Regency Pharma Limited
Pharmaceutical Importer · Mauritius · Antimalarial & Antiparasitic Focus · $8.9M Total Trade · DGFT Verified
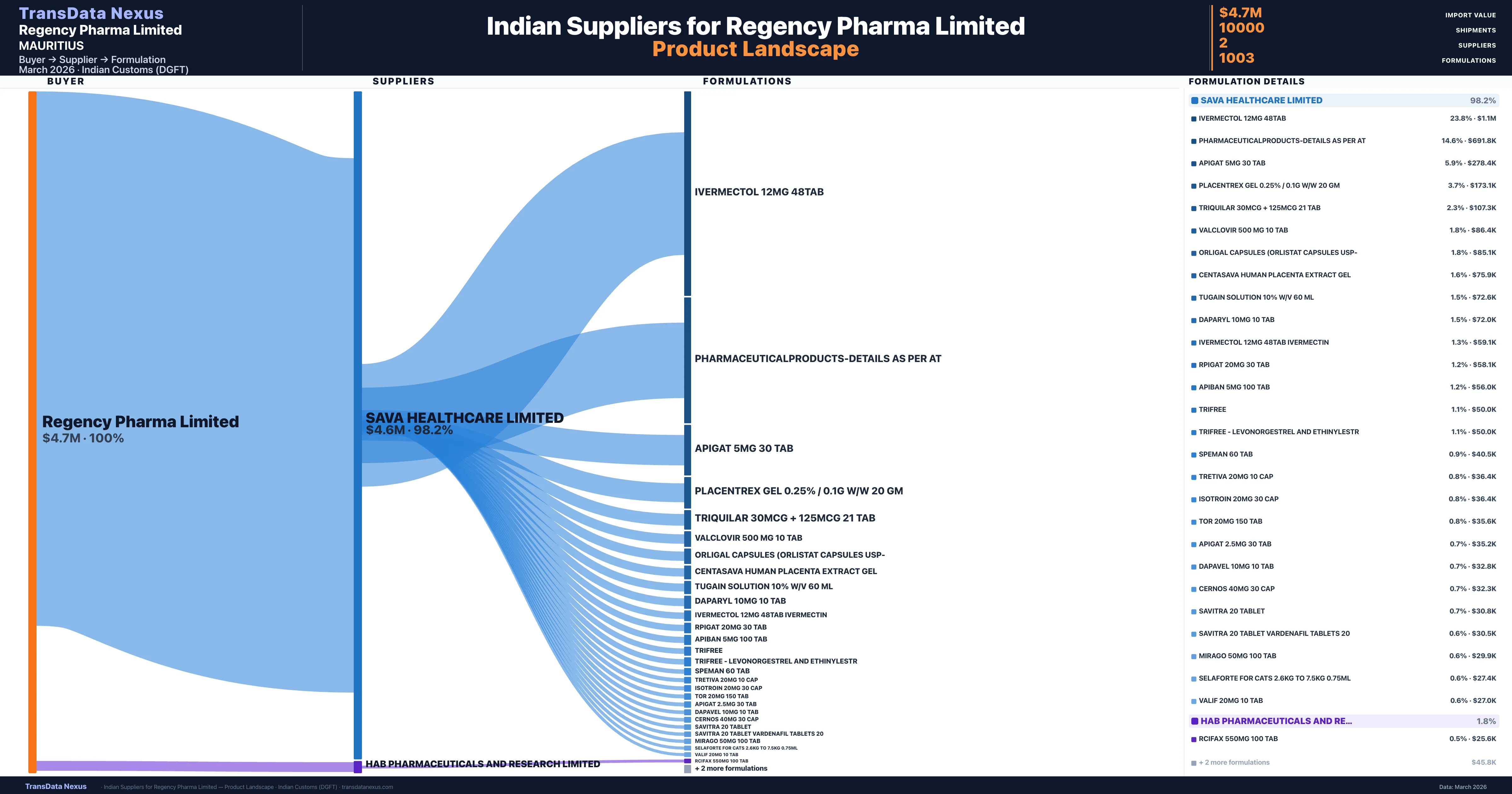
Regency Pharma Limited is a pharmaceutical importer based in Mauritius with a total trade value of $8.9M across 16 products in 9 therapeutic categories. Based on 3,690 verified import shipments from Indian Customs (DGFT) records, Regency Pharma Limited is the #1 buyer in 4 products including Ivermectin, Empagliflozin, Extract. Regency Pharma Limited sources from 2 verified Indian suppliers, with Sava Healthcare Limited accounting for 98.2% of imports.
