, blc72305a mfg.dt.apr-23 exp.dt.mar-28 | $150.0K | 3 |
| Isoniazid 300MG / rifapentine 300MG coated tablets (batch no:nie2359a mfg dt:10/2023 exp dt:09/2026) (17379 packs) | $150.0K | 3 |
| Pretomanid 200MG tablets (26's) (38193 packs : 26's bottle) | $150.0K | 3 |
| Isoniazid 300MG / rifapentine 300MG | $144.8K | 3 |
| Levofloxacin dispersible tablets 100 mg10x10'sx3499=349900 | $129.6K | 3 |
| Rifampin and isoniazid dispersible tablets-rifa 75 & iso 50 TAB 3x28t gdf (pedia) (batch no-a310705 ,a310739,a310784) | $123.5K | 3 |
| Isoniazid 300MG/rifapentine 300MG tablets Pack size:3x12's6557 Pack batch no: nie2323a/nie2324a manufacturing .dt.: may-2023 | $100.0K | 2 |
| Rifampicin 75MG /isoniazid 50MG dispersible tablets (batch no:nrt2369a,nrt2367b,nrt2368a mfg dt:11/23 exp dt:10/25 | $97.8K | 3 |
| Isoniazid 300MG and rifapentine 300MG | $50.0K | 1 |
| Lamivudine 30MG + zidovudine 60MG | $50.0K | 1 |
Sekretariat Ditjen Pencegahan DAN imports 58 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Sekretariat Ditjen Pencegahan DAN Import?
Top Products by Import Value
Sekretariat Ditjen Pencegahan DAN Therapeutic Categories — 2 Specializations
Sekretariat Ditjen Pencegahan DAN imports across 2 therapeutic categories, with Tuberculosis Medications (84.0%), Advanced Antibiotics (16.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Tuberculosis Medications
1 products · 84.0% · $1.1M
Advanced Antibiotics
1 products · 16.0% · $200.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Isoniazid | Tuberculosis Medications | $1.1M | 21 | 0.0% | 14 |
| 2 | Linezolid | Advanced Antibiotics | $200.0K | 4 | 0.5% | 9 |
Sekretariat Ditjen Pencegahan DAN imports 2 pharmaceutical products across 2 categories into Indonesia totaling $1.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Sekretariat Ditjen Pencegahan DAN.
Request DemoSekretariat Ditjen Pencegahan DAN — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Sekretariat Ditjen Pencegahan dan Pengendalian Penyakit (Sekretariat Ditjen P2P) is the Secretariat of the Directorate General of Disease Prevention and Control within Indonesia's Ministry of Health. This governmental body is responsible for coordinating and supporting administrative functions related to disease prevention and control programs across the nation. Its primary role includes the development and implementation of health policies, management of disease surveillance systems, and coordination of public health initiatives. While Sekretariat Ditjen P2P does not function as a pharmaceutical wholesaler or distributor, it plays a pivotal role in the procurement and distribution of medical supplies and pharmaceuticals necessary for public health programs. This includes the importation of essential medicines to support national health objectives.
2Distribution Network
As a governmental entity, Sekretariat Ditjen P2P's distribution network is designed to ensure the efficient delivery of medical supplies and pharmaceuticals throughout Indonesia. While specific warehouse locations and logistics capabilities are not publicly detailed, the organization collaborates with various stakeholders, including local health authorities and logistics providers, to facilitate the nationwide distribution of medical products. This collaborative approach ensures that essential medicines reach both urban and remote areas, supporting the country's public health infrastructure.
3Industry Role
Sekretariat Ditjen P2P operates within Indonesia's pharmaceutical supply chain primarily as a government procurement and distribution entity. Its role is to source and import essential medicines, ensuring their availability for public health programs and initiatives. By managing the procurement process, Sekretariat Ditjen P2P ensures that the Indonesian government can provide necessary medical treatments to its population, particularly in areas where private sector distribution may be limited.
