M/s. SUN Farmaceutica DO Brazil Ltd
Pharmaceutical Importer · Brazil · Advanced Oncology Focus · $1.3M Total Trade · DGFT Verified
M/s. SUN Farmaceutica DO Brazil Ltd is a pharmaceutical importer based in Brazil with a total trade value of $1.3M across 4 products in 1 therapeutic categories. Based on 29 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. M/s. SUN Farmaceutica DO Brazil Ltd sources from 1 verified Indian supplier, with Sun Pharmaceutical Industries Limited accounting for 100.0% of imports.
M/s. SUN Farmaceutica DO Brazil Ltd — Import Portfolio & Supplier Network
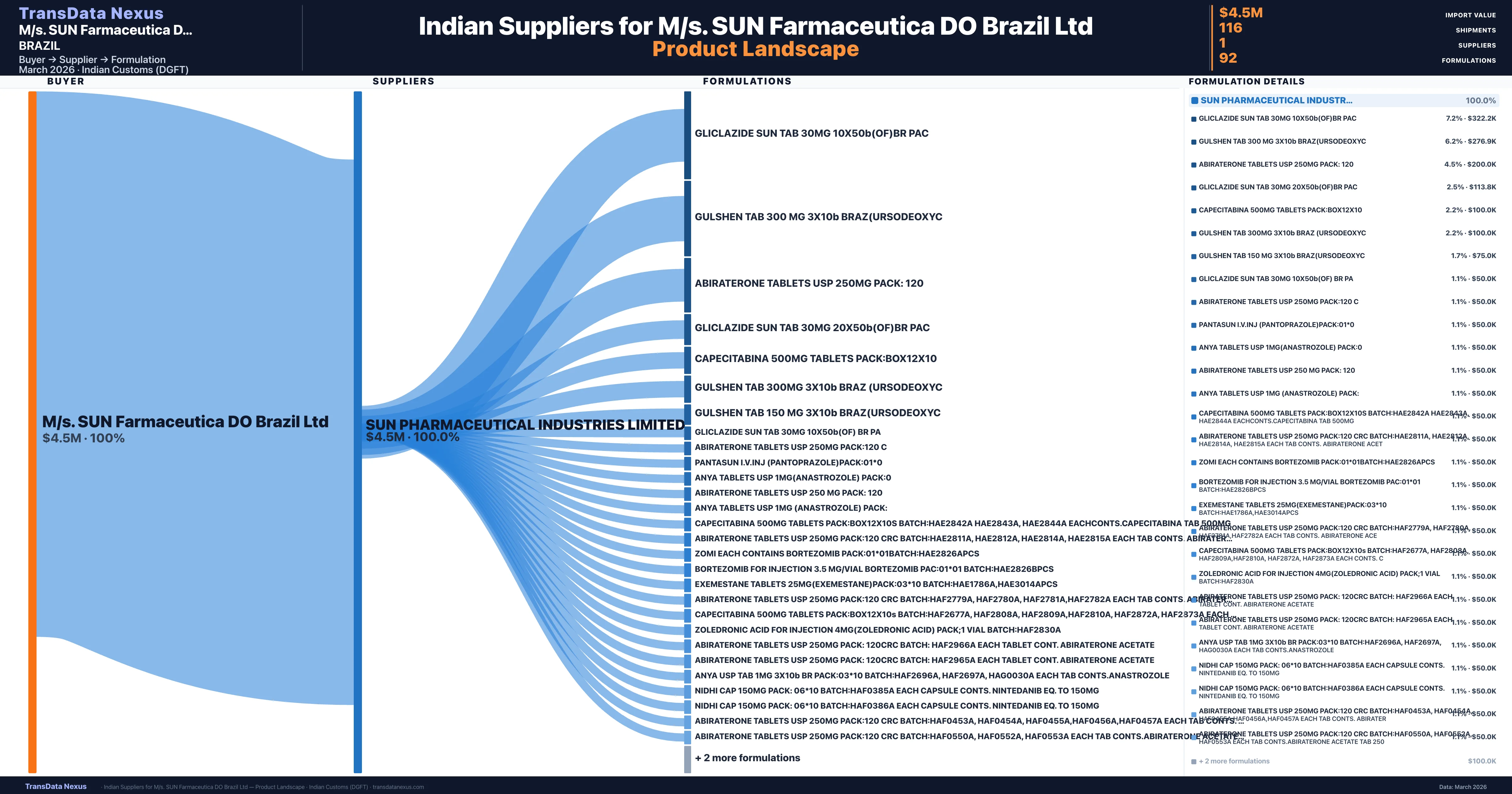
Who Are the Verified Indian Suppliers to M/s. SUN Farmaceutica DO Brazil Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Sun Pharmaceutical Industries Limited | $4.5M | 116 | 100.0% |
M/s. SUN Farmaceutica DO Brazil Ltd sources from 1 verified Indian supplier across 92 distinct formulations. The sourcing is highly concentrated — Sun Pharmaceutical Industries Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does M/s. SUN Farmaceutica DO Brazil Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Gliclazide sun TAB 30MG 10x50b(of)br | $322.2K | 7 |
| Gulshen TAB 300 MG 3x10b | $276.9K | 6 |
| Abiraterone tablets USP 250MG Pack | $200.0K | 4 |
| Gliclazide sun TAB 30MG 20x50b(of)br | $113.8K | 3 |
| Capecitabina 500MG tablets | $100.0K | 2 |
| Gulshen TAB 300MG 3x10b braz | $100.0K | 2 |
| Gulshen TAB 150 MG 3x10b | $75.0K | 2 |
| Gliclazide sun TAB 30MG 10x50b(of) br | $50.0K | 1 |
