Pharmacie Centrale DE Guinee,
Pharmaceutical Importer · Guinea · Combination Drugs Focus · $1.1M Total Trade · DGFT Verified
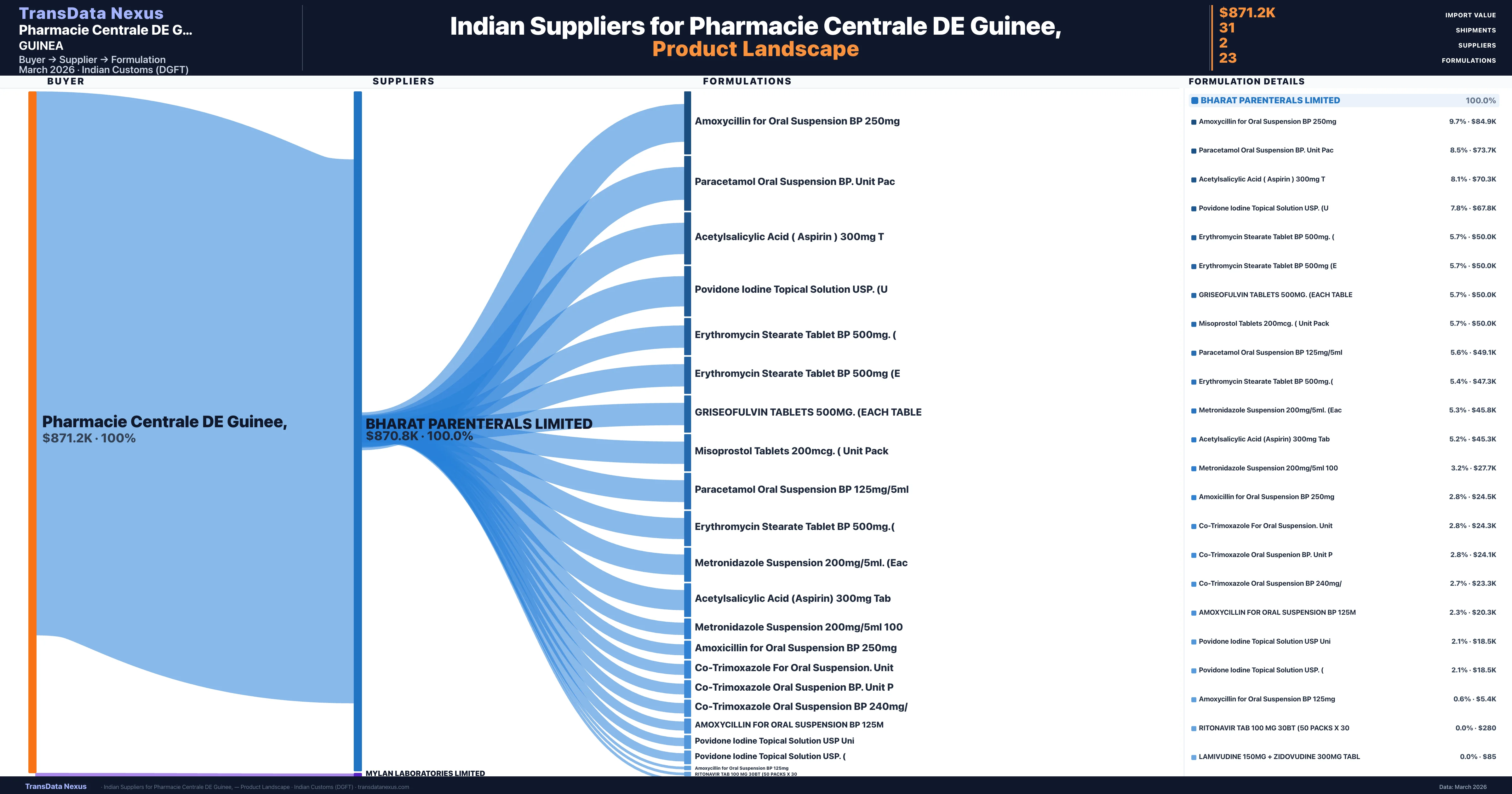
Pharmacie Centrale DE Guinee, is a pharmaceutical importer based in Guinea with a total trade value of $1.1M across 5 products in 3 therapeutic categories. Based on 41 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Pharmacie Centrale DE Guinee, sources from 2 verified Indian suppliers, with Bharat Parenterals Limited accounting for 100.0% of imports.
