Prince Pharma
Pharmaceutical Importer · Congo Dr · Advanced Antibiotics Focus · $163.7K Total Trade · DGFT Verified
Prince Pharma is a pharmaceutical importer based in Congo Dr with a total trade value of $163.7K across 2 products in 2 therapeutic categories. Based on 39 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Prince Pharma sources from 61 verified Indian suppliers, with Prashi Pharma Private Limited accounting for 40.0% of imports.
Prince Pharma — Import Portfolio & Supplier Network
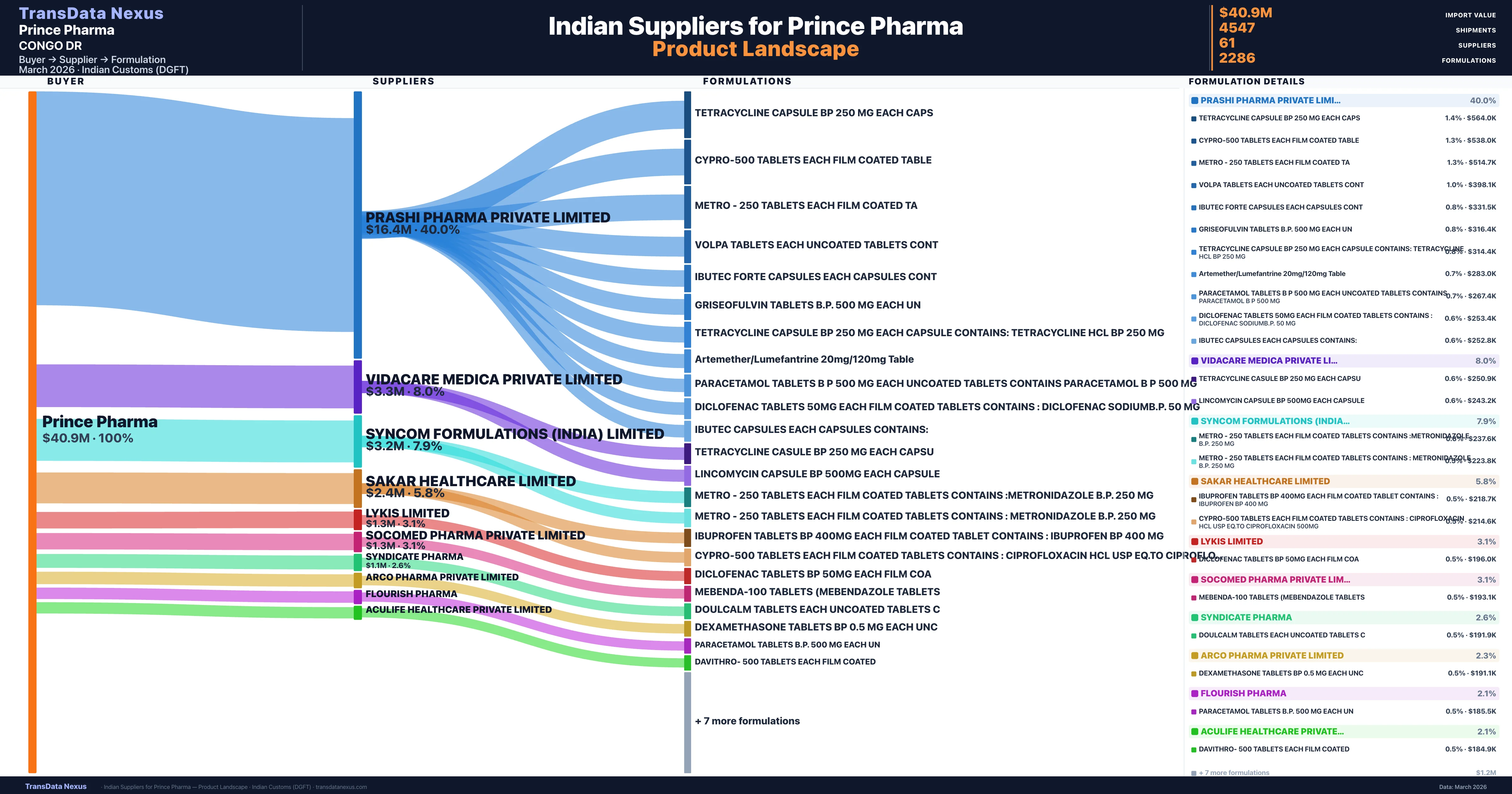
Who Are the Verified Indian Suppliers to Prince Pharma?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Prashi Pharma Private Limited | $16.4M | 1,417 | 40.0% |
| Vidacare Medica Private Limited | $3.3M | 281 | 8.0% |
| Syncom Formulations (india) Limited | $3.2M | 825 | 7.9% |
| Sakar Healthcare Limited | $2.4M | 208 | 5.8% |
| Lykis Limited | $1.3M | 216 | 3.1% |
| Socomed Pharma Private Limited | $1.3M | 193 | 3.1% |
| Syndicate Pharma | $1.1M | 109 | 2.6% |
| Arco Pharma Private Limited | $952.9K | 89 | 2.3% |
| Flourish Pharma | $875.3K | 53 | 2.1% |
| Aculife Healthcare Private Limited | $861.5K | 53 | 2.1% |
| Innova Captab Limited | $704.9K | 90 | 1.7% |
| Syncom Formulations India Limited | $661.2K | 156 | 1.6% |
| Halewood Laboratories Private Limited | $616.0K | 100 | 1.5% |
| Ipca Laboratories Limited | $575.5K | 38 | 1.4% |
| Laborate Pharmaceutical India Limited | $541.8K | 18 | 1.3% |
| Ambadnya Life Science Llp | $483.2K | 22 | 1.2% |
| East African India Overseas | $472.6K | 16 | 1.2% |
| Synokem Pharmaceuticals Limited | $457.6K | 119 | 1.1% |
| Kamla Lifesciences Limited | $425.7K | 54 | 1.0% |
| Neozen Healthcare Llp | $378.3K | 41 | 0.9% |
| Medico Remedies Limited | $327.3K | 15 | 0.8% |
| Maxwell Life Science Private Limited | $305.2K | 18 | 0.7% |