| Abiraterone tablets USP 250MG Pack:120 | $50.0K | 1 |
| Pantasun i.v.INJ (pantoprazole | $50.0K | 1 |
| Anya tablets USP 1MG(anastrozole) | $50.0K | 1 |
| Abiraterone tablets USP 250 MG Pack | $50.0K | 1 |
| Anya tablets USP 1MG (anastrozole) | $50.0K | 1 |
| Capecitabina 500MG tablets Pack:box12x10s batch:hae2842a hae2843a, hae2844a eachconts.capecitabina TAB 500MG | $50.0K | 1 |
| Abiraterone tablets USP 250MG Pack:120 crc batch:hae2811a, hae2812a, hae2814a, hae2815a Each TAB conts. abiraterone | $50.0K | 1 |
M/s. SUN Farmaceutica DO Brazil Ltd imports 92 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does M/s. SUN Farmaceutica DO Brazil Ltd Import?
Top Products by Import Value
M/s. SUN Farmaceutica DO Brazil Ltd Therapeutic Categories — 1 Specializations
M/s. SUN Farmaceutica DO Brazil Ltd imports across 1 therapeutic categories, with Advanced Oncology (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Oncology
4 products · 100.0% · $1.3M
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Anastrozole | Advanced Oncology | $587.3K | 15 | 0.5% | 7 |
| 2 | Capecitabine | Advanced Oncology | $250.0K | 5 | 0.3% | 13 |
| 3 | Temozolomide | Advanced Oncology | $250.0K | 5 | 1.1% | 5 |
| 4 | Bortezomib | Advanced Oncology | $200.0K | 4 | 0.9% | 11 |
M/s. SUN Farmaceutica DO Brazil Ltd imports 4 pharmaceutical products across 1 categories into Brazil totaling $1.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for M/s. SUN Farmaceutica DO Brazil Ltd.