Pharmacie Centrale DE Guinee, — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Pharmacie Centrale DE Guinee,?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Bharat Parenterals Limited | $870.8K | 29 | 100.0% |
| Mylan Laboratories Limited | $365 | 2 | 0.0% |
Pharmacie Centrale DE Guinee, sources from 2 verified Indian suppliers across 23 distinct formulations. The sourcing is highly concentrated — Bharat Parenterals Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Pharmacie Centrale DE Guinee, Import?
| Formulation | Value | Ships |
|---|---|---|
| Amoxycillin for oral suspension BP 250MG | $84.9K | 3 |
| Paracetamol oral suspension BP. unit | $73.7K | 2 |
| Acetylsalicylic acid ( aspirin ) 300MG | $70.3K | 3 |
| Povidone iodine topical solution USP. | $67.8K | 2 |
| Erythromycin stearate tablet BP 500MG. | $50.0K | 1 |
| Erythromycin stearate tablet BP 500MG | $50.0K | 1 |
| Griseofulvin tablets 500MG. (Each | $50.0K | 1 |
| Misoprostol tablets 200mcg. ( unit | $50.0K | 1 |
| Paracetamol oral suspension BP 125MG/5ML | $49.1K | 3 |
| Erythromycin stearate tablet BP | $47.3K | 1 |
| Metronidazole suspension 200MG/5ML. | $45.8K | 1 |
| Acetylsalicylic acid (aspirin) 300MG TAB | $45.3K | 1 |
| Metronidazole suspension 200MG/5ML | $27.7K | 1 |
| Amoxicillin for oral suspension BP 250MG | $24.5K | 1 |
| Co-trimoxazole for oral suspension. | $24.3K | 1 |
Pharmacie Centrale DE Guinee, imports 23 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Pharmacie Centrale DE Guinee, Import?
Top Products by Import Value
Pharmacie Centrale DE Guinee, Therapeutic Categories — 3 Specializations
Pharmacie Centrale DE Guinee, imports across 3 therapeutic categories, with Combination Drugs (39.0%), Advanced Antibiotics (37.1%), Antimalarial & Antiparasitic (23.9%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Combination Drugs
2 products · 39.0% · $420.2K
Advanced Antibiotics
2 products · 37.1% · $400.0K
Antimalarial & Antiparasitic
1 products · 23.9% · $257.8K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Mebendazole | Antimalarial & Antiparasitic | $257.8K | 18 | 1.9% | 8 |
| 2 | Trimethoprim | Advanced Antibiotics | $250.0K | 5 | 0.9% | 14 |
| 3 | Salicylic | Combination Drugs | $212.3K | 9 | 1.3% | 14 |
| 4 | Acetylsalicylic | Combination Drugs | $207.9K | 6 | 2.6% | 8 |
| 5 | Sulfamethoxazole | Advanced Antibiotics | $150.0K | 3 | 0.8% | 14 |
Pharmacie Centrale DE Guinee, imports 5 pharmaceutical products across 3 categories into Guinea totaling $1.1M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Pharmacie Centrale DE Guinee,.
Request DemoPharmacie Centrale DE Guinee, — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Pharmacie Centrale de Guinée (PCG) is a public enterprise established on December 17, 1992, by decree number D/92/133/PRG/SGG, following the dissolution of "Pharma-Guinée." Operating under the Ministry of Health, PCG's primary mission is to ensure the consistent and regular supply of quality health products to both public and private healthcare facilities across Guinea. This includes the procurement, storage, and distribution of essential medicines and medical supplies, aiming to enhance accessibility and affordability for the Guinean population.
Headquartered in Conakry, Guinea's capital, PCG functions as the central procurement agency for the government, playing a pivotal role in the nation's pharmaceutical distribution network. The organization is governed by a Board of Directors and managed by a General Directorate, overseeing various departments dedicated to its operational functions. In recent years, PCG has undertaken significant reforms to strengthen its capacity, including the inauguration of a new storage warehouse in November 2023, aimed at bolstering its logistical and operational capabilities.
