Reed - Lane Inc
Pharmaceutical Importer · United States · Respiratory & OTC Focus · $1.1M Total Trade · DGFT Verified
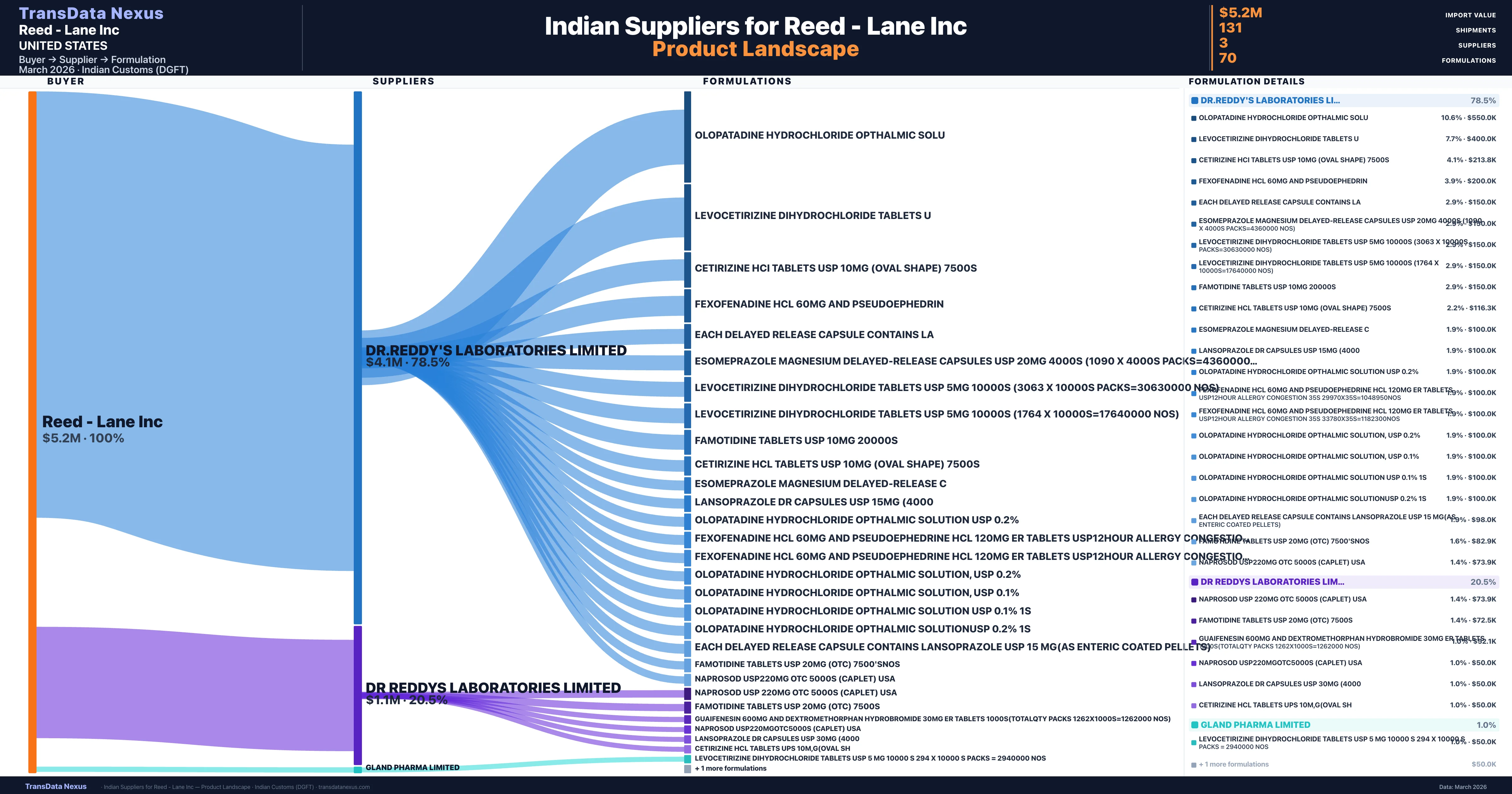
Reed - Lane Inc is a pharmaceutical importer based in United States with a total trade value of $1.1M across 4 products in 1 therapeutic categories. Based on 35 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Reed - Lane Inc sources from 3 verified Indian suppliers, with Dr.reddy's Laboratories Limited accounting for 78.5% of imports.
