National Medical Stores
Pharmaceutical Importer · Uganda · Antiviral & HIV Medications Focus · $12.2M Total Trade · DGFT Verified
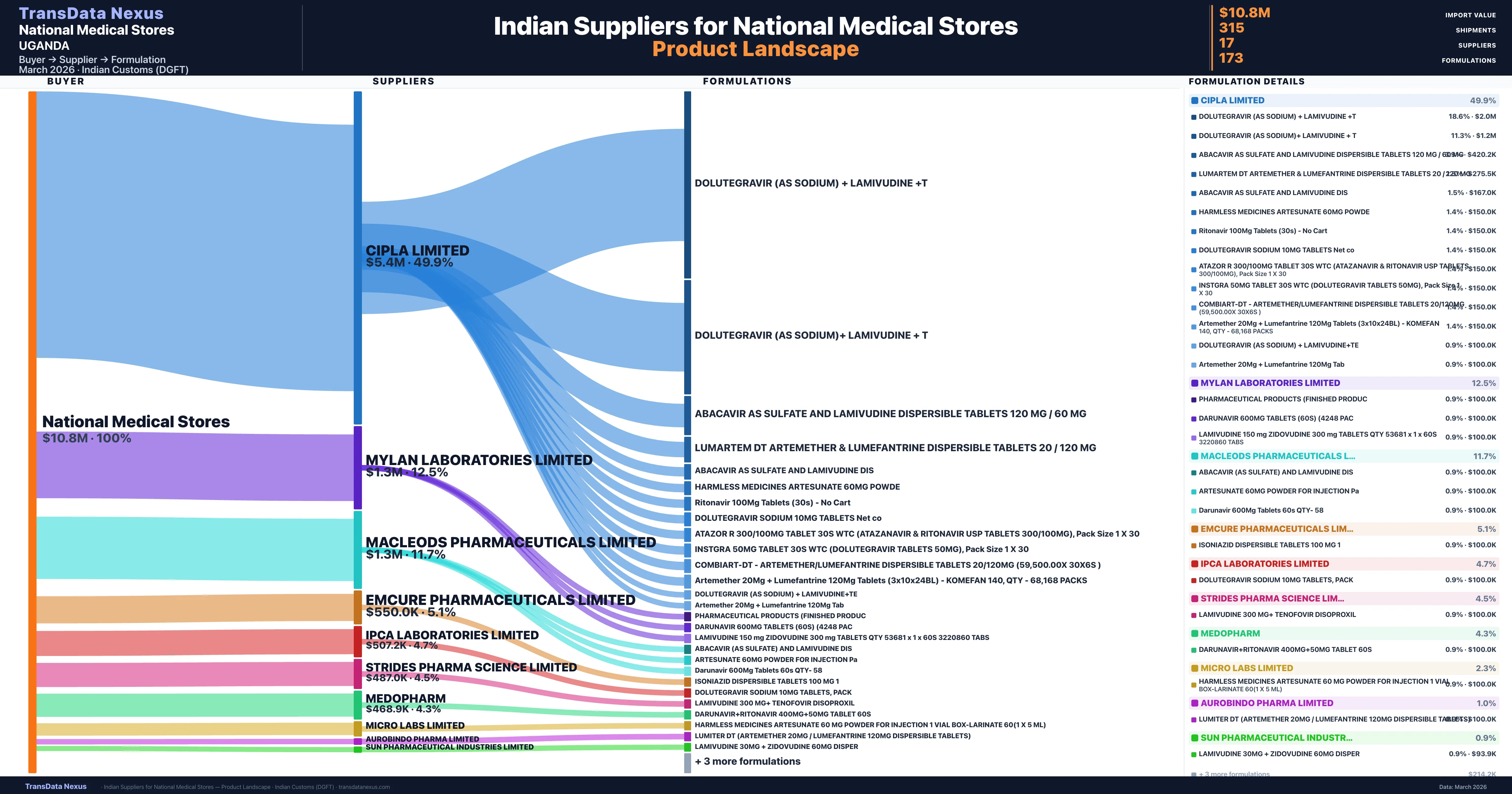
National Medical Stores is a pharmaceutical importer based in Uganda with a total trade value of $12.2M across 8 products in 3 therapeutic categories. Based on 244 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. National Medical Stores sources from 17 verified Indian suppliers, with Cipla Limited accounting for 49.9% of imports.
