Norvik Enterprises Limited
Pharmaceutical Importer · Kenya · Cardiovascular Focus · $12.8M Total Trade · DGFT Verified
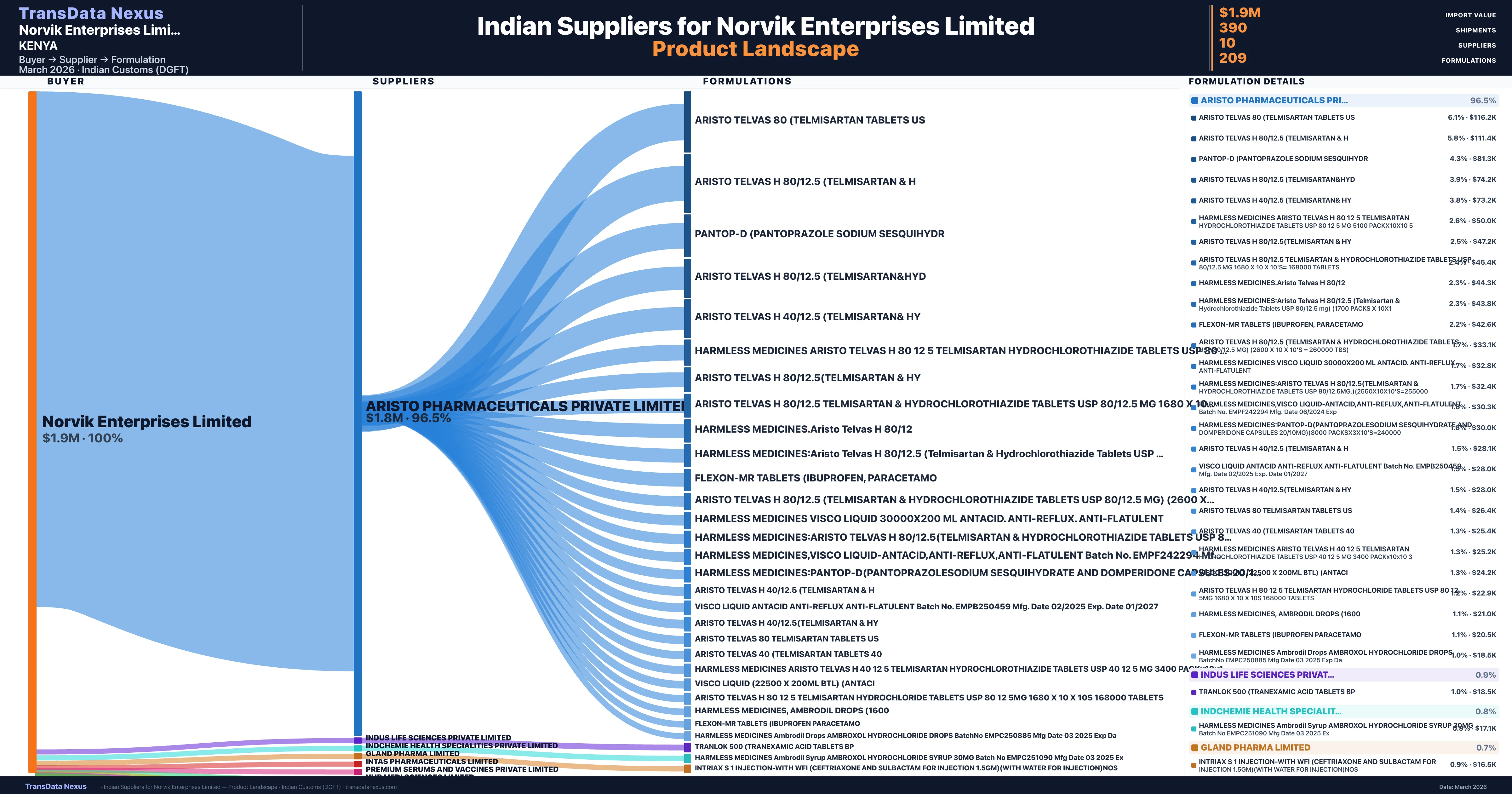
Norvik Enterprises Limited is a pharmaceutical importer based in Kenya with a total trade value of $12.8M across 5 products in 5 therapeutic categories. Based on 295 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Norvik Enterprises Limited sources from 10 verified Indian suppliers, with Aristo Pharmaceuticals Private Limited accounting for 96.5% of imports.
