Medecins Sans Frontieres Logistique
Pharmaceutical Importer · France · Other Focus · $809.7K Total Trade · DGFT Verified
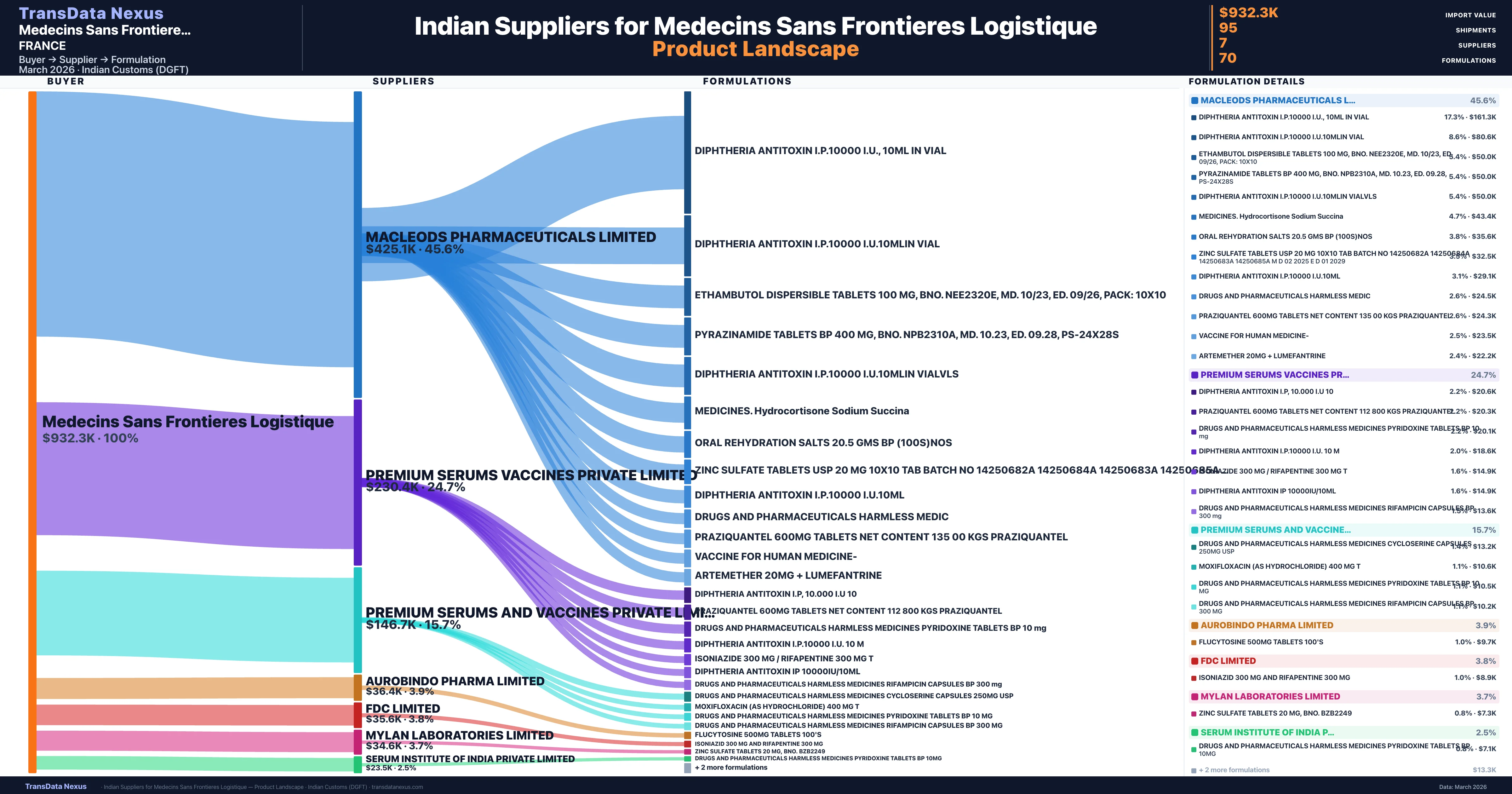
Medecins Sans Frontieres Logistique is a pharmaceutical importer based in France with a total trade value of $809.7K across 3 products in 3 therapeutic categories. Based on 23 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Medecins Sans Frontieres Logistique sources from 7 verified Indian suppliers, with Macleods Pharmaceuticals Limited accounting for 45.6% of imports.