National Medical Stores — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to National Medical Stores?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Cipla Limited | $5.4M | 156 | 49.9% |
| Mylan Laboratories Limited | $1.3M | 35 | 12.5% |
| Macleods Pharmaceuticals Limited | $1.3M | 30 | 11.7% |
| Emcure Pharmaceuticals Limited | $550.0K | 11 | 5.1% |
| Ipca Laboratories Limited | $507.2K | 11 | 4.7% |
| Strides Pharma Science Limited | $487.0K | 14 | 4.5% |
| Medopharm | $468.9K | 24 | 4.3% |
| Micro Labs Limited | $250.0K | 5 | 2.3% |
| Aurobindo Pharma Limited | $108.9K | 5 | 1.0% |
| Sun Pharmaceutical Industries Limited | $100.0K | 2 | 0.9% |
| Hetero Labs Limited | $100.0K | 2 | 0.9% |
| Manarti Exports | $67.3K | 15 | 0.6% |
| Panacea Biotec Limited | $50.0K | 1 | 0.5% |
| Maneesh Pharmaceuticals Limited | $50.0K | 1 | 0.5% |
| Lupin Limited | $50.0K | 1 | 0.5% |
| Stallion Laboratories Private Limited | $10.9K | 1 | 0.1% |
| Galentic Pharma India Private Limited | $4.3K | 1 | 0.0% |
National Medical Stores sources from 17 verified Indian suppliers across 173 distinct formulations. The supply base is diversified across 17 suppliers, reducing single-source dependency risk.
What Formulations Does National Medical Stores Import?
| Formulation | Value | Ships |
|---|---|---|
| Dolutegravir (as sodium) + lamivudine | $2.0M | 42 |
| Dolutegravir (as sodium)+ lamivudine + | $1.2M | 25 |
| Abacavir as sulfate and lamivudine dispersible tablets 120 MG / 60 MG | $420.2K | 10 |
| Lumartem dt artemether & lumefantrine dispersible tablets 20 / 120 MG | $275.5K | 7 |
| Abacavir as sulfate and lamivudine | $167.0K | 4 |
| Harmless medicines artesunate 60MG | $150.0K | 3 |
| Ritonavir 100MG tablets (30s) - no | $150.0K | 3 |
| Dolutegravir sodium 10MG tablets net | $150.0K | 3 |
| Atazor r 300/100MG tablet 30s wtc (atazanavir & ritonavir USP tablets 300/100MG), Pack size 1 x | $150.0K | 3 |
| Instgra 50MG tablet 30s wtc (dolutegravir tablets 50MG), Pack size 1 x | $150.0K | 3 |
| Combiart-dt - artemether/lumefantrine dispersible tablets 20/120MG (59,500.00x 30x6s ) | $150.0K | 3 |
| Artemether 20MG + lumefantrine 120MG tablets (3x10x24bl) - komefan 140, qty - 68,168 | $150.0K | 3 |
| Dolutegravir (as sodium) + | $100.0K | 2 |
| Artemether 20MG + lumefantrine 120MG TAB | $100.0K | 2 |
| Pharmaceutical products (finished | $100.0K | 2 |
National Medical Stores imports 173 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does National Medical Stores Import?
National Medical Stores Therapeutic Categories — 3 Specializations
National Medical Stores imports across 3 therapeutic categories, with Antiviral & HIV Medications (63.9%), Nutritional Supplements (18.0%), Antimalarial & Antiparasitic (18.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 90% of total imports.
Antiviral & HIV Medications
5 products · 63.9% · $7.8M
Nutritional Supplements
1 products · 18.0% · $2.2M
Antimalarial & Antiparasitic
2 products · 18.0% · $2.2M
Import Portfolio — Top 8 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Dolutegravir | Antiviral & HIV Medications | $4.8M | 97 | 0.1% | 7 |
| 2 | Sodium | Nutritional Supplements | $2.2M | 44 | 0.6% | 14 |
| 3 | Artemether | Antimalarial & Antiparasitic | $1.6M | 31 | 0.2% | 2 |
| 4 | Tenofovir | Antiviral & HIV Medications | $1.3M | 25 | 0.0% | 7 |
| 5 | Ritonavir | Antiviral & HIV Medications | $1.1M | 22 | 0.3% | 8 |
| 6 | Artesunate | Antimalarial & Antiparasitic | $650.0K | 13 | 0.4% | 3 |
| 7 | Atazanavir | Antiviral & HIV Medications | $300.0K | 6 | 0.4% | 14 |
| 8 | Darunavir | Antiviral & HIV Medications | $300.0K | 6 | 0.5% | 3 |
National Medical Stores imports 8 pharmaceutical products across 3 categories into Uganda totaling $12.2M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for National Medical Stores.