Norvik Enterprises Limited — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Norvik Enterprises Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Aristo Pharmaceuticals Private Limited | $1.8M | 374 | 96.5% |
| Indus Life Sciences Private Limited | $16.5K | 3 | 0.9% |
| Indchemie Health Specialities Private Limited | $14.7K | 4 | 0.8% |
| Gland Pharma Limited | $13.1K | 1 | 0.7% |
| Intas Pharmaceuticals Limited | $6.8K | 1 | 0.4% |
| Premium Serums And Vaccines Private Limited | $5.6K | 1 | 0.3% |
| Vhb Medi Sciences Limited | $4.2K | 1 | 0.2% |
| Apex Laboratories Private Limited | $3.1K | 3 | 0.2% |
| Medsource Ozone Biomedicals Private Limited | $2.4K | 1 | 0.1% |
| Premium Serums Vaccines Private Limited | $12 | 1 | 0.0% |
Norvik Enterprises Limited sources from 10 verified Indian suppliers across 209 distinct formulations. The sourcing is highly concentrated — Aristo Pharmaceuticals Private Limited accounts for 96.5% of total imports, indicating a strategic single-source relationship.
What Formulations Does Norvik Enterprises Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Aristo telvas 80 (telmisartan tablets | $116.2K | 16 |
| Aristo telvas h 80/12.5 (telmisartan & | $111.4K | 10 |
| Pantop-d (pantoprazole sodium | $81.3K | 10 |
| Aristo telvas h 80/12.5 | $74.2K | 6 |
| Aristo telvas h 40/12.5 (telmisartan& | $73.2K | 10 |
| Harmless medicines aristo telvas h 80 12 5 telmisartan hydrochlorothiazide tablets USP 80 12 5 MG 5100 packx10x10 | $50.0K | 1 |
| Aristo telvas h 80/12.5(telmisartan & | $47.2K | 4 |
| Aristo telvas h 80/12.5 telmisartan & hydrochlorothiazide tablets USP 80/12.5 MG 1680 x 10 x 10's= 168000 | $45.4K | 2 |
| Harmless medicines.aristo telvas h | $44.3K | 1 |
| Harmless medicines:aristo telvas h 80/12.5 (telmisartan & hydrochlorothiazide tablets USP 80/12.5 MG) (1700 packs x | $43.8K | 2 |
| Flexon-mr tablets (ibuprofen | $42.6K | 12 |
| Aristo telvas h 80/12.5 (telmisartan & hydrochlorothiazide tablets USP 80/12.5 MG) (2600 x 10 x 10's = 260000 tbs) | $33.1K | 1 |
| Harmless medicines visco liquid 30000x200 ML antacid. anti-reflux. | $32.8K | 1 |
| Harmless medicines:aristo telvas h 80/12.5(telmisartan & hydrochlorothiazide tablets USP 80/12.5MG. | $32.4K | 1 |
| Harmless medicines,visco liquid-antacid,anti-reflux,anti-flatulent batch no. empf242294 mfg. date 06/2024 | $30.3K | 1 |
Norvik Enterprises Limited imports 209 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Norvik Enterprises Limited Import?
Top Products by Import Value
Norvik Enterprises Limited Therapeutic Categories — 5 Specializations
Norvik Enterprises Limited imports across 5 therapeutic categories, with Cardiovascular (68.9%), Diuretics (24.1%), Gastrointestinal (5.6%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Cardiovascular
1 products · 68.9% · $8.8M
Diuretics
1 products · 24.1% · $3.1M
Gastrointestinal
1 products · 5.6% · $725.5K
Respiratory & OTC
1 products · 1.0% · $124.2K
Combination Drugs
1 products · 0.4% · $48.8K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Telmisartan | Cardiovascular | $8.8M | 177 | 0.1% | 4 |
| 2 | Hydrochlorothiazide | Diuretics | $3.1M | 62 | 0.8% | 9 |
| 3 | Domperidone | Gastrointestinal | $725.5K | 24 | 2.4% | 6 |
| 4 | Ambroxol | Respiratory & OTC | $124.2K | 16 | 0.8% | 17 |
| 5 | Ibuprofen Paracetamol | Combination Drugs | $48.8K | 16 | 2.5% | 5 |
Norvik Enterprises Limited imports 5 pharmaceutical products across 5 categories into Kenya totaling $12.8M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Norvik Enterprises Limited.