Regency Pharma Limited — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Regency Pharma Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Sava Healthcare Limited | $4.6M | 9,998 | 98.2% |
| Hab Pharmaceuticals And Research Limited | $85.1K | 2 | 1.8% |
Regency Pharma Limited sources from 2 verified Indian suppliers across 1,003 distinct formulations. The sourcing is highly concentrated — Sava Healthcare Limited accounts for 98.2% of total imports, indicating a strategic single-source relationship.
What Formulations Does Regency Pharma Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Ivermectol 12MG 48tab | $1.1M | 82 |
| Pharmaceuticalproducts-details as per | $691.8K | 28 |
| Apigat 5MG 30 TAB | $278.4K | 66 |
| Placentrex gel 0.25% / 0.1g w/w 20 GM | $173.1K | 55 |
| Triquilar 30mcg + 125mcg 21 TAB | $107.3K | 35 |
| Valclovir 500 MG 10 TAB | $86.4K | 35 |
| Orligal capsules (orlistat capsules | $85.1K | 2 |
| Centasava human placenta extract | $75.9K | 2 |
| Tugain solution 10% w/v 60 ML | $72.6K | 68 |
| Daparyl 10MG 10 TAB | $72.0K | 35 |
| Ivermectol 12MG 48tab | $59.1K | 3 |
| Rpigat 20MG 30 TAB | $58.1K | 42 |
| Apiban 5MG 100 TAB | $56.0K | 5 |
| Trifree | $50.0K | 1 |
| Trifree - levonorgestrel and | $50.0K | 1 |
Regency Pharma Limited imports 1,003 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Regency Pharma Limited Import?
Regency Pharma Limited Therapeutic Categories — 9 Specializations
Regency Pharma Limited imports across 9 therapeutic categories, with Antimalarial & Antiparasitic (29.9%), Advanced Diabetes Medications (21.7%), Diabetes & Endocrine (15.1%) representing the largest segments. The portfolio is concentrated — top 5 products = 82% of total imports.
Antimalarial & Antiparasitic
2 products · 29.9% · $2.7M
Advanced Diabetes Medications
6 products · 21.7% · $1.9M
Diabetes & Endocrine
1 products · 15.1% · $1.3M
Cardiovascular
1 products · 14.1% · $1.3M
Ayurvedic & Herbal Products
2 products · 10.7% · $959.0K
Diuretics
1 products · 3.4% · $308.1K
Immunosuppressants
1 products · 3.1% · $276.0K
Respiratory
1 products · 2.0% · $180.7K
Medical Devices & Diagnostics
1 products · 0.0% · $1.0K
Import Portfolio — Top 16 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ivermectin | Antimalarial & Antiparasitic | $2.5M | 363 | 28.4% | 1 |
| 2 | Empagliflozin | Diabetes & Endocrine | $1.3M | 688 | 15.0% | 1 |
| 3 | Dapagliflozin | Advanced Diabetes Medications | $1.3M | 692 | 2.7% | 7 |
| 4 | Apixaban | Cardiovascular | $1.3M | 306 | 0.7% | 19 |
| 5 | Extract | Ayurvedic & Herbal Products | $806.6K | 220 | 6.6% | 1 |
| 6 | Semaglutide | Advanced Diabetes Medications | $435.7K | 196 | 7.5% | 4 |
| 7 | Torsemide | Diuretics | $308.1K | 417 | 1.7% | 7 |
| 8 | Cyclosporine | Immunosuppressants | $276.0K | 320 | 2.3% | 7 |
| 9 | Tiotropium | Respiratory | $180.7K | 74 | 0.3% | 11 |
| 10 | Asava | Ayurvedic & Herbal Products | $152.4K | 6 | 33.3% | 1 |
| 11 | Pyrantel | Antimalarial & Antiparasitic | $148.3K | 36 | 4.5% | 6 |
| 12 | Linagliptin | Advanced Diabetes Medications | $71.9K | 70 | 1.8% | 4 |
| 13 | Acarbose | Advanced Diabetes Medications | $37.2K | 89 | 0.6% | 15 |
| 14 | Canagliflozin | Advanced Diabetes Medications | $24.1K | 111 | 2.7% | 9 |
| 15 | Saxagliptin | Advanced Diabetes Medications | $20.5K | 93 | 0.4% | 6 |
| 16 | Catheter | Medical Devices & Diagnostics | $1.0K | 9 | 0.9% | 7 |
