National Center FOR Hiv/aids
Pharmaceutical Importer · Cambodia · Antifungals Focus · $450.0K Total Trade · DGFT Verified
National Center FOR Hiv/aids is a pharmaceutical importer based in Cambodia with a total trade value of $450.0K across 2 products in 2 therapeutic categories. Based on 9 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. National Center FOR Hiv/aids sources from 5 verified Indian suppliers, with Mylan Laboratories Limited accounting for 56.7% of imports.
National Center FOR Hiv/aids — Import Portfolio & Supplier Network
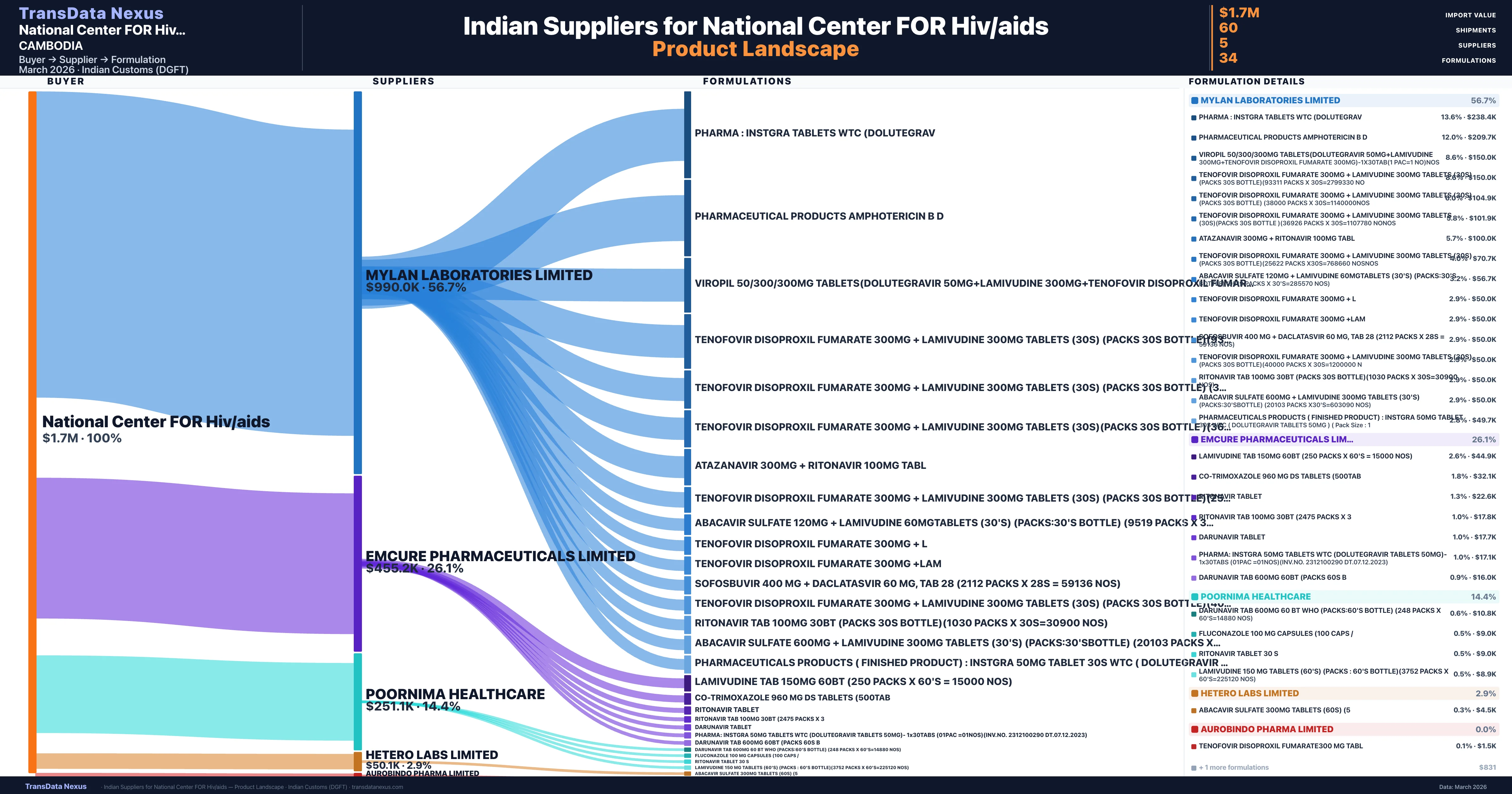
Who Are the Verified Indian Suppliers to National Center FOR Hiv/aids?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Mylan Laboratories Limited | $990.0K | 31 | 56.7% |
| Emcure Pharmaceuticals Limited | $455.2K | 11 | 26.1% |
| Poornima Healthcare | $251.1K | 12 | 14.4% |
| Hetero Labs Limited | $50.1K | 5 | 2.9% |
| Aurobindo Pharma Limited | $821 | 1 | 0.0% |
National Center FOR Hiv/aids sources from 5 verified Indian suppliers across 34 distinct formulations. The supply base is diversified across 5 suppliers, reducing single-source dependency risk.