Request DemoNorvik Enterprises Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Norvik Enterprises Limited is a pharmaceutical importer and distributor based in Kampala, Uganda. Established in 1998, the company was founded with the primary objective of meeting the increasing demand for affordable and high-quality pharmaceutical products. Over the years, Norvik has grown to become one of Uganda's leading importers of pharmaceutical, vaccine, and medical sundry products. The company imports products from reputable manufacturers worldwide, including the UK, USA, Europe, Asia, South Africa, and Australia. (norvik.ug)
Norvik Enterprises Limited operates a comprehensive distribution network across Uganda, supplying to various regions such as Soroti City, Mbarara City, Fort Portal City, Kasese City, Gulu City, Kabale City, Masaka City, Mbale City, Jinja City, and Hoima City. This extensive reach ensures that essential pharmaceutical products are accessible to both urban and remote areas. The company's services encompass procurement, warehousing, distribution, sales, and marketing, enabling a continuous flow of life-saving drugs and health products to facilities nationwide. (norvik.ug)
2Distribution Network
Norvik Enterprises Limited maintains warehousing facilities in the heart of Kampala, Uganda, which house over 100 brands of pharmaceutical products and surgical tools from various pharmaceutical companies. These products are well-distributed through the company's branches across the country, including locations in Mbarara and Mbale. The warehousing facilities are equipped with numerous fire extinguishers for safety, as well as machinery like ladder platforms and skilled manpower to ensure smooth and efficient operations. (norvik.ug)
3Industry Role
In Kenya's pharmaceutical supply chain, Norvik Enterprises Limited functions as a pharmaceutical importer and distributor. While the company is based in Uganda, it has been involved in importing pharmaceutical products from India, as evidenced by its trade data. This involvement indicates that Norvik Enterprises Limited plays a role in the regional distribution of pharmaceutical products, potentially supplying to neighboring countries, including Kenya. (en.nbd.ltd)
Supplier Relationship Intelligence — Norvik Enterprises Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Norvik Enterprises Limited's sourcing strategy exhibits a high concentration, with a significant portion of its imports originating from a single supplier, Aristo Pharmaceuticals Private Limited. This supplier accounts for $1.8 million in imports, representing 96.5% of the company's total shipments. Such dependency on a single supplier can pose risks, including potential supply chain disruptions if issues arise with that supplier. However, this concentration may also reflect a strategic choice to maintain consistent product quality and pricing. The stability of this relationship is crucial for Norvik's operations, and any changes in the supplier's circumstances could significantly impact the company's supply chain.
2Supply Chain Resilience
Norvik Enterprises Limited's supply chain resilience is closely tied to its reliance on Aristo Pharmaceuticals Private Limited. The company's limited engagement with other suppliers—such as Indus Life Sciences Private Limited, Indchemie Health Specialities Private Limited, and others—suggests a lack of diversified sourcing. This limited diversification may expose Norvik to risks associated with supply chain disruptions, such as production delays or quality issues from the primary supplier. Additionally, the company's focus on a narrow range of formulations, primarily in the cardiovascular and diuretics categories, indicates a specialized product portfolio. While this specialization can lead to expertise and efficiency, it also means that any disruptions in the supply of these specific products could have a more pronounced impact on Norvik's operations.
3Strategic Implications
The high concentration of sourcing from a single supplier, Aristo Pharmaceuticals Private Limited, positions Norvik Enterprises Limited to benefit from consistent product quality and potentially favorable pricing. However, this strategy also exposes the company to significant risks, including supply chain disruptions and reduced bargaining power with other suppliers. For Indian exporters seeking to become alternative suppliers to Norvik, there is an opportunity to diversify the company's supply base by offering competitive pricing, reliable delivery schedules, and high-quality products. Building a strong relationship with Norvik could lead to increased market share in the East African region, especially if the company is looking to mitigate risks associated with over-reliance on a single supplier.
Importing Pharmaceuticals into Kenya — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Kenya
1Regulatory Authority & Framework
In Kenya, the Pharmacy and Poisons Board (PPB) serves as the primary regulatory authority overseeing the importation, distribution, and sale of pharmaceutical products. The PPB ensures that all drugs and health products meet the required safety, efficacy, and quality standards before they enter the Kenyan market. The regulatory framework governing pharmaceutical imports includes the Pharmacy and Poisons Act (Cap. 244) and the Pharmacy and Poisons (Registration of Health Products and Technologies) Rules, which outline the procedures for drug registration, importation, and market authorization.