Request DemoNational Medical Stores — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
National Medical Stores (NMS) is a government-owned organization in Uganda, established in 1993 under the National Medical Stores Act. Headquartered in Entebbe, NMS is mandated to procure, store, and distribute essential medicines and medical supplies to all public health facilities across the country, including hospitals, health centers, and institutions associated with the police, army, and prisons. In August 2012, its mandate was further expanded to include the distribution of vaccines nationwide. (nms.go.ug)
Operating as a statutory corporation, NMS plays a pivotal role in Uganda's healthcare supply chain by ensuring the availability of quality medical commodities in public health facilities. The organization is governed by a 15-person board of directors, with a management team led by General Manager/CEO Moses Kamabare. (nms.go.ug)
2Distribution Network
NMS operates a comprehensive distribution network to ensure timely delivery of medical supplies across Uganda. The central warehouse is located in Kajjansi, approximately 21 kilometers northeast of the headquarters in Entebbe. This facility, completed in 2019, is equipped with state-of-the-art storage capabilities, including cold storage rooms and a quality control laboratory. Additionally, NMS has eight regional distribution centers strategically located in various districts to facilitate efficient delivery. (nms.go.ug)
The organization employs a GPS-monitored fleet to deliver medicines and supplies across all districts of Uganda, ensuring reliable and timely distribution to over 3,200 public health facilities. (nms.go.ug)
3Industry Role
NMS serves as the primary wholesaler and distributor of essential medicines and medical supplies to public health facilities in Uganda. Its role is central to the country's healthcare supply chain, ensuring that government health facilities have access to necessary medical commodities. By managing procurement, storage, and distribution, NMS acts as a critical intermediary between pharmaceutical suppliers and healthcare providers. (nms.go.ug)
Supplier Relationship Intelligence — National Medical Stores
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
NMS's sourcing strategy exhibits a high degree of concentration, with the top five products accounting for 89.8% of total imports from India. This concentration suggests a strategic focus on key therapeutic areas, potentially leveraging volume discounts and streamlined logistics. However, such dependency may pose risks if there are disruptions in the supply of these specific products. The shipment data indicates a stable relationship with Indian suppliers, as evidenced by the consistent number of shipments over the years. For instance, CIPLA LIMITED has been a significant supplier, accounting for 49.9% of the total import value with 156 shipments. (nms.go.ug)
2Supply Chain Resilience
NMS's supply chain resilience is bolstered by its diversified supplier base, comprising 17 unique Indian suppliers. This diversity mitigates risks associated with over-reliance on a single supplier. The organization's focus on finished pharmaceutical formulations, rather than raw APIs or bulk drugs, ensures that products meet the necessary quality standards for direct use in healthcare settings. Additionally, the establishment of a modern warehouse in Kajjansi enhances storage capabilities and supply chain resilience. (nms.go.ug)
3Strategic Implications
The concentrated sourcing pattern of NMS positions it to negotiate favorable terms with key suppliers, potentially securing better pricing and priority in supply. For Indian exporters, this presents an opportunity to strengthen relationships with NMS by ensuring consistent product quality and reliability. Suppliers that can offer a diverse range of products within the therapeutic areas prioritized by NMS may find increased opportunities for collaboration. (nms.go.ug)
Importing Pharmaceuticals into Uganda — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Uganda
1Regulatory Authority & Framework
The National Drug Authority (NDA) is the regulatory body responsible for overseeing the importation and distribution of pharmaceutical products in Uganda. Established under the National Drug Policy and Authority Act (Cap 206), the NDA ensures that medicines supplied in Uganda meet consistent high standards of quality, safety, and efficacy. (nda.or.ug)
The NDA's regulations stipulate that entities importing drugs into Uganda must possess a valid import license issued by the Authority. Additionally, a license to operate a pharmacy is required to be eligible for an import license. Each consignment of drugs must also receive a Verification Certificate from the NDA prior to importation. (nda.or.ug)
2Import Licensing & GMP
Import licensing in Uganda is governed by the National Drug Policy and Authority Act and associated regulations. Entities seeking to import drugs must obtain an import license from the NDA, which is valid for one year. To be eligible for this license, the entity must hold a valid license to operate a pharmacy. Furthermore, a Verification Certificate from the NDA is required for each consignment of drugs prior to importation. (nda.or.ug)
The NDA also enforces Good Manufacturing Practice (GMP) standards for pharmaceutical products. Manufacturers, including those from India, must comply with these standards to ensure product quality. The NDA conducts inspections of foreign manufacturing sites to verify compliance with GMP before approving marketing authorization applications. (nda.or.ug)