Regency Pharma Limited imports 16 pharmaceutical products across 9 categories into Mauritius totaling $8.9M. The company is the #1 buyer for 4 products: Ivermectin, Empagliflozin, Extract, Asava.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Regency Pharma Limited.
Request DemoRegency Pharma Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Regency Pharma Limited is a pharmaceutical importer and distributor based in Mauritius, specializing in the procurement and distribution of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The company operates as a primary wholesaler, sourcing a diverse range of pharmaceutical products to meet the needs of the Mauritian healthcare sector. Headquartered at Warehouse No.1, BPML Freeport Zone 9, Plaine Magnien SSR International, Mauritius, Regency Pharma Limited plays a pivotal role in ensuring the availability of essential medications across the island nation.
Established in 1991, Regency Pharma Limited has developed a robust network of suppliers and partners, primarily focusing on imports from India. The company's extensive import portfolio encompasses 16 distinct products across nine therapeutic categories, reflecting its commitment to addressing a wide array of medical needs within Mauritius. This strategic sourcing approach underscores Regency Pharma Limited's dedication to providing comprehensive pharmaceutical solutions to the Mauritian market.
2Distribution Network
Regency Pharma Limited's distribution network is strategically located to facilitate efficient logistics and timely delivery of pharmaceutical products throughout Mauritius. The company's primary warehouse, situated at Warehouse No.1, BPML Freeport Zone 9, Plaine Magnien SSR International, serves as the central hub for inventory management and distribution operations. This location offers proximity to key transportation routes, enhancing the company's ability to manage and distribute products effectively across the island.
While specific details regarding additional warehouse locations and logistics capabilities are not publicly disclosed, Regency Pharma Limited's centralized distribution model suggests a focus on optimizing supply chain operations to meet the demands of the Mauritian healthcare sector. The company's strategic positioning within the Freeport Zone indicates potential advantages in terms of customs procedures and import-export facilitation, contributing to the overall efficiency of its distribution network.
3Industry Role
In the pharmaceutical supply chain of Mauritius, Regency Pharma Limited functions as a primary wholesaler, sourcing a diverse array of finished pharmaceutical formulations from international suppliers, predominantly from India. The company's extensive import activities, encompassing 16 products across nine therapeutic categories, highlight its significant role in ensuring the availability of essential medications within the Mauritian market. By acting as a conduit between international manufacturers and local healthcare providers, Regency Pharma Limited plays a crucial role in maintaining the supply of critical pharmaceutical products to meet the needs of the Mauritian population.
The company's position as a primary wholesaler is further emphasized by its substantial import volume, with a total import value of $8.9 million USD and 3,690 shipments recorded from 2022 to 2026. This scale of operations underscores Regency Pharma Limited's capacity to manage and distribute a wide range of pharmaceutical products, reinforcing its integral position within the Mauritian pharmaceutical supply chain.