Reed - Lane Inc — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Reed - Lane Inc?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Dr.reddy's Laboratories Limited | $4.1M | 107 | 78.5% |
| Dr Reddys Laboratories Limited | $1.1M | 23 | 20.5% |
| Gland Pharma Limited | $50.0K | 1 | 1.0% |
Reed - Lane Inc sources from 3 verified Indian suppliers across 70 distinct formulations. Total import value: $5.2M across 131 shipments.
What Formulations Does Reed - Lane Inc Import?
| Formulation | Value | Ships |
|---|---|---|
| Olopatadine hydrochloride opthalmic | $550.0K | 11 |
| Levocetirizine dihydrochloride tablets | $400.0K | 8 |
| Cetirizine hci tablets USP 10MG (oval shape) 7500s | $213.8K | 6 |
| Fexofenadine hcl 60MG and | $200.0K | 4 |
| Each delayed release capsule contains | $150.0K | 3 |
| Esomeprazole magnesium delayed-release capsules USP 20MG 4000s (1090 x 4000s packs=4360000 nos) | $150.0K | 3 |
| Levocetirizine dihydrochloride tablets USP 5MG 10000s (3063 x 10000s packs=30630000 nos) | $150.0K | 3 |
| Levocetirizine dihydrochloride tablets USP 5MG 10000s (1764 x 10000s=17640000 nos) | $150.0K | 3 |
| Famotidine tablets USP 10MG 20000s | $150.0K | 3 |
| Cetirizine hcl tablets USP 10MG (oval shape) 7500s | $116.3K | 3 |
| Esomeprazole magnesium delayed-release | $100.0K | 2 |
| Lansoprazole dr capsules USP 15MG | $100.0K | 2 |
| Olopatadine hydrochloride opthalmic solution USP | $100.0K | 2 |
| Fexofenadine hcl 60MG and pseudoephedrine hcl 120MG er tablets usp12hour allergy congestion 35s | $100.0K | 2 |
| Fexofenadine hcl 60MG and pseudoephedrine hcl 120MG er tablets usp12hour allergy congestion 35s | $100.0K | 2 |
Reed - Lane Inc imports 70 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Reed - Lane Inc Import?
Top Products by Import Value
Reed - Lane Inc Therapeutic Categories — 1 Specializations
Reed - Lane Inc imports across 1 therapeutic categories, with Respiratory & OTC (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Respiratory & OTC
4 products · 100.0% · $1.1M
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Guaifenesin | Respiratory & OTC | $521.5K | 22 | 1.0% | 18 |
| 2 | Dextromethorphan | Respiratory & OTC | $228.2K | 5 | 1.5% | 13 |
| 3 | Ephedrine | Respiratory & OTC | $200.0K | 4 | 0.9% | 11 |
| 4 | Pseudoephedrine | Respiratory & OTC | $200.0K | 4 | 1.7% | 7 |
Reed - Lane Inc imports 4 pharmaceutical products across 1 categories into United States totaling $1.1M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Reed - Lane Inc.
Request DemoReed - Lane Inc — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Reed - Lane Inc is a privately held pharmaceutical contract packaging company headquartered in Wayne, New Jersey. Established in 1959, Reed - Lane specializes in providing innovative packaging solutions for prescription (Rx), over-the-counter (OTC), and generic pharmaceutical products. Their services encompass blister packaging, bottling, pouching, and convenience vial filling, catering to a diverse clientele in the pharmaceutical industry.
The company operates from a 132,000-square-foot facility in Wayne, NJ, featuring over 25 dedicated production suites. These facilities are equipped with individual air handling and HEPA filtration systems, temperature-controlled storage for bulk and finished goods, and stringent environmental and security monitoring, including over 150 high-definition CCTV cameras. Reed - Lane is licensed by the U.S. Drug Enforcement Administration (DEA) to package controlled substances classified as Schedule III–V drugs.