Supplier Relationship Intelligence — Sekretariat Ditjen Pencegahan DAN
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Sekretariat Ditjen P2P's sourcing strategy exhibits a high degree of concentration, with a portfolio focused on two primary products: Isoniazid and Linezolid. This concentrated approach suggests a strategic decision to prioritize these medications, likely due to their critical role in treating tuberculosis and other serious infections. The substantial import value of Isoniazid ($1.1 million) and Linezolid ($200,000) indicates a significant commitment to these therapies. However, this concentration also presents potential risks, such as supply chain disruptions or price fluctuations affecting these specific products. The reliance on a limited number of suppliers for these medications may impact the stability and resilience of the supply chain.
2Supply Chain Resilience
The resilience of Sekretariat Ditjen P2P's supply chain is influenced by its reliance on a select group of Indian pharmaceutical suppliers. The top suppliers include MACLEODS PHARMACEUTICALS LIMITED, LUPIN LIMITED, and MANEESH PHARMACEUTICALS LIMITED, among others. This limited supplier base could pose challenges in the event of supply disruptions or regulatory changes affecting these manufacturers. Additionally, the focus on a narrow range of formulations may limit the organization's ability to adapt to changing public health needs or emerging diseases. To enhance supply chain resilience, diversifying the supplier base and expanding the range of imported formulations could be beneficial.
3Strategic Implications
Sekretariat Ditjen P2P's concentrated sourcing strategy positions it to effectively meet the specific therapeutic needs of the Indonesian population, particularly in the treatment of tuberculosis and serious infections. This focus allows for streamlined procurement processes and potentially better pricing agreements with suppliers. However, the limited supplier base and product range may expose the organization to risks related to supply chain disruptions or changes in international trade policies. For Indian pharmaceutical exporters, this presents an opportunity to engage with a key government buyer by offering alternative products or formulations that align with Indonesia's public health priorities, thereby expanding their market presence.
Importing Pharmaceuticals into Indonesia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Indonesia
1Regulatory Authority & Framework
Indonesia's pharmaceutical industry is regulated by the Badan Pengawas Obat dan Makanan (BPOM), the National Agency of Drug and Food Control. BPOM oversees the registration, importation, and distribution of pharmaceutical products to ensure they meet safety, efficacy, and quality standards. The regulatory framework governing pharmaceutical imports includes the Decree of the Minister of Health No. 1010/08, which outlines the registration and import procedures for pharmaceutical products. Additionally, the Regulation of the President of the Republic of Indonesia No. 80/2017 grants BPOM the authority to supervise both pre-market and post-market processes of drugs and foods. These regulations are designed to protect public health by ensuring that all pharmaceutical products in the Indonesian market are safe and effective.
2Import Licensing & GMP
Importing pharmaceutical products into Indonesia requires compliance with specific licensing and certification requirements. Importers must obtain an import business license and, for certain products, an import certificate (Surat Keterangan Impor or SKI) issued by BPOM. Additionally, imported drugs must have a distribution license and comply with existing laws and regulations. The products must meet criteria such as proven efficacy and safety, quality standards in accordance with Good Manufacturing Practices (GMP), and accurate labeling. Parties applying for a distribution license must submit various documents, including administrative, quality, non-clinical, and clinical documents, to BPOM for evaluation.
3Quality & Labeling
Imported pharmaceutical products must adhere to stringent quality and labeling requirements to ensure consumer safety. Batch testing is mandatory to verify the quality and safety of the products. Stability requirements ensure that the products maintain their efficacy and safety throughout their shelf life. Labeling must be in the Indonesian language and provide objective, non-misleading information to ensure appropriate and safe use of the drugs. Additionally, serialization mandates may be implemented to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, Indonesia has implemented several regulatory changes affecting pharmaceutical imports. In October 2023, the government announced restrictions on certain imported commodities, including traditional medicines and health supplements, to protect local industries and consumers. This move aims to reduce the influx of imported products that could disadvantage local producers. Additionally, in June 2025, the Ministry of Trade issued Regulation No. 20/2025, establishing import controls for chemicals, hazardous substances, and certain minerals, which may impact the importation of pharmaceutical raw materials and finished products. These regulatory changes underscore the government's focus on strengthening domestic industries and ensuring the safety and quality of imported pharmaceutical products. (setkab.go.id)
Sekretariat Ditjen Pencegahan DAN — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Sekretariat Ditjen P2P's product strategy focuses on importing essential medicines critical for public health programs, particularly in the treatment of tuberculosis and serious infections. The emphasis on Isoniazid and Linezolid reflects a strategic decision to address the high burden of these diseases in Indonesia. The market demand for these products is driven by the need for effective treatments to combat infectious diseases, which remain a significant public health challenge in the country. By prioritizing these therapeutic areas, Sekretariat Ditjen P2P aims to improve health outcomes and support the national health agenda.