| Laborate Pharmaceuticals India Limited | $278.7K | 17 | 0.7% |
| Lyka Labs Limited | $252.4K | 15 | 0.6% |
| Makcur Laboratories Limited | $230.2K | 13 | 0.6% |
| Nem Laboratories Private Limited | $212.4K | 20 | 0.5% |
| Sparsh Bio-tech Private Limited | $192.3K | 8 | 0.5% |
| Flagship Biotech International Private Limited | $185.2K | 18 | 0.5% |
| Galen Pharmaceuticals Limited | $179.5K | 32 | 0.4% |
| Macleods Pharmaceuticals Limited | $173.6K | 4 | 0.4% |
Prince Pharma sources from 61 verified Indian suppliers across 2,286 distinct formulations. The supply base is diversified across 61 suppliers, reducing single-source dependency risk.
What Formulations Does Prince Pharma Import?
| Formulation | Value | Ships |
|---|---|---|
| Tetracycline capsule BP 250 MG Each CAPS | $564.0K | 19 |
| Cypro-500 tablets Each film coated | $538.0K | 22 |
| Metro - 250 tablets Each film coated | $514.7K | 25 |
| Volpa tablets Each uncoated tablets | $398.1K | 26 |
| Ibutec forte capsules Each capsules | $331.5K | 12 |
| Griseofulvin tablets b.p. 500 MG Each | $316.4K | 10 |
| Tetracycline capsule BP 250 MG Each capsule contains: tetracycline hcl BP 250 MG | $314.4K | 18 |
| Artemether/lumefantrine 20MG/120MG | $283.0K | 12 |
| Paracetamol tablets b p 500 MG Each uncoated tablets contains paracetamol b p 500 MG | $267.4K | 7 |
| Diclofenac tablets 50MG Each film coated tablets contains : diclofenac sodiumb.p. 50 MG | $253.4K | 12 |
| Ibutec capsules Each capsules | $252.8K | 10 |
| Tetracycline casule BP 250 MG Each | $250.9K | 9 |
| Lincomycin capsule BP 500MG Each | $243.2K | 9 |
| Metro - 250 tablets Each film coated tablets contains :metronidazole b.p. 250 MG | $237.6K | 14 |
| Metro - 250 tablets Each film coated tablets contains : metronidazole b.p. 250 MG | $223.8K | 15 |
Prince Pharma imports 2,286 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Prince Pharma Import?