Supplier Relationship Intelligence — Regency Pharma Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Regency Pharma Limited exhibits a high degree of sourcing concentration, with a significant majority of its pharmaceutical imports originating from India. The company's total import value from India stands at $8.9 million USD, encompassing 3,690 shipments across 16 products. This sourcing strategy underscores India's pivotal role as a supplier of pharmaceutical products to Mauritius, aligning with the broader trend of Indian pharmaceuticals dominating the Mauritian market.
The concentration of imports from a single country, particularly one as dominant as India in the Mauritian pharmaceutical market, presents both strategic advantages and potential risks. On the positive side, this focused sourcing approach may lead to streamlined logistics, favorable pricing, and established supplier relationships. However, it also exposes Regency Pharma Limited to risks associated with geopolitical factors, regulatory changes, and supply chain disruptions within India. The company's reliance on a limited number of suppliers—Sava Healthcare Limited and HAB Pharmaceuticals and Research Limited—further intensifies this concentration risk. While the data indicates a stable relationship with these suppliers, any disruptions could significantly impact Regency Pharma Limited's ability to meet the pharmaceutical needs of the Mauritian market.
2Supply Chain Resilience
Regency Pharma Limited's supply chain resilience is closely tied to its sourcing strategy, which is heavily concentrated on imports from India. The company's primary suppliers, Sava Healthcare Limited and HAB Pharmaceuticals and Research Limited, account for the majority of its pharmaceutical imports. This concentrated supplier base may enhance operational efficiency but also introduces vulnerabilities related to supplier dependency.
The company's import data reveals a diverse range of 1,003 unique formulations, indicating a broad product portfolio. However, the heavy reliance on a limited number of suppliers for these formulations suggests potential challenges in supply chain diversification. To bolster resilience, Regency Pharma Limited may consider exploring additional suppliers and diversifying its sourcing strategies. This approach could mitigate risks associated with supply chain disruptions and enhance the company's ability to maintain a consistent supply of pharmaceutical products to the Mauritian market.
3Strategic Implications
Regency Pharma Limited's concentrated sourcing pattern, characterized by a heavy reliance on imports from India and a limited number of suppliers, has several strategic implications. For the company, this approach may lead to cost efficiencies and streamlined operations due to established supplier relationships and familiarity with the Indian pharmaceutical market. However, the concentration also exposes Regency Pharma Limited to risks associated with supply chain disruptions, regulatory changes, and geopolitical factors affecting India.
For Indian pharmaceutical exporters, Regency Pharma Limited's sourcing pattern presents both opportunities and challenges. The company's established relationships with Indian suppliers indicate a preference for sourcing from India, potentially facilitating entry for new Indian exporters. However, the existing supplier base's dominance may pose challenges for new entrants seeking to establish a foothold in Regency Pharma Limited's supply chain. Understanding the company's sourcing dynamics and identifying areas where additional suppliers can add value will be crucial for Indian exporters aiming to collaborate with Regency Pharma Limited.
Importing Pharmaceuticals into Mauritius — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Mauritius
1Regulatory Authority & Framework
In Mauritius, the regulation of pharmaceutical products is primarily governed by the Pharmacy Act of 1983 and the Consumer Protection (Price and Supplies Control) Act of 1998. The Pharmacy Act 1983 establishes the legal framework for the authorization, marketing, sale, and supply of pharmaceutical products, defining "pharmaceutical product" as any drug, medicine, preparation, poison, or therapeutic substance intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease in humans or animals.
The Consumer Protection (Price and Supplies Control) Act of 1998 complements the Pharmacy Act by regulating the pricing and supply of pharmaceutical products, ensuring that consumers have access to essential medications at fair prices. These legislative frameworks collectively aim to maintain the quality, safety, and efficacy of pharmaceutical products available in Mauritius, safeguarding public health and ensuring the integrity of the pharmaceutical supply chain.