2Distribution Network
PCG's distribution network is centered in Conakry, with its main warehouse located in the Dixinn district. This strategic location facilitates efficient storage and distribution of pharmaceutical products throughout Guinea. The organization has also expanded its logistical infrastructure by inaugurating a new storage facility in November 2023, enhancing its capacity to manage and distribute medical supplies effectively.
3Industry Role
As the central procurement agency for the Guinean government, PCG serves as the primary wholesaler of pharmaceutical products in the country. It is responsible for sourcing, storing, and distributing medicines and medical supplies to both public and private healthcare institutions, ensuring a steady supply of quality health products nationwide. This central role positions PCG as a critical entity in Guinea's pharmaceutical supply chain, directly impacting the availability and accessibility of essential medicines for the population.
Supplier Relationship Intelligence — Pharmacie Centrale DE Guinee,
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
PCG's sourcing strategy exhibits a high degree of concentration, with a significant portion of its pharmaceutical imports originating from India. Between 2022 and 2026, PCG imported finished pharmaceutical formulations totaling $1.1 million USD from India, comprising 41 shipments across five products in three therapeutic categories. The top five products imported include Mebendazole ($258K), Trimethoprim ($250K), Salicylic ($212K), Acetylsalicylic ($208K), and Sulfamethoxazole ($150K). This concentration indicates a strategic choice to leverage India's robust pharmaceutical manufacturing capabilities, ensuring a consistent supply of quality medicines.
The relationship stability with Indian suppliers appears strong, as evidenced by the consistent volume of imports over the years. The primary supplier, Bharat Parenterals Limited, accounted for $871K across 29 shipments, while Mylan Laboratories Limited contributed $365K through two shipments. This sustained engagement suggests a reliable and ongoing partnership, reducing the risks associated with supplier dependency.
2Supply Chain Resilience
PCG's supply chain resilience is bolstered by its diversified sourcing strategy, primarily from India, which is known for its stringent regulatory standards and quality manufacturing practices. The consistent importation of finished pharmaceutical formulations from India indicates a stable and reliable supply chain. However, the limited number of suppliers—two verified Indian suppliers—may pose risks in terms of supply chain flexibility and responsiveness to market changes. To enhance resilience, PCG could consider diversifying its supplier base and exploring alternative sourcing options.
3Strategic Implications
PCG's concentrated sourcing from India positions it to benefit from competitive pricing and high-quality pharmaceutical products. This strategic alignment with India's pharmaceutical industry enhances the organization's ability to provide affordable and effective medicines to the Guinean population. For Indian exporters, this presents an opportunity to strengthen existing partnerships and explore avenues for expanding their product offerings to meet the diverse needs of PCG.
Importing Pharmaceuticals into Guinea — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Guinea
1Regulatory Authority & Framework
In Guinea, the regulatory authority overseeing pharmaceutical imports is the Ministry of Health, which is responsible for ensuring the safety, efficacy, and quality of medical products entering the country. Key legislation governing pharmaceutical imports includes the Law L/2018/024/AN of June 20, 2018, which pertains to medicines, health products, and the practice of pharmacy. This law outlines the regulatory framework for the importation, distribution, and use of pharmaceutical products in Guinea, ensuring compliance with international standards and safeguarding public health.
2Import Licensing & GMP
Import licensing requirements in Guinea mandate that all pharmaceutical products undergo a registration process with the Ministry of Health before being imported and distributed. This process ensures that imported medicines meet the country's safety and efficacy standards. Guinea recognizes Good Manufacturing Practice (GMP) certifications from reputable international bodies, including the World Health Organization (WHO) and the European Union (EU). Pharmaceutical companies seeking to supply PCG must ensure their products are manufactured in facilities holding valid GMP certifications from these recognized authorities.
3Quality & Labeling
Imported pharmaceutical products are subject to batch testing to verify their quality and compliance with Guinea's health standards. Stability requirements are enforced to ensure that medicines maintain their efficacy throughout their shelf life. Labeling must be in French, the official language of Guinea, and include essential information such as dosage instructions, manufacturing details, and expiration dates. Serialization mandates are in place to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, Guinea has implemented several regulatory changes affecting pharmaceutical imports. These include stricter enforcement of GMP certification requirements, updated labeling standards to include additional safety information, and enhanced batch testing protocols to improve the detection of substandard products. These reforms aim to strengthen the pharmaceutical supply chain and ensure the delivery of safe and effective medicines to the Guinean population.
