Prince Farma, LDA
Pharmaceutical Importer · Angola · Nutritional Supplements Focus · $7.8M Total Trade · DGFT Verified
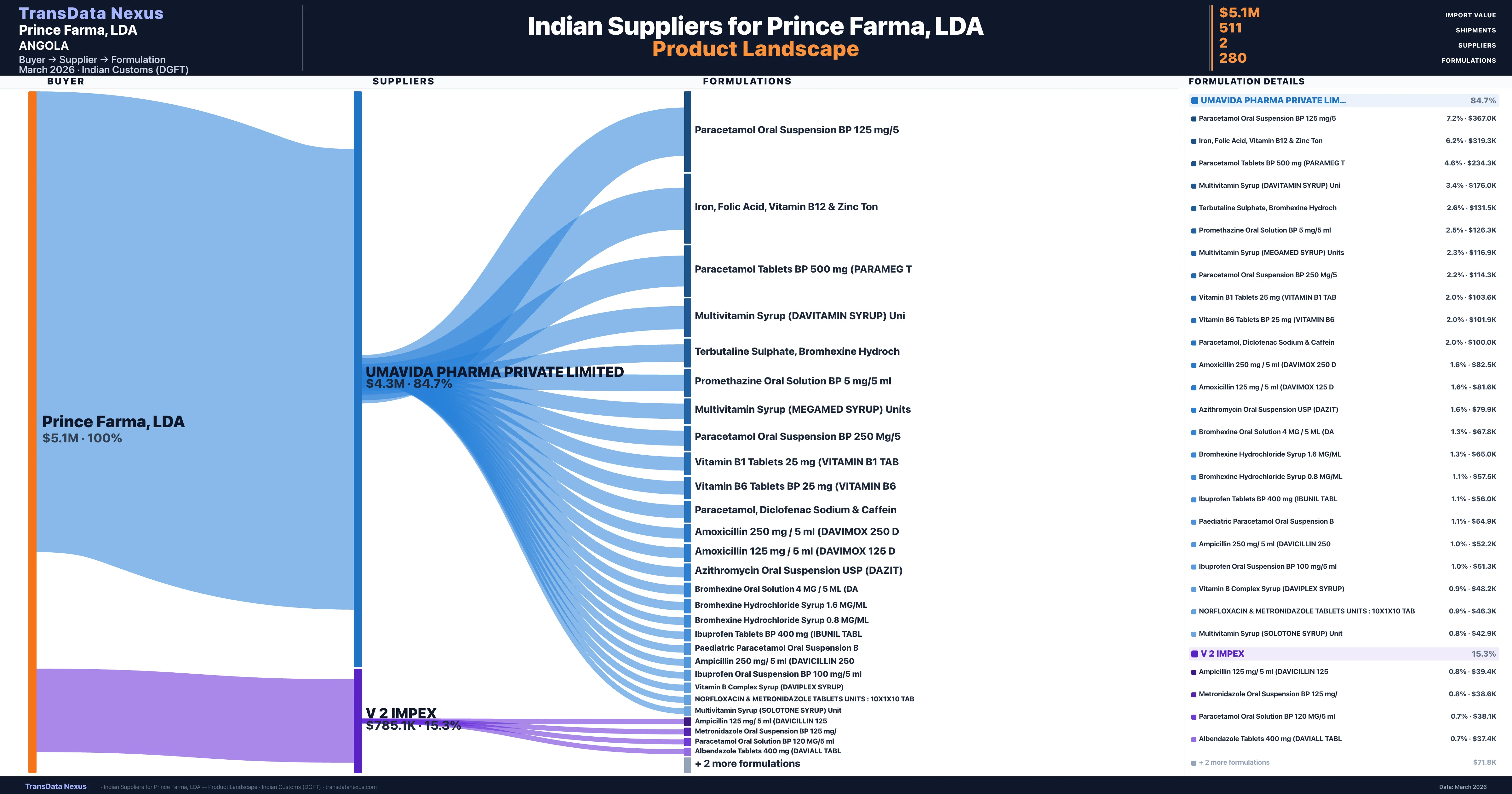
Prince Farma, LDA is a pharmaceutical importer based in Angola with a total trade value of $7.8M across 7 products in 4 therapeutic categories. Based on 158 verified import shipments from Indian Customs (DGFT) records, Prince Farma, LDA is the #1 buyer in 4 products including Vitamin, Bromhexine, Folic. Prince Farma, LDA sources from 2 verified Indian suppliers, with Umavida Pharma Private Limited accounting for 84.7% of imports.