What Formulations Does National Center FOR Hiv/aids Import?
| Formulation | Value | Ships |
|---|---|---|
| Pharma : instgra tablets wtc | $238.4K | 5 |
| Pharmaceutical products amphotericin b | $209.7K | 6 |
| Viropil 50/300/300MG tablets(dolutegravir 50MG+lamivudine 300MG+tenofovir disoproxil fumarate 300MG)-1x30tab(1 pac=1 no | $150.0K | 3 |
| Tenofovir disoproxil fumarate 300MG + lamivudine 300MG tablets (30s) (packs 30s bottle)(93311 packs x 30s=2799330 | $150.0K | 3 |
| Tenofovir disoproxil fumarate 300MG + lamivudine 300MG tablets (30s) (packs 30s bottle) (38000 packs x | $104.9K | 3 |
| Tenofovir disoproxil fumarate 300MG + lamivudine 300MG tablets (30s)(packs 30s bottle )(36926 packs x 30s=1107780 | $101.9K | 3 |
| Atazanavir 300MG + ritonavir 100MG | $100.0K | 2 |
| Tenofovir disoproxil fumarate 300MG + lamivudine 300MG tablets (30s) (packs 30s bottle)(25622 packs x30s=768660 | $70.7K | 3 |
| Abacavir sulfate 120MG + lamivudine 60mgtablets (30's) (packs:30's bottle) (9519 packs x 30's=285570 nos) | $56.7K | 2 |
| Tenofovir disoproxil fumarate 300MG + | $50.0K | 1 |
| Tenofovir disoproxil fumarate 300MG | $50.0K | 1 |
| Sofosbuvir 400 MG + daclatasvir 60 MG, TAB 28 (2112 packs x 28s = 59136 nos) | $50.0K | 1 |
| Tenofovir disoproxil fumarate 300MG + lamivudine 300MG tablets (30s) (packs 30s bottle)(40000 packs x 30s=1200000 | $50.0K | 1 |
| Ritonavir TAB 100MG 30bt (packs 30s bottle)(1030 packs x 30s=30900 nos) | $50.0K | 1 |
| Abacavir sulfate 600MG + lamivudine 300MG tablets (30's) (packs:30'sbottle) (20103 packs x30's=603090 nos) | $50.0K | 1 |
National Center FOR Hiv/aids imports 34 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does National Center FOR Hiv/aids Import?
Top Products by Import Value
National Center FOR Hiv/aids Therapeutic Categories — 2 Specializations
National Center FOR Hiv/aids imports across 2 therapeutic categories, with Antifungals (66.7%), Antiviral & HIV Medications (33.3%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antifungals
1 products · 66.7% · $300.0K
Antiviral & HIV Medications
1 products · 33.3% · $150.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Amphotericin | Antifungals | $300.0K | 6 | 0.3% | 16 |
| 2 | Atazanavir | Antiviral & HIV Medications | $150.0K | 3 | 0.2% | 13 |
National Center FOR Hiv/aids imports 2 pharmaceutical products across 2 categories into Cambodia totaling $450.0K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for National Center FOR Hiv/aids.