2Distribution Network
Reed - Lane's distribution network is primarily centered within the United States, with its main operations based in Wayne, New Jersey. The company's 132,000-square-foot facility serves as the hub for its packaging and distribution activities. While specific details about additional warehouse locations or logistics capabilities beyond this facility are not publicly disclosed, Reed - Lane's extensive warehousing space and fully validated Enterprise Resource Planning (ERP) system suggest a robust infrastructure capable of managing significant distribution needs.
3Industry Role
Reed - Lane Inc functions as a contract packaging service provider within the pharmaceutical supply chain. Unlike wholesalers or distributors, Reed - Lane specializes in the packaging of pharmaceutical products, including both prescription and over-the-counter medications. Their role is to prepare and package these products for distribution, ensuring compliance with industry standards and regulations. This positions Reed - Lane as a critical intermediary between pharmaceutical manufacturers and the end-users of these products.
Supplier Relationship Intelligence — Reed - Lane Inc
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Reed - Lane Inc's sourcing strategy exhibits a high degree of concentration, with a significant portion of their imported pharmaceutical products originating from India. Between 2022 and 2026, Reed - Lane imported finished pharmaceutical formulations valued at $1.1 million from India, encompassing 35 shipments and 70 unique formulations. The top four products imported include Guaifenesin ($522K), Dextromethorphan ($228K), Ephedrine ($200K), and Pseudoephedrine ($200K), all within the respiratory and OTC therapeutic category. This concentration indicates a strategic focus on specific products and suppliers.
The primary supplier, DR.REDDY'S LABORATORIES LIMITED, accounted for 78.5% of the total import value, followed by DR REDDYS LABORATORIES LIMITED with 20.5%, and GLAND PHARMA LIMITED with 1.0%. This heavy reliance on a limited number of suppliers may pose risks related to supply chain disruptions, pricing volatility, and dependency on supplier performance. However, it also suggests a strategic choice to maintain strong, focused relationships with key suppliers to ensure product quality and consistency.
2Supply Chain Resilience
Reed - Lane Inc's supply chain resilience is closely tied to its sourcing concentration from Indian suppliers. The company's reliance on a few key suppliers, particularly DR.REDDY'S LABORATORIES LIMITED, indicates a deep integration with these partners. While this can lead to streamlined operations and potential cost benefits, it also exposes Reed - Lane to risks associated with supply chain disruptions, such as production delays, regulatory changes, or geopolitical issues affecting India. The limited number of suppliers may also impact the company's ability to diversify its supply chain in response to market changes or disruptions.
3Strategic Implications
Reed - Lane Inc's concentrated sourcing strategy positions the company to leverage strong relationships with key Indian suppliers, potentially securing favorable terms and ensuring consistent product quality. However, this approach also exposes Reed - Lane to risks associated with supply chain disruptions, pricing volatility, and dependency on supplier performance. For Indian exporters, understanding Reed - Lane's sourcing patterns presents an opportunity to strengthen existing partnerships and explore avenues for diversification within Reed - Lane's product portfolio. By offering complementary products or services, Indian exporters can enhance their value proposition and potentially increase their share in Reed - Lane's supply chain.
Importing Pharmaceuticals into United States — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United States
1Regulatory Authority & Framework
In the United States, the Food and Drug Administration (FDA) serves as the primary regulatory authority overseeing the importation of pharmaceutical products. The FDA ensures that all drugs entering the U.S. market meet standards for quality, safety, and effectiveness. The Federal Food, Drug, and Cosmetic Act (FD&C Act) provides the legal framework governing the importation of drugs, including requirements for drug registration, labeling, and manufacturing practices. The FDA's Regulatory Procedures Manual and Investigations Operations Manual outline the procedures for the importation and examination of drugs, including the authority to refuse admission to products that do not comply with FDA regulations.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products entering the United States are governed by the FDA. Importers must ensure that their products comply with FDA regulations, including those related to Good Manufacturing Practices (GMP). The FDA recognizes GMP certifications from various international standards, such as EU GMP, WHO GMP, and PIC/S, provided they meet FDA criteria. Importers must verify that their foreign suppliers hold valid GMP certifications and that their manufacturing processes align with FDA standards. Additionally, importers must obtain wholesale distribution authorization and ensure that their products are properly labeled and registered with the FDA.
