Merckle Gmbh
Pharmaceutical Importer · Germany · Analgesics & Antipyretics Focus · $3.0M Total Trade · DGFT Verified
Merckle Gmbh is a pharmaceutical importer based in Germany with a total trade value of $3.0M across 3 products in 3 therapeutic categories. Based on 73 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Merckle Gmbh sources from 16 verified Indian suppliers, with Centaur Pharmaceuticals Private Limited accounting for 20.2% of imports.
Merckle Gmbh — Import Portfolio & Supplier Network
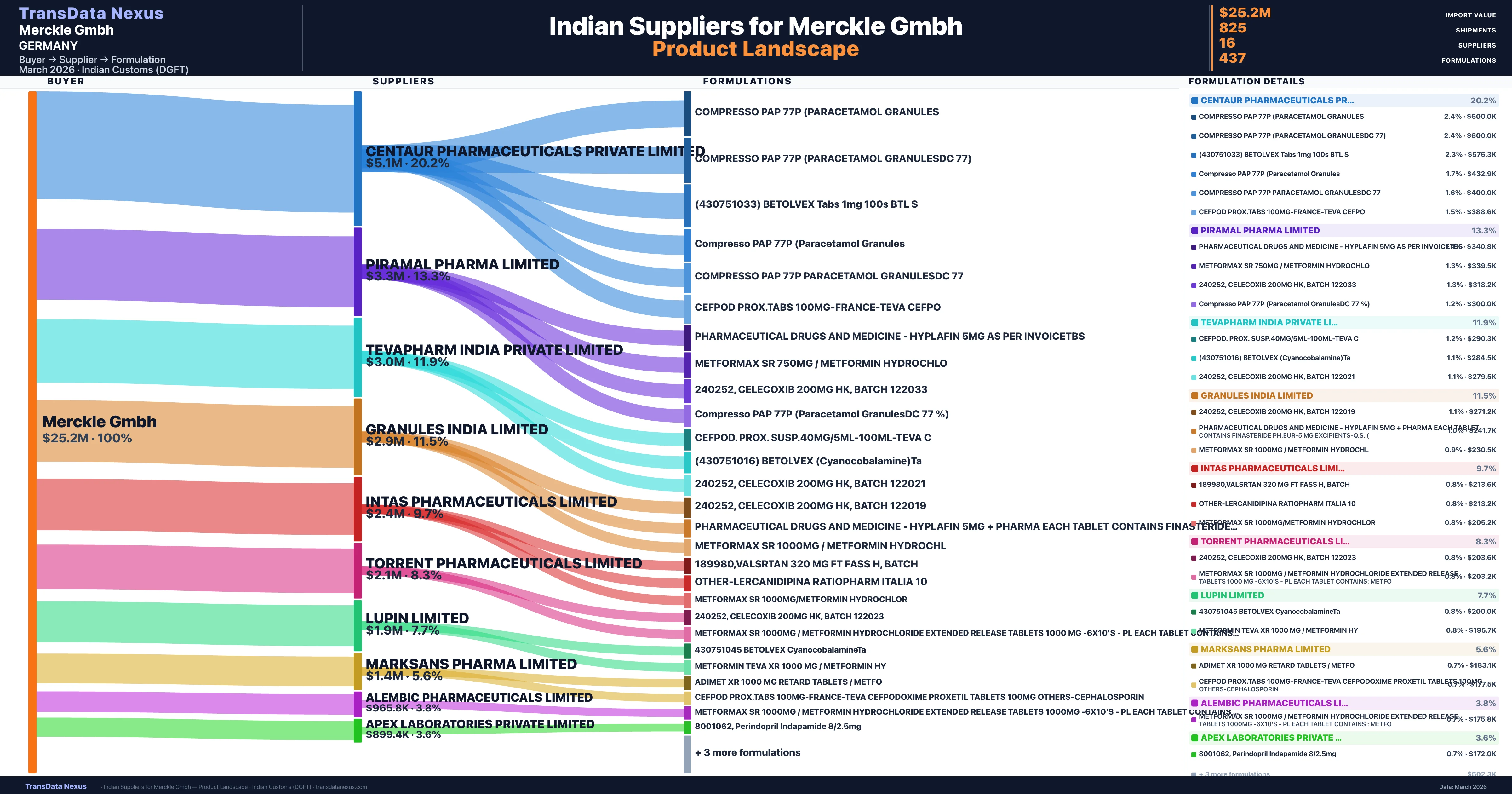
Who Are the Verified Indian Suppliers to Merckle Gmbh?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Centaur Pharmaceuticals Private Limited | $5.1M | 138 | 20.2% |
| Piramal Pharma Limited | $3.3M | 124 | 13.3% |
| Tevapharm India Private Limited | $3.0M | 93 | 11.9% |
| Granules India Limited | $2.9M | 61 | 11.5% |
| Intas Pharmaceuticals Limited | $2.4M | 138 | 9.7% |
| Torrent Pharmaceuticals Limited | $2.1M | 68 | 8.3% |
| Lupin Limited | $1.9M | 51 | 7.7% |
| Marksans Pharma Limited | $1.4M | 39 | 5.6% |
| Alembic Pharmaceuticals Limited | $965.8K | 37 | 3.8% |
| Apex Laboratories Private Limited | $899.4K | 29 | 3.6% |
| Ipca Laboratories Limited | $430.2K | 19 | 1.7% |
| Watson Pharma Private Limited | $341.6K | 15 | 1.4% |
| Mylan Laboratories Limited | $225.8K | 6 | 0.9% |
| Usv Private Limited | $57.1K | 5 | 0.2% |
| Rpg Life Sciences Limited | $50.0K | 1 | 0.2% |
| Sri Krishna Pharmaceuticals Limited | $50.0K | 1 | 0.2% |
Merckle Gmbh sources from 16 verified Indian suppliers across 437 distinct formulations. The supply base is diversified across 16 suppliers, reducing single-source dependency risk.
What Formulations Does Merckle Gmbh Import?
| Formulation | Value | Ships |
|---|---|---|
| Compresso pap 77p (paracetamol | $600.0K | 12 |