Prince Farma, LDA — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Prince Farma, LDA?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Umavida Pharma Private Limited | $4.3M | 401 | 84.7% |
| V 2 Impex | $785.1K | 110 | 15.3% |
Prince Farma, LDA sources from 2 verified Indian suppliers across 280 distinct formulations. The sourcing is highly concentrated — Umavida Pharma Private Limited accounts for 84.7% of total imports, indicating a strategic single-source relationship.
What Formulations Does Prince Farma, LDA Import?
| Formulation | Value | Ships |
|---|---|---|
| Paracetamol oral suspension BP 125 | $367.0K | 15 |
| Iron, folic acid, vitamin b12 & zinc | $319.3K | 11 |
| Paracetamol tablets BP 500 MG (parameg | $234.3K | 11 |
| Multivitamin syrup (davitamin syrup) | $176.0K | 7 |
| Terbutaline sulphate, bromhexine | $131.5K | 5 |
| Promethazine oral solution BP 5 MG/5 ML | $126.3K | 5 |
| Multivitamin syrup (megamed syrup) | $116.9K | 6 |
| Paracetamol oral suspension BP 250 | $114.3K | 6 |
| Vitamin b1 tablets 25 MG (vitamin b1 TAB | $103.6K | 4 |
| Vitamin b6 tablets BP 25 MG (vitamin | $101.9K | 4 |
| Paracetamol, diclofenac sodium & | $100.0K | 2 |
| Amoxicillin 250 MG / 5 ML (davimox 250 | $82.5K | 7 |
| Amoxicillin 125 MG / 5 ML (davimox 125 | $81.6K | 7 |
| Azithromycin oral suspension USP (dazit) | $79.9K | 6 |
| Bromhexine oral solution 4 MG / 5 ML | $67.8K | 5 |
Prince Farma, LDA imports 280 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Prince Farma, LDA Import?
Top Products by Import Value
Prince Farma, LDA Therapeutic Categories — 4 Specializations
Prince Farma, LDA imports across 4 therapeutic categories, with Nutritional Supplements (56.4%), Respiratory & OTC (23.7%), Vitamins & Supplements (16.7%) representing the largest segments. The portfolio is concentrated — top 5 products = 92% of total imports.
Nutritional Supplements
2 products · 56.4% · $4.4M
Respiratory & OTC
2 products · 23.7% · $1.9M
Vitamins & Supplements
2 products · 16.7% · $1.3M
Antibiotics
1 products · 3.2% · $251.9K
Import Portfolio — Top 7 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Vitamin | Nutritional Supplements | $3.6M | 73 | 2.5% | 1 |
| 2 | Bromhexine | Respiratory & OTC | $1.5M | 30 | 9.2% | 1 |
| 3 | Folic | Nutritional Supplements | $750.0K | 15 | 1.3% | 1 |
| 4 | Iron | Vitamins & Supplements | $700.0K | 14 | 0.1% | 16 |
| 5 | Zinc | Vitamins & Supplements | $600.0K | 12 | 0.8% | 2 |
| 6 | Terbutaline | Respiratory & OTC | $350.0K | 7 | 10.0% | 1 |
| 7 | Tetracycline | Antibiotics | $251.9K | 7 | 1.5% | 7 |
Prince Farma, LDA imports 7 pharmaceutical products across 4 categories into Angola totaling $7.8M. The company is the #1 buyer for 4 products: Vitamin, Bromhexine, Folic, Terbutaline.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Prince Farma, LDA.
Request DemoPrince Farma, LDA — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Prince Farma, LDA is a leading pharmaceutical company based in Luanda, Angola, specializing in the distribution, sales, and marketing of pharmaceutical products. Established in 2005, the company has built a strong reputation for providing high-quality and affordable medicines to the Angolan market. Prince Farma's product portfolio includes a wide range of pharmaceutical formulations, such as tablets, capsules, syrups, and injections, sourced from reputable manufacturers worldwide. The company is ISO 9001:2015 certified, reflecting its commitment to maintaining high standards in quality management systems.
As a primary wholesaler and distributor, Prince Farma plays a pivotal role in Angola's pharmaceutical supply chain. The company collaborates with various international suppliers to import finished pharmaceutical formulations, ensuring a steady supply of essential medicines to meet the healthcare needs of the Angolan population. By focusing on quality and affordability, Prince Farma has become a trusted partner for healthcare providers and patients across the country.