2Import Licensing & GMP
Pharmaceutical products imported into Mauritius must comply with the regulations set forth by the Pharmacy Board, operating under the Ministry of Health and Wellness. Only wholesalers licensed by the Pharmacy Board are authorized to import pharmaceutical products, ensuring that all imported medications meet the required standards for quality and safety.
Import licensing requirements include the submission of detailed documentation, such as certificates of analysis, Good Manufacturing Practice (GMP) certificates, and other relevant regulatory approvals. GMP certificates from recognized authorities, such as the World Health Organization (WHO) or the European Union (EU), are typically required to demonstrate that the manufacturing processes adhere to internationally accepted standards. Additionally, wholesalers must obtain wholesale distribution authorization from the Pharmacy Board, which involves meeting specific criteria related to storage facilities, personnel qualifications, and operational procedures.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure they meet the quality standards set by the Pharmacy Board. This testing verifies the identity, purity, strength, and quality of the medications, ensuring they are safe and effective for consumer use. Stability requirements are also enforced to ensure that products maintain their efficacy and safety throughout their shelf life.
Labeling requirements stipulate that all pharmaceutical products must display information in English and French, the official languages of Mauritius. Labels must include details such as the product name, active ingredients, dosage form, strength, batch number, manufacturing and expiry dates, storage conditions, and the name and address of the manufacturer or distributor. Serialization mandates may also be implemented to enhance traceability and prevent counterfeit products from entering the market, although specific requirements can vary and should be verified with the Pharmacy Board.
4Recent Regulatory Changes
As of March 2026, there have been no publicly disclosed significant policy changes affecting Indian pharmaceutical imports into Mauritius between 2024 and 2026. The regulatory environment remains stable, with existing frameworks under the Pharmacy Act 1983 and the Consumer Protection (Price and Supplies Control) Act of 1998 continuing to govern the importation and distribution of pharmaceutical products. However, stakeholders are advised to monitor communications from the Ministry of Health and Wellness and the Pharmacy Board for any future updates or amendments to existing regulations.
Regency Pharma Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Regency Pharma Limited's product strategy focuses on importing a diverse range of pharmaceutical formulations across multiple therapeutic categories, including antimalarial and antiparasitic agents, advanced diabetes medications, and other diabetes and endocrine treatments. This strategic focus aligns with the prevalent health concerns in Mauritius, where diseases such as malaria
Frequently Asked Questions — Regency Pharma Limited
What products does Regency Pharma Limited import from India?
Regency Pharma Limited imports 16 pharmaceutical products across 9 categories. Top imports: Ivermectin ($2.5M), Empagliflozin ($1.3M), Dapagliflozin ($1.3M), Apixaban ($1.3M), Extract ($806.6K).
Who supplies pharmaceuticals to Regency Pharma Limited from India?
Regency Pharma Limited sources from 2 verified Indian suppliers. The primary supplier is Sava Healthcare Limited (98.2% of imports, $4.6M).
What is Regency Pharma Limited's total pharmaceutical import value?
Regency Pharma Limited's total pharmaceutical import value from India is $8.9M, based on 3,690 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Regency Pharma Limited focus on?
Regency Pharma Limited imports across 9 categories. The largest: Antimalarial & Antiparasitic (29.9%), Advanced Diabetes Medications (21.7%), Diabetes & Endocrine (15.1%).
Get Full Regency Pharma Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Regency Pharma Limited identified across shipments using consignee name normalization, aggregating 3 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Regency Pharma Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 3,690 individual customs records matching Regency Pharma Limited.
- 5.Supplier Verification: Regency Pharma Limited sources from 2 verified Indian suppliers across 1,003 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
16 Products Tracked
9 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 3 company name variants from customs records. For current shipment-level data, contact TransData Nexus.