2Import Licensing & GMP
Importers of pharmaceutical products into Kenya are required to obtain a product registration certificate from the Pharmacy and Poisons Board (PPB) for each drug they intend to import. This process involves submitting an application that includes detailed information about the product, its manufacturing process, and evidence of compliance with Good Manufacturing Practice (GMP) standards. The PPB recognizes GMP certificates from competent regulatory authorities, such as the World Health Organization (WHO) and the European Medicines Agency (EMA). Additionally, importers must obtain an import permit for each consignment, processed through the Kenya National TradeNet System (KESWS). (infotradekenya.go.ke)
3Quality & Labeling
Pharmaceutical products imported into Kenya must undergo batch testing to ensure they meet the required quality standards. Stability studies are also conducted to confirm that the products maintain their efficacy and safety throughout their shelf life. Labeling requirements include providing information in English and Kiswahili, detailing the product's name, active ingredients, dosage form, strength, batch number, manufacturing date, expiry date, storage conditions, and the manufacturer's details. Serialization mandates may be implemented to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
In January 2025, the Ministry of Health, through the Pharmacy and Poisons Board (PPB), issued a directive banning the importation, distribution, sale, or handling of unregistered pharmaceutical products in Kenya, particularly those referencing the Indian Pharmacopoeia. This move was aimed at curbing the influx of unregistered drugs into the Kenyan market and ensuring that all pharmaceutical products meet the country's regulatory standards. (citizen.digital)
Norvik Enterprises Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Norvik Enterprises Limited's product strategy focuses on importing pharmaceutical formulations in the cardiovascular, diuretics, and gastrointestinal therapeutic categories. The company's top five imported products include Telmisartan, Hydrochlorothiazide, Domperidone, Ambroxol, and Ibuprofen Paracetamol, with Telmisartan accounting for the largest import value at $8.8 million. This focus aligns with the prevalent health concerns in the East African region, where cardiovascular diseases and related conditions are significant public health issues. The demand for these products is driven by the need for effective treatments for hypertension, heart failure, and other cardiovascular conditions.
2Sourcing Profile
Norvik Enterprises Limited's sourcing strategy is heavily concentrated on importing finished pharmaceutical formulations from India, with a particular emphasis on products manufactured by Aristo Pharmaceuticals Private Limited. This approach suggests a preference for sourcing from suppliers that offer a comprehensive range of products meeting international quality standards. The company's limited engagement with other suppliers indicates a strategic choice to maintain consistency in product quality and supply reliability.
3Market Positioning
Based on its product mix, Norvik Enterprises Limited primarily serves the wholesale distribution segment of the Kenyan pharmaceutical market. The company's focus on importing and distributing pharmaceutical formulations positions it to supply a wide range of healthcare facilities, including hospitals, clinics, and pharmacies, with essential medications. By ensuring a steady supply of high-quality pharmaceutical products, Norvik plays a crucial role in meeting the healthcare needs of the East African region.
Seller's Guide — How to Become a Supplier to Norvik Enterprises Limited
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Norvik Enterprises Limited, especially those offering products in the cardiovascular, diuretics, and gastrointestinal therapeutic categories. By providing competitive pricing, reliable delivery schedules, and adherence to international quality standards, new suppliers can position themselves as viable alternatives to existing suppliers. Diversifying Norvik's supplier base can enhance supply chain resilience and mitigate risks associated with over-reliance on a single supplier.
2Requirements & Qualifications
Indian exporters seeking to supply pharmaceutical products to Norvik Enterprises Limited and the Kenyan market must ensure that their products are registered with the Pharmacy and Poisons Board (PPB) in Kenya. This involves obtaining a product registration certificate and complying with the PPB's requirements, including providing evidence of Good Manufacturing Practice (GMP) certification from recognized authorities such as the World Health Organization (WHO) or the European Medicines Agency (EMA). Additionally, exporters must adhere to Kenya's import regulations, including obtaining import permits through the Kenya National TradeNet System (KESWS).
3How to Approach
To establish a relationship with Norvik Enterprises Limited, Indian exporters should initiate contact by providing detailed product information, including specifications, GMP certifications,
Frequently Asked Questions — Norvik Enterprises Limited
What products does Norvik Enterprises Limited import from India?
Norvik Enterprises Limited imports 5 pharmaceutical products across 5 categories. Top imports: Telmisartan ($8.8M), Hydrochlorothiazide ($3.1M), Domperidone ($725.5K), Ambroxol ($124.2K), Ibuprofen Paracetamol ($48.8K).
Who supplies pharmaceuticals to Norvik Enterprises Limited from India?
Norvik Enterprises Limited sources from 10 verified Indian suppliers. The primary supplier is Aristo Pharmaceuticals Private Limited (96.5% of imports, $1.8M).
What is Norvik Enterprises Limited's total pharmaceutical import value?
Norvik Enterprises Limited's total pharmaceutical import value from India is $12.8M, based on 295 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Norvik Enterprises Limited focus on?
Norvik Enterprises Limited imports across 5 categories. The largest: Cardiovascular (68.9%), Diuretics (24.1%), Gastrointestinal (5.6%).
Get Full Norvik Enterprises Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Norvik Enterprises Limited identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Norvik Enterprises Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 295 individual customs records matching Norvik Enterprises Limited.
- 5.Supplier Verification: Norvik Enterprises Limited sources from 10 verified Indian suppliers across 209 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
5 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.