3Quality & Labeling
Pharmaceutical products imported into Uganda must undergo batch testing to ensure they meet the required quality standards. Stability requirements are enforced to guarantee that products remain effective throughout their shelf life. Labeling must be in English, and include essential information such as dosage instructions, storage conditions, and expiration dates. Serialization mandates are implemented to enhance traceability and prevent counterfeit products from entering the market. (nda.or.ug)
4Recent Regulatory Changes
Between 2024 and 2026, the NDA has implemented stricter enforcement of import licensing requirements, emphasizing the need for valid import licenses and compliance with GMP standards. These changes aim to enhance the safety and efficacy of pharmaceutical products in Uganda. Additionally, the NDA has increased post-market surveillance to monitor the quality and safety of drugs available in the market. (nda.or.ug)
National Medical Stores — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
NMS's focus on antiviral and HIV medications (63.9%), nutritional supplements (18.0%), and antimalarial and antiparasitic drugs (18.0%) aligns with Uganda's public health priorities. The high import values of products like Dolutegravir ($4.8M) and Artemether ($1.6M) reflect the significant demand for these treatments. This strategic product selection is driven by the prevalence of HIV/AIDS and malaria in Uganda, necessitating a steady supply of effective medications. (nms.go.ug)
2Sourcing Profile
NMS's sourcing strategy emphasizes finished pharmaceutical formulations, ensuring that imported products are ready for direct use in healthcare settings. India's robust pharmaceutical manufacturing sector, known for its compliance with international quality standards, makes it a preferred source for these formulations. The diverse supplier base, including companies like CIPLA LIMITED and MYLAN LABORATORIES LIMITED, provides NMS with a reliable supply chain for essential medicines. (nms.go.ug)
3Market Positioning
By focusing on essential medicines and medical supplies, NMS serves the public healthcare sector in Uganda, including government hospitals, health centers, and institutions associated with the police, army, and prisons. Its role as a primary wholesaler and distributor positions it as a critical player in ensuring the availability of quality medical commodities in the public health system. (nms.go.ug)
Seller's Guide — How to Become a Supplier to National Medical Stores
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with NMS, especially those offering a diverse range of products within the therapeutic areas prioritized by NMS. Suppliers that can demonstrate consistent product quality, reliability, and compliance with regulatory standards may find increased opportunities for collaboration. However, the existing supplier base is well-established, so new entrants must offer compelling value propositions to gain a foothold. (nms.go.ug)
2Requirements & Qualifications
Indian exporters seeking to supply NMS must ensure that their products comply with the NDA's GMP standards. They must also obtain marketing authorization from the NDA, which involves inspections of manufacturing sites and product evaluations. Additionally, products must meet the NDA's quality control requirements, including batch testing and stability assessments. (nda.or.ug)
3How to Approach
Indian exporters should initiate contact with NMS by demonstrating their product's compliance with NDA standards and their ability to meet the specific needs of the Ugandan market. Participating in tenders and responding to NMS's procurement notices can provide avenues for collaboration. Understanding the regulatory filing process with the NDA and ensuring timely submission of required documentation is crucial.
Frequently Asked Questions — National Medical Stores
What products does National Medical Stores import from India?
National Medical Stores imports 8 pharmaceutical products across 3 categories. Top imports: Dolutegravir ($4.8M), Sodium ($2.2M), Artemether ($1.6M), Tenofovir ($1.3M), Ritonavir ($1.1M).
Who supplies pharmaceuticals to National Medical Stores from India?
National Medical Stores sources from 17 verified Indian suppliers. The primary supplier is Cipla Limited (49.9% of imports, $5.4M).
What is National Medical Stores's total pharmaceutical import value?
National Medical Stores's total pharmaceutical import value from India is $12.2M, based on 244 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does National Medical Stores focus on?
National Medical Stores imports across 3 categories. The largest: Antiviral & HIV Medications (63.9%), Nutritional Supplements (18.0%), Antimalarial & Antiparasitic (18.0%).
Get Full National Medical Stores Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: National Medical Stores identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as National Medical Stores's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 244 individual customs records matching National Medical Stores.
- 5.Supplier Verification: National Medical Stores sources from 17 verified Indian suppliers across 173 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
8 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.