Medecins Sans Frontieres Logistique — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Medecins Sans Frontieres Logistique?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Macleods Pharmaceuticals Limited | $425.1K | 67 | 45.6% |
| Premium Serums Vaccines Private Limited | $230.4K | 9 | 24.7% |
| Premium Serums And Vaccines Private Limited | $146.7K | 7 | 15.7% |
| Aurobindo Pharma Limited | $36.4K | 7 | 3.9% |
| Fdc Limited | $35.6K | 1 | 3.8% |
| Mylan Laboratories Limited | $34.6K | 3 | 3.7% |
| Serum Institute Of India Private Limited | $23.5K | 1 | 2.5% |
Medecins Sans Frontieres Logistique sources from 7 verified Indian suppliers across 70 distinct formulations. The supply base is diversified across 7 suppliers, reducing single-source dependency risk.
What Formulations Does Medecins Sans Frontieres Logistique Import?
| Formulation | Value | Ships |
|---|---|---|
| Diphtheria antitoxin i.p.10000 i.u., 10ML in | $161.3K | 5 |
| Diphtheria antitoxin i.p.10000 i.u.10mlin | $80.6K | 4 |
| Ethambutol dispersible tablets 100 MG, bno. nee2320e, md. 10/23, ed. 09/26, Pack | $50.0K | 1 |
| Pyrazinamide tablets BP 400 MG, bno. npb2310a, md. 10.23, ed. 09.28, ps-24x28s | $50.0K | 1 |
| Diphtheria antitoxin i.p.10000 i.u.10mlin | $50.0K | 1 |
| Medicines. hydrocortisone sodium | $43.4K | 1 |
| Oral rehydration salts 20.5 gms BP (100s | $35.6K | 1 |
| Zinc sulfate tablets USP 20 MG 10x10 TAB batch no 14250682a 14250684a 14250683a 14250685a m d 02 2025 e d 01 | $32.5K | 1 |
| Diphtheria antitoxin i.p.10000 i.u.10ML | $29.1K | 1 |
| Drugs and pharmaceuticals harmless | $24.5K | 7 |
| Praziquantel 600MG tablets net content 135 00 kgs | $24.3K | 1 |
| Vaccine for human | $23.5K | 1 |
| Artemether 20MG + | $22.2K | 1 |
| Diphtheria antitoxin i.p, 10.000 i.u | $20.6K | 1 |
| Praziquantel 600MG tablets net content 112 800 kgs | $20.3K | 1 |
Medecins Sans Frontieres Logistique imports 70 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Medecins Sans Frontieres Logistique Import?
Top Products by Import Value
Medecins Sans Frontieres Logistique Therapeutic Categories — 3 Specializations
Medecins Sans Frontieres Logistique imports across 3 therapeutic categories, with Other (51.3%), Tuberculosis Medications (43.2%), Antimalarial & Antiparasitic (5.5%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Other
1 products · 51.3% · $415.1K
Tuberculosis Medications
1 products · 43.2% · $350.0K
Antimalarial & Antiparasitic
1 products · 5.5% · $44.6K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Antitoxin | Other | $415.1K | 14 | 3.1% | 6 |
| 2 | Pyrazinamide | Tuberculosis Medications | $350.0K | 7 | 0.0% | 18 |
| 3 | Praziquantel | Antimalarial & Antiparasitic | $44.6K | 2 | 1.5% | 8 |
Medecins Sans Frontieres Logistique imports 3 pharmaceutical products across 3 categories into France totaling $809.7K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Medecins Sans Frontieres Logistique.
Request DemoMedecins Sans Frontieres Logistique — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Médecins Sans Frontières Logistique (MSF Logistique) is a non-profit organization established in 1986 under French law, operating as an association. It serves as one of the two central procurement units for the Médecins Sans Frontières (MSF) movement, ensuring the supply of medical and non-medical materials for both routine programs and emergency interventions, including responses to epidemics, natural disasters, and conflicts. MSF Logistique manages the entire supply chain, from procurement to transportation to the destination country.