| Compresso pap 77p (paracetamol granulesdc 77) | $600.0K | 12 |
| (430751033) betolvex tabs 1MG 100s btl | $576.3K | 12 |
| Compresso pap 77p (paracetamol | $432.9K | 9 |
| Compresso pap 77p paracetamol granulesdc | $400.0K | 8 |
| Cefpod prox.tabs 100MG-france-teva | $388.6K | 8 |
| Pharmaceutical drugs and medicine - hyplafin 5MG as per | $340.8K | 9 |
| Metformax sr 750MG / metformin | $339.5K | 8 |
| 240252, celecoxib 200MG hk, batch | $318.2K | 8 |
| Compresso pap 77p (paracetamol granulesdc 77 %) | $300.0K | 6 |
| Cefpod. prox. SUSP.40MG/5ML-100ML-teva | $290.3K | 7 |
| (430751016) betolvex (cyanocobalamine | $284.5K | 6 |
| 240252, celecoxib 200MG hk, batch | $279.5K | 6 |
| 240252, celecoxib 200MG hk, batch | $271.2K | 6 |
| Pharmaceutical drugs and medicine - hyplafin 5MG + pharma Each tablet contains finasteride PH.EUR-5 MG excipients-q.s. | $241.7K | 9 |
Merckle Gmbh imports 437 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Merckle Gmbh Import?
Top Products by Import Value
Merckle Gmbh Therapeutic Categories — 3 Specializations
Merckle Gmbh imports across 3 therapeutic categories, with Analgesics & Antipyretics (57.6%), Advanced Antibiotics (25.9%), CNS & Psychiatric (16.5%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Analgesics & Antipyretics
1 products · 57.6% · $1.7M
Advanced Antibiotics
1 products · 25.9% · $785.6K
CNS & Psychiatric
1 products · 16.5% · $500.0K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Celecoxib | Analgesics & Antipyretics | $1.7M | 41 | 1.3% | 9 |
| 2 | Cefpodoxime | Advanced Antibiotics | $785.6K | 22 | 1.0% | 15 |
| 3 | Clonazepam | CNS & Psychiatric | $500.0K | 10 | 0.4% | 5 |
Merckle Gmbh imports 3 pharmaceutical products across 3 categories into Germany totaling $3.0M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Merckle Gmbh.