Request DemoNational Center FOR Hiv/aids — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
The National Center for HIV/AIDS, Dermatology and STDs (NCHADS) is a Cambodian government agency operating under the Ministry of Health. Established to manage the treatment and prevention of HIV/AIDS and sexually transmitted diseases, NCHADS plays a pivotal role in Cambodia's public health infrastructure. Its main offices are located in Phnom Penh, the capital city. (nchads.gov.kh)
NCHADS is responsible for developing and disseminating policies, strategies, and guidelines for the implementation of the Continuum of Care in Cambodia. This includes planning, coordinating, and supervising the scaling-up and expansion of HIV/AIDS and STD services. Additionally, NCHADS assists in identifying the needs for drugs and equipment for the Continuum of Care and monitors and evaluates the different components to improve the quality of care and services.
2Distribution Network
As a government agency, NCHADS operates a centralized distribution network for pharmaceuticals and medical supplies related to HIV/AIDS and STDs. While specific details about warehouse locations and logistics capabilities are not publicly disclosed, NCHADS collaborates with various health departments and organizations to ensure the effective distribution of medical supplies across Cambodia. This collaboration includes working with Provincial Health Departments and other local NGOs to support people living with HIV.
3Industry Role
NCHADS functions as a central authority in Cambodia's pharmaceutical supply chain, primarily focusing on the procurement, distribution, and management of HIV/AIDS and STD-related medical products. While it does not operate as a traditional wholesaler or distributor, NCHADS coordinates with various stakeholders, including the Ministry of Health, Provincial Health Departments, and non-governmental organizations, to ensure the availability and distribution of essential medical supplies.
Supplier Relationship Intelligence — National Center FOR Hiv/aids
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
NCHADS's sourcing strategy reveals a high concentration in its pharmaceutical imports, with a total import value of $450,000 USD from India across nine shipments. The top two products imported are Amphotericin ($300,000 USD) and Atazanavir ($150,000 USD), accounting for 66.7% and 33.3% of the total import value, respectively. This concentration indicates a strategic focus on specific therapeutic areas critical to HIV/AIDS treatment.
The shipment data suggests a stable relationship with Indian suppliers, as evidenced by the consistent import volumes over the period. The primary suppliers include Mylan Laboratories Limited, Emcure Pharmaceuticals Limited, and Poornima Healthcare, which collectively account for 97.8% of the total import value. This indicates a reliance on a select group of suppliers for essential HIV/AIDS medications.
2Supply Chain Resilience
NCHADS's supply chain resilience is supported by its partnerships with reputable Indian pharmaceutical manufacturers, such as Mylan Laboratories Limited, Emcure Pharmaceuticals Limited, and Poornima Healthcare. These suppliers are known for their adherence to Good Manufacturing Practices (GMP) and international quality standards, ensuring the reliability and safety of the imported medications. However, the concentration of imports from a limited number of suppliers may pose risks in the event of supply disruptions. Diversifying the supplier base and establishing backup suppliers could enhance the resilience of NCHADS's supply chain.
3Strategic Implications
The concentrated sourcing pattern of NCHADS positions it as a significant purchaser of specific HIV/AIDS medications from India, potentially offering leverage in negotiations with suppliers. For Indian exporters, this presents an opportunity to strengthen relationships with NCHADS by ensuring consistent supply and quality, thereby securing a stable market for their products. Additionally, understanding NCHADS's procurement needs and aligning product offerings accordingly can enhance the competitiveness of Indian exporters in the Cambodian market.
Importing Pharmaceuticals into Cambodia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Cambodia
1Regulatory Authority & Framework
The Ministry of Health (MoH) is the primary regulatory authority overseeing pharmaceutical imports in Cambodia. The Law on the Management of Pharmaceuticals governs the importation, distribution, and sale of pharmaceutical products, including requirements for import licenses and product registration. According to this law, authorization from the MoH is required for the importation of pharmaceuticals, and all imported pharmaceutical products must be registered with the MoH before distribution. (wipo.int)
2Import Licensing & GMP
Import licensing for pharmaceuticals in Cambodia is managed by the MoH. All imported medicines and medical products must be registered with the MoH, which involves submitting an application form, GMP or ISO certificates, a free sale certificate, a letter of authorization, and the product’s manual. The registration process typically takes three to six months, depending on documentation and MoH workload.