Pharmacie Centrale DE Guinee, — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
PCG's focus on importing combination drugs, advanced antibiotics, and antimalarial and antiparasitic medications aligns with the prevalent health challenges in Guinea. Combination drugs are essential for treating complex infections, advanced antibiotics are crucial for combating resistant strains, and antimalarial and antiparasitic drugs are vital due to the high incidence of these diseases in the region. The market demand for these products is driven by the need to address public health concerns effectively and improve patient outcomes.
2Sourcing Profile
PCG's sourcing strategy emphasizes the procurement of generic drugs, particularly finished pharmaceutical formulations, to ensure affordability and accessibility. The preference for formulations that address a broad spectrum of diseases reflects a comprehensive approach to healthcare provision. India's established pharmaceutical industry, with its capacity for large-scale production and adherence to international quality standards, makes it a suitable partner for PCG's procurement needs.
3Market Positioning
PCG serves a diverse segment of the Guinean market, including retail pharmacies, hospitals, government tenders, and wholesale distribution. By supplying a wide range of pharmaceutical products, it ensures that various healthcare providers have access to essential medicines, thereby supporting the overall healthcare infrastructure in Guinea.
Seller's Guide — How to Become a Supplier to Pharmacie Centrale DE Guinee,
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with PCG, especially those offering products that complement the existing portfolio or address unmet needs in the Guinean market. Gaps in PCG's current sourcing include a limited number of suppliers and a narrow product range in certain therapeutic areas. Indian exporters can explore these opportunities by ensuring compliance with Guinea's regulatory requirements and demonstrating the quality and efficacy of their products.
2Requirements & Qualifications
Indian exporters aiming to supply PCG must ensure their products are manufactured in facilities holding valid GMP certifications from recognized authorities such as the WHO or EU. Additionally, compliance with Guinea's import licensing requirements, including product registration with the Ministry of Health, is mandatory. Products must meet Guinea's quality standards, including batch testing and stability requirements, and labeling must be in French, adhering to the country's regulations.
3How to Approach
To establish a relationship with PCG, Indian exporters should initiate contact through official channels, providing detailed product information and demonstrating compliance with Guinea's regulatory standards. Participating in tenders and responding to procurement notices issued by PCG can facilitate entry into the market. Developing a comprehensive regulatory filing strategy, including obtaining necessary certifications and approvals, is crucial. Timelines for approval can vary; therefore, maintaining open communication with PCG and the Ministry of Health is essential to navigate the regulatory process effectively.
Frequently Asked Questions — Pharmacie Centrale DE Guinee,
What products does Pharmacie Centrale DE Guinee, import from India?
Pharmacie Centrale DE Guinee, imports 5 pharmaceutical products across 3 categories. Top imports: Mebendazole ($257.8K), Trimethoprim ($250.0K), Salicylic ($212.3K), Acetylsalicylic ($207.9K), Sulfamethoxazole ($150.0K).
Who supplies pharmaceuticals to Pharmacie Centrale DE Guinee, from India?
Pharmacie Centrale DE Guinee, sources from 2 verified Indian suppliers. The primary supplier is Bharat Parenterals Limited (100.0% of imports, $870.8K).
What is Pharmacie Centrale DE Guinee,'s total pharmaceutical import value?
Pharmacie Centrale DE Guinee,'s total pharmaceutical import value from India is $1.1M, based on 41 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Pharmacie Centrale DE Guinee, focus on?
Pharmacie Centrale DE Guinee, imports across 3 categories. The largest: Combination Drugs (39.0%), Advanced Antibiotics (37.1%), Antimalarial & Antiparasitic (23.9%).
Get Full Pharmacie Centrale DE Guinee, Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Pharmacie Centrale DE Guinee, identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Pharmacie Centrale DE Guinee,'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 41 individual customs records matching Pharmacie Centrale DE Guinee,.
- 5.Supplier Verification: Pharmacie Centrale DE Guinee, sources from 2 verified Indian suppliers across 23 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.