Request DemoMerckle Gmbh — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Merckle GmbH is a German pharmaceutical company specializing in the manufacturing and distribution of generic medicines. Established in 1881, the company has evolved into a significant player in the pharmaceutical industry. In 2010, Merckle GmbH became part of Teva Pharmaceutical Industries, a global leader in generic pharmaceuticals.
The company's headquarters are located at Graf-Arco-Straße 3, 89079 Ulm, Germany. Merckle GmbH operates production facilities in Ulm and Blaubeuren, ensuring a robust manufacturing capacity to meet both domestic and international demands.
2Distribution Network
Merckle GmbH's distribution network is extensive, encompassing multiple warehouses and logistics centers across Germany. This strategic positioning enables efficient nationwide coverage, facilitating timely delivery of pharmaceutical products to various stakeholders, including pharmacies, hospitals, and healthcare providers. While specific details about their international distribution are not publicly disclosed, their affiliation with Teva Pharmaceutical Industries suggests a global reach, leveraging Teva's established international logistics and distribution channels.
3Industry Role
Within Germany's pharmaceutical supply chain, Merckle GmbH functions primarily as a pharmaceutical manufacturer and distributor. The company focuses on producing generic medications, which are then distributed to wholesalers, pharmacies, and healthcare institutions. This role positions Merckle GmbH as a key contributor to the accessibility and affordability of essential medicines in the German market.
Supplier Relationship Intelligence — Merckle Gmbh
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Merckle GmbH's sourcing strategy exhibits a high degree of concentration, with a significant portion of their pharmaceutical imports originating from India. This concentrated sourcing approach can offer benefits such as cost efficiencies and streamlined supply chain management. However, it also introduces potential risks, including exposure to supply disruptions due to geopolitical factors, regulatory changes, or natural disasters in the sourcing region. The company's reliance on a limited number of suppliers may also impact its ability to negotiate favorable terms or adapt to market changes swiftly.
2Supply Chain Resilience
The resilience of Merckle GmbH's Indian supply chain is a critical factor in maintaining uninterrupted product availability. While the company has established relationships with multiple Indian suppliers, the overall supply chain's robustness depends on factors such as the diversification of suppliers, the stability of shipping routes, and the adherence to international regulatory standards. Ensuring compliance with Good Manufacturing Practice (GMP) and other quality standards is essential to mitigate risks associated with product quality and regulatory compliance.
3Strategic Implications
Merckle GmbH's concentrated sourcing pattern from India positions the company to leverage cost advantages and supplier expertise. However, this strategy also necessitates a proactive approach to risk management, including diversifying the supplier base and developing contingency plans to address potential supply chain disruptions. For Indian exporters, understanding Merckle GmbH's sourcing preferences and quality requirements presents an opportunity to establish partnerships, provided they can meet the necessary standards and regulatory compliance criteria.
Importing Pharmaceuticals into Germany — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Germany
1Regulatory Authority & Framework
In Germany, the Federal Institute for Drugs and Medical Devices (BfArM) serves as the primary regulatory authority overseeing the approval and distribution of pharmaceutical products. The German Medicines Act (AMG) provides the legal framework governing the manufacturing, approval, and marketing of medicinal products. For Indian generics to be marketed in Germany, they must obtain a national marketing authorization from BfArM, ensuring compliance with the AMG and EU regulations. (pspharmaservice.de)
2Import Licensing & GMP
Importing pharmaceutical products into Germany requires adherence to specific licensing and quality standards. Importers must obtain an import permit in accordance with § 72 of the AMG, which mandates compliance with personnel and material requirements similar to those for manufacturing medicinal products. Additionally, imported products must meet Good Manufacturing Practice (GMP) standards recognized by the European Union, WHO, or PIC/S to ensure product quality and safety.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality and efficacy. Stability studies are required to ensure that products maintain their intended quality throughout their shelf life. Labeling must be in German, providing clear information on dosage, administration, and potential side effects. Serialization mandates are in place to prevent counterfeit products and ensure traceability within the supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, Germany has implemented several regulatory changes affecting pharmaceutical imports. These include stricter compliance requirements for GMP, enhanced scrutiny of import permits, and updated labeling standards to improve patient safety and product traceability. Importers must stay informed about these changes to maintain compliance and ensure uninterrupted market access.