Headquartered in Mérignac, Nouvelle-Aquitaine, France, MSF Logistique operates as a pharmaceutical establishment and customs warehouse, recognized by the European Union as a humanitarian purchasing center. This status enables it to efficiently manage the procurement and distribution of medical supplies for MSF's global operations. (clodura.ai)
2Distribution Network
MSF Logistique's distribution network is strategically designed to support MSF's humanitarian missions worldwide. While specific warehouse locations are not publicly disclosed, the organization maintains facilities in France, including its headquarters in Mérignac. These facilities are equipped to handle the storage and distribution of medical supplies, ensuring timely delivery to MSF's field operations. The logistics capabilities encompass procurement, storage, and transportation, facilitating the rapid deployment of medical aid to regions in need. (clodura.ai)
3Industry Role
In France's pharmaceutical supply chain, MSF Logistique functions as a central procurement and logistics entity for the MSF movement. It is not a wholesaler or distributor in the traditional commercial sense but operates as a specialized logistics intermediary. Its primary role is to source, store, and distribute medical supplies to support MSF's humanitarian missions globally. This includes managing the supply chain for both routine programs and emergency interventions, ensuring that medical aid reaches those in need promptly and efficiently. (clodura.ai)
Supplier Relationship Intelligence — Medecins Sans Frontieres Logistique
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
MSF Logistique's sourcing strategy from India reveals a high concentration on a limited number of suppliers. The organization has engaged with seven unique Indian suppliers, with the top three accounting for 88.5% of the total import value. This concentration indicates a strategic choice to establish strong, reliable partnerships with key suppliers, ensuring consistent quality and supply. The shipment data, with 23 total shipments, suggests a stable and ongoing relationship with these suppliers, facilitating efficient procurement processes. However, such concentration also presents a potential risk; any disruption with these key suppliers could impact MSF Logistique's ability to deliver medical supplies effectively.
2Supply Chain Resilience
MSF Logistique's supply chain resilience is supported by its partnerships with multiple Indian suppliers, providing a degree of diversification. The organization imports 70 unique pharmaceutical formulations, indicating a broad product range that can mitigate risks associated with dependency on a single supplier or product. While the top three suppliers dominate the import value, the presence of additional suppliers offers alternative sources in case of disruptions. The shipment data, with 23 total shipments, reflects a consistent and reliable supply chain. However, the resilience of this supply chain also depends on the stability and reliability of the Indian pharmaceutical industry, including adherence to international quality standards and regulatory compliance.
3Strategic Implications
MSF Logistique's sourcing pattern from India positions it to leverage cost-effective procurement while maintaining a diverse product portfolio. The concentration on a few key suppliers allows for streamlined operations and potentially better terms. For Indian exporters, this presents an opportunity to become alternative suppliers, especially if they can offer competitive pricing, high-quality products, and reliable delivery schedules. Establishing relationships with MSF Logistique could lead to long-term partnerships, contributing to the organization's mission of providing timely medical aid.
Importing Pharmaceuticals into France — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for France
1Regulatory Authority & Framework
In France, the regulatory authority overseeing pharmaceutical imports is the French National Agency for Medicines and Health Products Safety (ANSM). The French Public Health Code governs the importation of medicinal products, stipulating that any medicinal product without a marketing authorization requires an import authorization for each import operation. This ensures that all imported pharmaceuticals meet the necessary safety and efficacy standards before reaching the French market.
2Import Licensing & GMP
Import licensing in France requires that each import operation of finished pharmaceutical products be authorized by the ANSM. This process ensures that imported medicines comply with French and European Union regulations. Good Manufacturing Practice (GMP) certificates from recognized authorities, such as the European Medicines Agency (EMA) or the World Health Organization (WHO), are essential for the importation of pharmaceutical products. These certifications confirm that the manufacturing processes meet international quality standards. Additionally, wholesale distribution authorization is required for entities involved in the distribution of medicinal products within France, ensuring that all intermediaries in the supply chain adhere to regulatory standards.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality, safety, and efficacy. Stability requirements ensure that medicines maintain their intended properties throughout their shelf life. Labeling must be in French, providing clear information on usage, dosage, and safety precautions. Serialization mandates are in place to track and trace pharmaceutical products, enhancing transparency and preventing counterfeit medicines from entering the supply chain. These measures are crucial for maintaining the integrity of the pharmaceutical supply chain and protecting public health.