Request DemoM/s. SUN Farmaceutica DO Brazil Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
M/s. SUN Farmaceutica DO Brazil Ltd is a pharmaceutical importer and wholesaler based in Brazil, specializing in the distribution of finished pharmaceutical formulations. Established on August 13, 2021, the company operates under the legal entity CNPJ 05.035.244/0003-95, with its headquarters located at Rua Francisco de S e Melo, 252, Glp 1 Armazém 11, Cordovil, Rio de Janeiro, RJ, 21010-410. (cnpj.biz) The primary activity of SUN Farmaceutica DO Brazil Ltd is the wholesale trade of human medicines and drugs, as classified under the CNAE code 46.44-3-01. (econodata.com.br)
The company is a subsidiary of SUN Pharmaceutical Industries Limited, a leading global pharmaceutical manufacturer headquartered in India. This affiliation underscores SUN Farmaceutica DO Brazil Ltd's strategic role in facilitating the distribution of SUN Pharma's products within the Brazilian market. By leveraging its parent company's extensive product portfolio and manufacturing capabilities, SUN Farmaceutica DO Brazil Ltd aims to enhance the availability and accessibility of quality pharmaceutical products across Brazil.
2Distribution Network
SUN Farmaceutica DO Brazil Ltd maintains a centralized distribution hub at its Rio de Janeiro facility, which serves as the primary point for receiving, storing, and dispatching pharmaceutical products. The company's logistics operations are designed to ensure efficient nationwide distribution, leveraging Brazil's extensive transportation infrastructure, including road, rail, and port systems. While specific details regarding additional warehouse locations or logistics capabilities are not publicly disclosed, the company's strategic positioning in Rio de Janeiro facilitates access to major ports and transportation routes, enabling effective distribution throughout Brazil.
3Industry Role
In Brazil's pharmaceutical supply chain, SUN Farmaceutica DO Brazil Ltd functions as a primary wholesaler and importer. By sourcing finished pharmaceutical formulations from its parent company, SUN Pharmaceutical Industries Limited, the company plays a crucial role in bridging the manufacturing capabilities of its parent with the distribution needs of the Brazilian market. This position allows SUN Farmaceutica DO Brazil Ltd to ensure a steady supply of quality medications to various stakeholders, including hospitals, pharmacies, and healthcare providers across the country.
Supplier Relationship Intelligence — M/s. SUN Farmaceutica DO Brazil Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
SUN Farmaceutica DO Brazil Ltd exhibits a high degree of supplier concentration, sourcing all its pharmaceutical imports exclusively from SUN Pharmaceutical Industries Limited. This single-source dependency reflects a strategic choice to maintain consistency in product quality and supply chain reliability. The company's import data indicates a total import value of $1.3 million USD from India, encompassing 29 shipments and 92 unique formulations across four products in the Advanced Oncology therapeutic category. The top five products imported include Anastrozole ($587K), Capecitabine ($250K), Temozolomide ($250K), and Bortezomib ($200K). This focused sourcing strategy suggests a stable and well-established relationship between SUN Farmaceutica DO Brazil Ltd and SUN Pharmaceutical Industries Limited, ensuring a consistent supply of specialized oncology medications.
2Supply Chain Resilience
The resilience of SUN Farmaceutica DO Brazil Ltd's supply chain is closely tied to its exclusive sourcing arrangement with SUN Pharmaceutical Industries Limited. This singular supplier relationship may present risks related to supply chain disruptions, such as manufacturing delays or logistical challenges. However, the strategic alignment between the two entities likely includes contingency planning and robust communication channels to mitigate potential disruptions. The company's focus on a limited number of formulations within the Advanced Oncology category indicates a specialized supply chain that may benefit from streamlined operations and focused quality control measures. While the lack of publicly available information on backup suppliers or alternative sourcing strategies limits a comprehensive assessment, the existing supplier relationship appears to be a cornerstone of SUN Farmaceutica DO Brazil Ltd's supply chain resilience.
3Strategic Implications
The concentrated sourcing pattern of SUN Farmaceutica DO Brazil Ltd, relying solely on SUN Pharmaceutical Industries Limited, positions the company to leverage the strengths and capabilities of its parent company. This strategic alignment facilitates access to a comprehensive portfolio of oncology medications, potentially enhancing the company's competitive edge in the Brazilian market. For Indian exporters seeking to become alternative suppliers, the existing supplier concentration presents an opportunity to introduce complementary products or formulations that align with SUN Farmaceutica DO Brazil Ltd's focus on oncology. Establishing a diversified supplier base could provide the company with greater flexibility and risk mitigation strategies, while also offering Indian exporters a foothold in the Brazilian pharmaceutical market.