2Sourcing Profile
Sekretariat Ditjen P2P's sourcing strategy is centered on importing generic drugs from India, focusing on specific formulations such as Isoniazid and Linezolid. India's well-established pharmaceutical industry, known for its compliance with international GMP standards, aligns with Indonesia's regulatory requirements. This sourcing approach ensures the availability of cost-effective and quality-assured medicines to meet the therapeutic needs of the Indonesian population.
3Market Positioning
Based on its product mix, Sekretariat Ditjen P2P serves the public health sector in Indonesia, focusing on government tenders and wholesale distribution to healthcare facilities. By importing essential medicines, the organization plays a crucial role in ensuring that public health programs have access to necessary treatments, thereby supporting the government's efforts to provide equitable healthcare services across the country.
Seller's Guide — How to Become a Supplier to Sekretariat Ditjen Pencegahan DAN
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Sekretariat Ditjen P2P by offering alternative products or formulations that align with Indonesia's public health priorities. Expanding the range of imported formulations could help address emerging health challenges and diversify the supply chain. Additionally, offering products that meet Indonesia's regulatory standards and quality requirements would be essential for establishing a successful partnership.
2Requirements & Qualifications
Indian exporters seeking to supply Sekretariat Ditjen P2P and the Indonesian market must ensure that their products are registered with BPOM and comply with Indonesian regulations. This includes obtaining necessary import licenses, adhering to GMP standards, and ensuring that products meet quality, safety, and efficacy criteria. Additionally, products must have appropriate labeling in the Indonesian language and provide objective, non-misleading information to ensure appropriate and safe use.
3How to Approach
To establish a relationship with Sekretariat Ditjen P2P, Indian exporters should begin by ensuring that their products are registered with BPOM and comply with Indonesian regulations. Participating in government tenders and engaging in direct communication with Sekretariat Ditjen P2P can facilitate the procurement process.
Frequently Asked Questions — Sekretariat Ditjen Pencegahan DAN
What products does Sekretariat Ditjen Pencegahan DAN import from India?
Sekretariat Ditjen Pencegahan DAN imports 2 pharmaceutical products across 2 categories. Top imports: Isoniazid ($1.1M), Linezolid ($200.0K).
Who supplies pharmaceuticals to Sekretariat Ditjen Pencegahan DAN from India?
Sekretariat Ditjen Pencegahan DAN sources from 7 verified Indian suppliers. The primary supplier is Macleods Pharmaceuticals Limited (44.2% of imports, $1.6M).
What is Sekretariat Ditjen Pencegahan DAN's total pharmaceutical import value?
Sekretariat Ditjen Pencegahan DAN's total pharmaceutical import value from India is $1.3M, based on 25 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Sekretariat Ditjen Pencegahan DAN focus on?
Sekretariat Ditjen Pencegahan DAN imports across 2 categories. The largest: Tuberculosis Medications (84.0%), Advanced Antibiotics (16.0%).
Get Full Sekretariat Ditjen Pencegahan DAN Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Sekretariat Ditjen Pencegahan DAN identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Sekretariat Ditjen Pencegahan DAN's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 25 individual customs records matching Sekretariat Ditjen Pencegahan DAN.
- 5.Supplier Verification: Sekretariat Ditjen Pencegahan DAN sources from 7 verified Indian suppliers across 58 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.