2Distribution Network
Prince Farma's distribution network is strategically designed to ensure efficient delivery of pharmaceutical products throughout Angola. The company's headquarters are located in Luanda, the capital city, serving as the central hub for operations. While specific details about additional warehouse locations are not publicly disclosed, the company's extensive experience and established partnerships suggest a well-organized logistics system capable of reaching various regions within Angola. Prince Farma's collaboration with renowned logistics firms further enhances its distribution capabilities, ensuring timely and reliable delivery of products to meet the demands of the healthcare sector.
3Industry Role
In Angola's pharmaceutical supply chain, Prince Farma, LDA functions primarily as a wholesaler and distributor. The company imports finished pharmaceutical formulations from international suppliers, including those in India, and distributes them to various healthcare providers, such as hospitals, clinics, and pharmacies. By focusing on the importation and distribution of finished products, Prince Farma ensures a consistent supply of essential medicines to the Angolan market, playing a crucial role in meeting the country's healthcare needs.
Supplier Relationship Intelligence — Prince Farma, LDA
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Prince Farma's sourcing strategy exhibits a high degree of concentration, with a significant portion of its pharmaceutical imports originating from India. Between 2022 and 2026, the company imported finished pharmaceutical formulations totaling $7.8 million USD across 158 shipments, with 92.3% of this value concentrated in the top five products: vitamins, bromhexine, folic acid, iron, and zinc. This concentration indicates a strategic focus on specific therapeutic areas, likely driven by market demand and the company's established supplier relationships.
The primary suppliers, UMAVIDA PHARMA PRIVATE LIMITED and V 2 IMPEX, account for 84.7% and 15.3% of the total import value, respectively. This supplier concentration suggests a reliance on a limited number of partners for sourcing key products. While such concentration can lead to favorable terms and streamlined logistics, it also presents potential risks, including supply chain disruptions due to geopolitical issues, regulatory changes, or operational challenges faced by these suppliers.
2Supply Chain Resilience
Prince Farma's supply chain resilience is closely tied to its sourcing strategy, which heavily depends on a limited number of suppliers from India. The company's import data indicates that UMAVIDA PHARMA PRIVATE LIMITED and V 2 IMPEX are the primary sources for its pharmaceutical imports, accounting for 84.7% and 15.3% of the total import value, respectively. This concentrated supplier base may expose Prince Farma to risks associated with supply chain disruptions, such as delays, quality issues, or regulatory changes affecting these suppliers.
To enhance supply chain resilience, it would be prudent for Prince Farma to diversify its supplier base by engaging with additional manufacturers from other countries. This strategy could mitigate potential risks and ensure a more stable and reliable supply of pharmaceutical products. Additionally, evaluating the regulatory compliance and quality standards of existing and potential suppliers is essential to maintain the high-quality standards that Prince Farma is known for.
3Strategic Implications
Prince Farma's concentrated sourcing pattern, primarily from two Indian suppliers, positions the company to leverage strong relationships and potentially favorable terms. However, this strategy also exposes the company to risks associated with supply chain disruptions, such as delays, quality issues, or regulatory changes affecting these suppliers.
For Indian exporters, understanding Prince Farma's sourcing preferences and the therapeutic areas of focus can present opportunities to strengthen existing partnerships or establish new ones. By ensuring compliance with international quality standards and maintaining consistent supply capabilities, Indian exporters can position themselves as reliable partners for Prince Farma, potentially expanding their market presence in Angola.
Importing Pharmaceuticals into Angola — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Angola
1Regulatory Authority & Framework
In Angola, the pharmaceutical sector is regulated by the Regulatory Agency for Medicines and Healthcare Technologies (ARMED), operating under the Ministry of Health. ARMED is responsible for regulating and supervising the sector, licensing pharmaceuticals and health technologies for human use, ensuring the quality of healthcare products and services entering Angola, and safeguarding public health.
Key legislation governing pharmaceutical imports includes the General Law of Medicines and Medical Devices (2019), which establishes guidelines for the regulation of medicines and medical devices in Angola. This law outlines the requirements for importing medicines, including the need for special authorization from the Ministry of Health for unapproved or unavailable medicines, as well as compliance with international regulations and standards.