3Quality & Labeling
Imported pharmaceutical products must adhere to FDA requirements for quality and labeling. This includes compliance with batch testing and stability requirements to ensure product safety and efficacy. Labeling must meet FDA standards, including accurate and truthful information, appropriate language requirements, and adherence to serialization mandates for tracking and tracing products through the supply chain. The FDA reviews drug labeling at the time of import to verify compliance with regulations, and may refuse entry to products that are misbranded or do not meet labeling requirements.
4Recent Regulatory Changes
Between 2024 and 2026, the FDA has implemented several policy changes affecting the importation of pharmaceutical products from India. These include updates to the Section 804 Importation Program, which allows for the importation of certain prescription drugs from Canada, and revisions to the FDA's Import Procedures Manual to enhance the efficiency and effectiveness of import operations. Additionally, the FDA has increased inspections and enforcement actions to ensure compliance with the Drug Supply Chain Security Act (DSCSA), impacting the importation of pharmaceutical products. These changes aim to strengthen the integrity of the pharmaceutical supply chain and ensure the safety and efficacy of imported drugs.
Reed - Lane Inc — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Reed - Lane Inc's focus on importing finished pharmaceutical formulations from India, particularly in the respiratory and OTC therapeutic categories, aligns with market demand for these products. The top four imported products—Guaifenesin, Dextromethorphan, Ephedrine, and Pseudoephedrine—are commonly used in over-the-counter cough and cold medications, indicating a strategic emphasis on high-demand consumer health products. This product strategy allows Reed - Lane to cater to a significant segment of the U.S. pharmaceutical market, addressing consumer needs for effective and accessible respiratory treatments.
2Sourcing Profile
Reed - Lane Inc's sourcing strategy is centered on importing finished pharmaceutical formulations from India, focusing on specific products within the respiratory and OTC therapeutic categories. This approach leverages India's established pharmaceutical manufacturing capabilities and cost advantages. By concentrating on a select range of products, Reed - Lane can maintain quality control and streamline its supply chain operations. India's role as a major supplier of generic pharmaceuticals to the U.S. market positions it as a key partner in Reed - Lane's procurement strategy.
3Market Positioning
Based on its product mix, Reed - Lane Inc serves the U.S. over-the-counter pharmaceutical market, focusing on respiratory treatments. By importing and packaging popular OTC medications, Reed - Lane positions itself as a contract packager that facilitates the availability of these products to consumers through various distribution channels, including retail pharmacies and online platforms. This market positioning enables Reed - Lane to meet consumer demand for accessible and effective health solutions.
Seller's Guide — How to Become a Supplier to Reed - Lane Inc
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Reed - Lane Inc, particularly in the respiratory and OTC therapeutic categories. Reed - Lane's concentrated sourcing strategy suggests potential for diversification within its product portfolio. Indian exporters can explore opportunities to supply additional formulations or variations of existing products to meet evolving market demands. By understanding Reed - Lane's product focus and quality standards, Indian suppliers can tailor their offerings to align with Reed - Lane's requirements, potentially increasing their presence in the U.S
Frequently Asked Questions — Reed - Lane Inc
What products does Reed - Lane Inc import from India?
Reed - Lane Inc imports 4 pharmaceutical products across 1 categories. Top imports: Guaifenesin ($521.5K), Dextromethorphan ($228.2K), Ephedrine ($200.0K), Pseudoephedrine ($200.0K).
Who supplies pharmaceuticals to Reed - Lane Inc from India?
Reed - Lane Inc sources from 3 verified Indian suppliers. The primary supplier is Dr.reddy's Laboratories Limited (78.5% of imports, $4.1M).
What is Reed - Lane Inc's total pharmaceutical import value?
Reed - Lane Inc's total pharmaceutical import value from India is $1.1M, based on 35 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Reed - Lane Inc focus on?
Reed - Lane Inc imports across 1 categories. The largest: Respiratory & OTC (100.0%).
Get Full Reed - Lane Inc Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Reed - Lane Inc identified across shipments using consignee name normalization, aggregating 3 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Reed - Lane Inc's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 35 individual customs records matching Reed - Lane Inc.
- 5.Supplier Verification: Reed - Lane Inc sources from 3 verified Indian suppliers across 70 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 3 company name variants from customs records. For current shipment-level data, contact TransData Nexus.