Merckle Gmbh — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Merckle GmbH's focus on importing analgesics, advanced antibiotics, and CNS & psychiatric medications aligns with the therapeutic areas of high demand in Germany. The prevalence of chronic pain, infectious diseases, and mental health conditions in the population drives the need for effective and affordable treatments in these categories. By concentrating on these areas, Merckle GmbH addresses critical healthcare needs while positioning itself competitively in the market.
2Sourcing Profile
Merckle GmbH's sourcing strategy emphasizes generic drug procurement, particularly from India, known for its robust pharmaceutical manufacturing capabilities. The company's preference for finished pharmaceutical formulations, such as tablets, capsules, syrups, and injections, indicates a focus on ready-to-market products that meet stringent quality standards. This approach allows Merckle GmbH to efficiently expand its product portfolio and meet market demands.
3Market Positioning
Based on its product mix, Merckle GmbH serves a broad segment of the German pharmaceutical market, including retail pharmacies, hospitals, and government tenders. The company's emphasis on essential therapeutic areas ensures its products are integral to various healthcare settings, contributing to the overall healthcare infrastructure and patient care in Germany.
Seller's Guide — How to Become a Supplier to Merckle Gmbh
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with Merckle GmbH, provided they can meet the company's quality standards and regulatory requirements. Gaps in Merckle GmbH's current sourcing may exist in specific formulations or packaging options, presenting avenues for collaboration. Prospective suppliers should focus on demonstrating product quality, regulatory compliance, and the ability to meet supply chain demands to establish a successful partnership.
2Requirements & Qualifications
Indian exporters aiming to supply Merckle GmbH and the German market must obtain a national marketing authorization from BfArM, ensuring compliance with the AMG and EU regulations. Products must adhere to GMP standards recognized by the European Union, WHO, or PIC/S. Additionally, labeling must be in German, and products must meet serialization requirements to comply with German regulations.
3How to Approach
To build a relationship with Merckle GmbH, Indian exporters should initiate contact through formal channels, providing detailed product information, quality certifications, and evidence of regulatory compliance. Participating in relevant tenders and industry events can enhance visibility and facilitate connections. Developing a comprehensive regulatory filing strategy, including obtaining necessary approvals and certifications, is crucial. Timelines for approval can vary; therefore, proactive communication and adherence to regulatory processes are essential for successful market entry.
Frequently Asked Questions — Merckle Gmbh
What products does Merckle Gmbh import from India?
Merckle Gmbh imports 3 pharmaceutical products across 3 categories. Top imports: Celecoxib ($1.7M), Cefpodoxime ($785.6K), Clonazepam ($500.0K).
Who supplies pharmaceuticals to Merckle Gmbh from India?
Merckle Gmbh sources from 16 verified Indian suppliers. The primary supplier is Centaur Pharmaceuticals Private Limited (20.2% of imports, $5.1M).
What is Merckle Gmbh's total pharmaceutical import value?
Merckle Gmbh's total pharmaceutical import value from India is $3.0M, based on 73 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Merckle Gmbh focus on?
Merckle Gmbh imports across 3 categories. The largest: Analgesics & Antipyretics (57.6%), Advanced Antibiotics (25.9%), CNS & Psychiatric (16.5%).
Get Full Merckle Gmbh Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Merckle Gmbh identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Merckle Gmbh's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 73 individual customs records matching Merckle Gmbh.
- 5.Supplier Verification: Merckle Gmbh sources from 16 verified Indian suppliers across 437 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.