2Import Licensing & GMP
Import licensing for pharmaceutical products in Angola is managed by the Ministry of Health, with specific requirements outlined in the General Law of Medicines and Medical Devices (2019). Importers must obtain special authorization from the Ministry of Health to import medicines, especially those not approved or unavailable in the country. This process involves submitting a formal request, medical prescriptions, detailed medical reports, and documentation about the medicine, including its composition, dosage, manufacturer, and country of origin.
Regarding Good Manufacturing Practice (GMP) certification, Angola recognizes international standards such as EU GMP, WHO GMP, and PIC/S. Pharmaceutical products imported into Angola must comply with these standards to ensure quality and safety. Importers are required to verify that their suppliers hold valid GMP certifications from recognized authorities to meet regulatory requirements.
3Quality & Labeling
Imported pharmaceutical products in Angola must undergo batch testing to ensure they meet the required quality standards. Stability requirements are enforced to guarantee that medicines remain effective and safe throughout their shelf life. Labeling must be in Portuguese, the official language of Angola, and should include essential information such as product name, active ingredients, dosage instructions, manufacturer details, and expiration date. Serialization mandates may apply to track and trace products within the supply chain, enhancing transparency and preventing counterfeit products.
4Recent Regulatory Changes
Between 2024 and 2026, Angola has implemented policy changes affecting pharmaceutical imports, including the establishment of the Regulatory Agency for Medicines and Healthcare Technologies (ARMED) under the Ministry of Health. ARMED is responsible for regulating and supervising the pharmaceutical sector, licensing pharmaceuticals and health technologies for human use, ensuring the quality of healthcare products entering Angola, and safeguarding public health.
Additionally, Presidential Decree No. 202/21 of 26 August 2021 has streamlined the licensing process for distribution points, reducing bureaucracy and facilitating the entry of new players into the market.
Prince Farma, LDA — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Prince Farma's product strategy focuses on therapeutic areas with high demand in Angola, including nutritional supplements, respiratory and over-the-counter (OTC) medications, and vitamins and supplements. The company's import data indicates that between 2022 and 2026, 56.4% of its imports were in the nutritional supplements category, 23.7% in respiratory and OTC, and 16.7% in vitamins and supplements. This strategic focus aligns with the healthcare needs of the Angolan population, addressing prevalent health concerns and conditions.
The top five imported products—vitamins, bromhexine, folic acid, iron, and zinc—are essential in managing various health conditions, such as nutritional deficiencies, respiratory issues, and anemia. By concentrating on these products, Prince Farma aims to provide comprehensive healthcare solutions that meet the needs of its diverse clientele.
2Sourcing Profile
Prince Farma's sourcing strategy emphasizes importing finished pharmaceutical formulations from international suppliers, particularly from India. The company's import data reveals that between 2022 and 2026, it imported a total of 280 unique formulations, with a significant concentration in the top five products. This approach allows Prince Farma to offer a diverse range of products while maintaining a focus on high-demand therapeutic areas.
India's well-established pharmaceutical industry, known for its adherence to international quality standards and competitive pricing, makes it a strategic sourcing partner for Prince Farma. By leveraging these advantages, Prince Farma can ensure a consistent supply of quality medicines to the Angolan market.
Frequently Asked Questions — Prince Farma, LDA
What products does Prince Farma, LDA import from India?
Prince Farma, LDA imports 7 pharmaceutical products across 4 categories. Top imports: Vitamin ($3.6M), Bromhexine ($1.5M), Folic ($750.0K), Iron ($700.0K), Zinc ($600.0K).
Who supplies pharmaceuticals to Prince Farma, LDA from India?
Prince Farma, LDA sources from 2 verified Indian suppliers. The primary supplier is Umavida Pharma Private Limited (84.7% of imports, $4.3M).
What is Prince Farma, LDA's total pharmaceutical import value?
Prince Farma, LDA's total pharmaceutical import value from India is $7.8M, based on 158 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Prince Farma, LDA focus on?
Prince Farma, LDA imports across 4 categories. The largest: Nutritional Supplements (56.4%), Respiratory & OTC (23.7%), Vitamins & Supplements (16.7%).
Get Full Prince Farma, LDA Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Prince Farma, LDA identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Prince Farma, LDA's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 158 individual customs records matching Prince Farma, LDA.
- 5.Supplier Verification: Prince Farma, LDA sources from 2 verified Indian suppliers across 280 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
7 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.