4Recent Regulatory Changes
Between 2024 and 2026, France implemented several regulatory changes affecting pharmaceutical imports. These included stricter requirements for GMP certifications, enhanced batch testing protocols, and updated labeling standards to improve transparency and patient safety. Additionally, serialization mandates were expanded to include more detailed tracking of pharmaceutical products throughout the supply chain. These changes aim to strengthen the regulatory framework governing pharmaceutical imports, ensuring that all medicines entering the French market meet the highest standards of quality and safety.
Medecins Sans Frontieres Logistique — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
MSF Logistique's focus on importing antitoxins, pyrazinamide, and praziquantel aligns with its mission to provide essential medical treatments for conditions prevalent in regions affected by conflict, epidemics, and natural disasters. Antitoxins are critical for treating various toxin-induced illnesses, pyrazinamide is a key component in tuberculosis treatment regimens, and praziquantel is essential for treating parasitic infections. The market demand for these products is driven by the need to address public health emergencies and provide ongoing medical care in underserved areas.
2Sourcing Profile
MSF Logistique's sourcing strategy emphasizes the procurement of generic drugs, focusing on formulations that are essential for treating infectious diseases and other health conditions prevalent in crisis settings. The organization imports a diverse range of formulations, indicating a preference for products that can be adapted to the specific needs of different regions. India's role as a major supplier of affordable and high-quality generic pharmaceuticals makes it a key partner in MSF Logistique's procurement strategy.
3Market Positioning
MSF Logistique serves the humanitarian medical aid segment within France's pharmaceutical market. Its imports are directed towards supporting MSF's global missions, including providing medical care in conflict zones, disaster-stricken areas, and regions facing epidemics. The organization's focus is on ensuring the availability of essential medicines for MSF's field operations, rather than serving the traditional retail pharmacy or hospital markets.
Seller's Guide — How to Become a Supplier to Medecins Sans Frontieres Logistique
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with MSF Logistique, especially if they can offer competitive pricing, high-quality products, and reliable delivery schedules. The existing sourcing pattern indicates a preference for established suppliers, but MSF Logistique's diverse product portfolio suggests potential gaps that new suppliers could fill. Building relationships with MSF Logistique could lead to long-term partnerships, contributing to the organization's mission of providing timely medical aid.
2Requirements & Qualifications
Indian exporters seeking to supply MSF Logistique must ensure that their products comply with French and European Union regulations, including obtaining necessary import licenses and GMP certifications from recognized authorities. Products must meet quality standards, undergo batch testing, and adhere to labeling requirements in French. Additionally, exporters should be prepared to meet serialization mandates for tracking and tracing pharmaceutical products throughout the supply chain.
3How to Approach
Indian exporters interested in supplying MSF Logistique should begin by understanding the organization's procurement processes and requirements. Engaging in direct communication to express interest, providing detailed product information, and demonstrating compliance with regulatory standards are essential steps. Participating in relevant tenders and staying informed about MSF Logistique's procurement needs can enhance the chances of establishing a partnership. Understanding the organization's mission and aligning product offerings with its humanitarian objectives will be crucial in building a successful relationship.
Frequently Asked Questions — Medecins Sans Frontieres Logistique
What products does Medecins Sans Frontieres Logistique import from India?
Medecins Sans Frontieres Logistique imports 3 pharmaceutical products across 3 categories. Top imports: Antitoxin ($415.1K), Pyrazinamide ($350.0K), Praziquantel ($44.6K).
Who supplies pharmaceuticals to Medecins Sans Frontieres Logistique from India?
Medecins Sans Frontieres Logistique sources from 7 verified Indian suppliers. The primary supplier is Macleods Pharmaceuticals Limited (45.6% of imports, $425.1K).
What is Medecins Sans Frontieres Logistique's total pharmaceutical import value?
Medecins Sans Frontieres Logistique's total pharmaceutical import value from India is $809.7K, based on 23 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Medecins Sans Frontieres Logistique focus on?
Medecins Sans Frontieres Logistique imports across 3 categories. The largest: Other (51.3%), Tuberculosis Medications (43.2%), Antimalarial & Antiparasitic (5.5%).
Get Full Medecins Sans Frontieres Logistique Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Medecins Sans Frontieres Logistique identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Medecins Sans Frontieres Logistique's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 23 individual customs records matching Medecins Sans Frontieres Logistique.
- 5.Supplier Verification: Medecins Sans Frontieres Logistique sources from 7 verified Indian suppliers across 70 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.