3Quality & Labeling
Imported pharmaceutical products must undergo laboratory testing by the MoH to ensure quality and safety. All imported pharmaceutical products are required to have at least 18 months validity before the expiry date. Medical devices are classified into four categories according to their levels of risk: low, fairly low, fairly high, and high. Companies can register their imported products online via the MoH registration platform known as the Cambodia Pharmaceutical Online Registration System (CamPORS).
4Recent Regulatory Changes
Between 2024 and 2026, Cambodia has implemented several policy changes affecting pharmaceutical imports. These include the introduction of the Cambodia Pharmaceutical Online Registration System (CamPORS) to streamline the registration process for imported pharmaceutical products. Additionally, the MoH has emphasized the need for GMP or ISO certificates from exporting companies to ensure product quality. These changes aim to enhance the efficiency and safety of pharmaceutical imports into Cambodia.
National Center FOR Hiv/aids — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
NCHADS's focus on importing Amphotericin and Atazanavir aligns with its mission to provide comprehensive care for individuals living with HIV/AIDS. Amphotericin is essential for treating fungal infections, which are common among immunocompromised patients, while Atazanavir is a critical component of antiretroviral therapy. The demand for these products is driven by the need to manage opportunistic infections and maintain viral suppression in HIV-positive individuals.
2Sourcing Profile
NCHADS's sourcing strategy emphasizes the procurement of generic antiretroviral medications, primarily from India, known for its robust pharmaceutical manufacturing sector. The focus on specific formulations, such as Amphotericin and Atazanavir, indicates a targeted approach to meet the therapeutic needs of the HIV/AIDS population in Cambodia. This strategy ensures access to essential medications while managing costs effectively.
3Market Positioning
By importing and distributing essential HIV/AIDS medications, NCHADS serves the public health sector in Cambodia, including government hospitals, clinics, and health programs. Its role is crucial in ensuring the availability of life-saving treatments for individuals living with HIV/AIDS, thereby positioning it as a key player in the Cambodian healthcare system.
Seller's Guide — How to Become a Supplier to National Center FOR Hiv/aids
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with NCHADS by offering high-quality, cost-effective generic antiretroviral medications. To identify gaps in NCHADS's current sourcing, potential suppliers should analyze the therapeutic areas and formulations currently imported and assess the demand for additional products. Building relationships with NCHADS and understanding its procurement processes can facilitate entry into the Cambodian market.
2Requirements & Qualifications
Indian exporters seeking to supply NCHADS must ensure their products are registered with the Cambodian MoH, which involves providing GMP or ISO certificates, a free sale certificate, a letter of authorization, and the product’s manual. Additionally, products must have at least 18 months validity before the expiry date. Compliance with these requirements is essential for successful market entry.
3How to Approach
To establish a relationship with NCHADS, Indian exporters should initiate contact through formal channels, such as the MoH's registration platform (CamPORS), to understand the registration process and requirements. Participating in tenders and responding to calls for proposals can also be effective strategies. Understanding the regulatory framework and ensuring compliance with all requirements will facilitate a smoother entry into the Cambodian market.
Frequently Asked Questions — National Center FOR Hiv/aids
What products does National Center FOR Hiv/aids import from India?
National Center FOR Hiv/aids imports 2 pharmaceutical products across 2 categories. Top imports: Amphotericin ($300.0K), Atazanavir ($150.0K).
Who supplies pharmaceuticals to National Center FOR Hiv/aids from India?
National Center FOR Hiv/aids sources from 5 verified Indian suppliers. The primary supplier is Mylan Laboratories Limited (56.7% of imports, $990.0K).
What is National Center FOR Hiv/aids's total pharmaceutical import value?
National Center FOR Hiv/aids's total pharmaceutical import value from India is $450.0K, based on 9 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does National Center FOR Hiv/aids focus on?
National Center FOR Hiv/aids imports across 2 categories. The largest: Antifungals (66.7%), Antiviral & HIV Medications (33.3%).
Get Full National Center FOR Hiv/aids Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: National Center FOR Hiv/aids identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as National Center FOR Hiv/aids's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 9 individual customs records matching National Center FOR Hiv/aids.
- 5.Supplier Verification: National Center FOR Hiv/aids sources from 5 verified Indian suppliers across 34 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.