Top Products by Import Value
Prince Pharma Therapeutic Categories — 2 Specializations
Prince Pharma imports across 2 therapeutic categories, with Advanced Antibiotics (54.3%), CNS & Psychiatric (45.7%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
1 products · 54.3% · $88.8K
CNS & Psychiatric
1 products · 45.7% · $74.9K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Norfloxacin | Advanced Antibiotics | $88.8K | 20 | 1.1% | 12 |
| 2 | Diazepam | CNS & Psychiatric | $74.9K | 19 | 1.0% | 15 |
Prince Pharma imports 2 pharmaceutical products across 2 categories into Congo Dr totaling $163.7K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Prince Pharma.
Request DemoPrince Pharma — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Prince Pharma is a leading pharmaceutical company based in Kinshasa, the capital of the Democratic Republic of Congo (DRC). Established over 15 years ago, the company specializes in importing and distributing a wide range of generic and branded medicines across various therapeutic areas. Their product portfolio includes both prescription and over-the-counter medications, catering to diverse healthcare needs within the DRC. (princepharma.co)
The company's medicines are manufactured in WHO-approved facilities in India and China, ensuring high efficacy and affordability. Prince Pharma's extensive distribution network ensures that their products are accessible throughout the country, including remote areas. Their commitment to continuous medical education for healthcare providers and addressing unmet healthcare needs underscores their role in enhancing the quality of healthcare in the DRC. (princepharma.co)
2Distribution Network
Prince Pharma operates a modern, ISO 9001:2015-certified warehouse equipped with Warehouse Management System (WMS) software and an automated system that streamlines picking and optimizes inventory. This facility forecasts demand and maintains controlled temperatures for perishable goods through real-time temperature monitoring. Their distribution network comprises numerous outlets and warehouses, ensuring effortless product availability throughout the DRC for optimal service. (princepharma.co)
3Industry Role
In the DRC's pharmaceutical supply chain, Prince Pharma functions primarily as a pharmaceutical importer and distributor. By sourcing medicines from international manufacturers and ensuring their availability across the country, they play a crucial role in meeting the healthcare needs of the Congolese population. Their extensive distribution network and commitment to quality make them a significant player in the DRC's pharmaceutical sector. (princepharma.co)
Supplier Relationship Intelligence — Prince Pharma
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Prince Pharma's sourcing strategy reveals a high concentration of imports from a select group of Indian suppliers. The top five suppliers account for 100% of their imports, with PRASHI PHARMA PRIVATE LIMITED leading at 40% of the total import value. This indicates a strategic choice to maintain strong, stable relationships with key suppliers, ensuring consistent product quality and supply. However, such concentration also poses risks; any disruption with these suppliers could significantly impact Prince Pharma's operations. The shipment data suggests a stable relationship, as evidenced by the consistent number of shipments over the years. Nonetheless, diversifying the supplier base could mitigate potential risks associated with over-reliance on a few suppliers.
2Supply Chain Resilience
Prince Pharma's supply chain resilience is bolstered by its partnerships with reputable Indian pharmaceutical manufacturers, all of which are WHO-approved. This ensures adherence to international quality standards. The company's modern warehousing facilities, equipped with automated systems and real-time monitoring, further enhance supply chain efficiency. However, the heavy reliance on a limited number of suppliers may expose the company to risks if any of these suppliers face operational challenges. To strengthen resilience, establishing relationships with additional suppliers and diversifying the product portfolio could be beneficial.
3Strategic Implications
Prince Pharma's sourcing pattern, characterized by a concentrated supplier base, positions the company to leverage strong relationships for favorable terms and consistent product quality. For Indian exporters, this presents an opportunity to become key suppliers by meeting Prince Pharma's quality standards and supply requirements. However, to mitigate potential risks associated with supplier concentration, Prince Pharma may consider diversifying its supplier base, which could open avenues for new Indian exporters to enter the market.
Importing Pharmaceuticals into Congo Dr — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Congo Dr
1Regulatory Authority & Framework
The Congolese Pharmaceutical Regulatory Authority (ACOREP) is responsible for regulating pharmaceutical imports and exports in the DRC. ACOREP oversees the authorization and control of drug imports, ensuring compliance with national and international standards. The regulatory framework includes obtaining an import license from an authorized commercial bank and undergoing a pre-shipment inspection by Bureau Veritas for shipments valued over $2,500.
2Import Licensing & GMP
Importers in the DRC must secure an import license from an authorized commercial bank before the physical shipment of goods. A pre-shipment inspection is mandatory for all imports valued at $2,500 FOB or greater. While Good Manufacturing Practice (GMP) certification is officially required, enforcement is often inconsistent; a Certificate of Pharmaceutical Product (CPP) is a critical document.
3Quality & Labeling
Imported pharmaceutical products must undergo quality control processes, including initial screening conducted locally using methods like the GPHF Minilab, with more comprehensive analysis performed at WHO-prequalified laboratories abroad. All documentation and labeling must be in French, as it is the official language of the DRC. Non-compliance with these requirements can lead to significant sanctions, including product recalls and fines.
4Recent Regulatory Changes
As of March 5, 2020, ACOREP has been in the process of assuming the responsibilities of the Directorate of Pharmacy and Medicine (DPM), including regulating clinical trials and authorizing and controlling drug imports and exports in the DRC. The transition from DPM to ACOREP is still in process as of this update.
Prince Pharma — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Prince Pharma's focus on importing advanced antibiotics and CNS & psychiatric medications aligns with the significant healthcare needs in the DRC. The high import values of Norfloxacin and Diazepam indicate substantial market demand for these therapeutic areas. This strategic focus enables Prince Pharma to address critical health challenges and position itself as a key supplier in these segments.
2Sourcing Profile
Prince Pharma's sourcing strategy emphasizes importing finished pharmaceutical formulations from WHO-approved manufacturers in India and China. This approach ensures adherence to international quality standards and meets the therapeutic needs of the DRC market. The company's reliance on a select group of suppliers reflects a strategic choice to maintain strong, stable relationships, although it also presents potential risks associated with supplier concentration.
3Market Positioning
Based on its product mix, Prince Pharma serves a broad segment of the DRC market, including retail pharmacies, hospitals, and government tenders. The company's extensive distribution network and focus on essential therapeutic areas position it as a key player in the Congolese pharmaceutical sector, catering to diverse healthcare needs across the country.
Seller's Guide — How to Become a Supplier to Prince Pharma
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to enter the DRC market by meeting Prince Pharma's quality standards and supply requirements. Diversifying the supplier base could mitigate potential risks associated with over-reliance on a few suppliers. Additionally, expanding the product portfolio to include other therapeutic areas could address unmet healthcare needs in the DRC, presenting growth opportunities for both Prince Pharma and potential suppliers.
2Requirements & Qualifications
Indian exporters seeking to supply Prince Pharma and the DRC market must ensure their products meet international quality standards and obtain necessary certifications, such as GMP and CPP. Compliance with DRC import regulations, including obtaining an import license and undergoing pre-shipment inspections, is mandatory. All documentation and labeling must be in French to meet regulatory requirements.
3How to Approach
To establish a relationship with Prince Pharma, Indian exporters should ensure their products meet the required quality standards and certifications. Engaging in direct communication to understand Prince Pharma's specific needs and participating in tenders can facilitate entry into the DRC market. Understanding and complying with the DRC's import regulations, including obtaining the necessary import licenses and ensuring all documentation is in French, is crucial. Building a strong, reliable partnership with Prince Pharma can lead to sustained success in the Congolese pharmaceutical market.
Frequently Asked Questions — Prince Pharma
What products does Prince Pharma import from India?
Prince Pharma imports 2 pharmaceutical products across 2 categories. Top imports: Norfloxacin ($88.8K), Diazepam ($74.9K).
Who supplies pharmaceuticals to Prince Pharma from India?
Prince Pharma sources from 61 verified Indian suppliers. The primary supplier is Prashi Pharma Private Limited (40.0% of imports, $16.4M).
What is Prince Pharma's total pharmaceutical import value?
Prince Pharma's total pharmaceutical import value from India is $163.7K, based on 39 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Prince Pharma focus on?
Prince Pharma imports across 2 categories. The largest: Advanced Antibiotics (54.3%), CNS & Psychiatric (45.7%).
Get Full Prince Pharma Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Prince Pharma identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Prince Pharma's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 39 individual customs records matching Prince Pharma.
- 5.Supplier Verification: Prince Pharma sources from 61 verified Indian suppliers across 2,286 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.