Importing Pharmaceuticals into Brazil — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Brazil
1Regulatory Authority & Framework
In Brazil, the National Health Surveillance Agency (ANVISA) is the primary regulatory body overseeing the pharmaceutical sector. ANVISA is responsible for ensuring the safety, efficacy, and quality of pharmaceutical products through rigorous evaluation and monitoring processes. Key legislation governing pharmaceutical imports includes the Brazilian Health Regulatory Agency Act and the National Health Surveillance Agency Act, which outline the requirements for product registration, importation, and distribution. The marketing authorization pathway for Indian generics involves obtaining ANVISA approval, which requires comprehensive documentation, including clinical trial data, manufacturing information, and evidence of bioequivalence. This regulatory framework ensures that imported pharmaceutical products meet the necessary standards for public health and safety.
2Import Licensing & GMP
Import licensing requirements in Brazil mandate that pharmaceutical products, including finished formulations, be registered with ANVISA prior to importation. This process involves submitting detailed dossiers that demonstrate compliance with Brazilian regulations and standards. Good Manufacturing Practice (GMP) certification is a critical component of the import licensing process. ANVISA recognizes GMP certifications from reputable international bodies, such as the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Pharmaceutical companies seeking to export to Brazil must ensure that their manufacturing facilities hold valid GMP certifications from these recognized organizations to facilitate the importation process. Additionally, wholesale distribution authorization is required for entities involved in the distribution of pharmaceutical products within Brazil, ensuring that all stakeholders in the supply chain adhere to regulatory standards.
3Quality & Labeling
Imported pharmaceutical products in Brazil are subject to batch testing and stability requirements to ensure their safety and efficacy. ANVISA mandates that imported drugs undergo quality control testing upon arrival, which may include assessments of purity, potency, and stability. Labeling requirements stipulate that product labels be in Portuguese and include essential information such as dosage instructions, active ingredients, expiration dates, and storage conditions. Serialization mandates are also in place to facilitate traceability and prevent counterfeit products from entering the market. These regulations are designed to protect public health by ensuring that pharmaceutical products meet the necessary standards for quality and safety.
4Recent Regulatory Changes
Between 2024 and 2026, Brazil implemented several policy changes affecting pharmaceutical imports, particularly concerning the registration and approval processes for foreign-manufactured drugs. ANVISA introduced streamlined procedures for the registration of generic medications, aiming to expedite market entry and enhance competition. Additionally, there was an increased emphasis on the recognition of international GMP certifications, allowing for a more efficient importation process for foreign manufacturers. These regulatory changes are intended to improve the availability of quality pharmaceutical products in Brazil and encourage the participation of international suppliers in the Brazilian market.
M/s. SUN Farmaceutica DO Brazil Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
SUN Farmaceutica DO Brazil Ltd's product strategy focuses exclusively on the Advanced Oncology therapeutic category, importing finished pharmaceutical formulations such as Anastrozole, Capecitabine, Temozolomide, and Bortezomib. This strategic focus aligns with the growing demand for oncology treatments in Brazil, driven by an increasing incidence of cancer and a need for effective therapies. By concentrating on this specialized area, the company aims to leverage its parent company's expertise and product portfolio to meet the specific needs of the Brazilian oncology market. This targeted approach allows SUN Farmaceutica DO Brazil Ltd to establish a strong presence in a niche segment, potentially leading to enhanced market share and recognition among healthcare providers and patients.
2Sourcing Profile
SUN Farmaceutica DO Brazil Ltd's sourcing strategy is centered on importing generic drugs from its parent company, SUN Pharmaceutical Industries Limited. The company imports finished pharmaceutical formulations, indicating a preference for ready-to-market products that require minimal additional processing. This approach allows SUN Farmaceutica DO Brazil Ltd to efficiently distribute high-quality medications without the complexities associated with raw material sourcing or manufacturing. India's established pharmaceutical manufacturing capabilities and recognized GMP certifications make it a suitable partner for SUN Farmaceutica DO Brazil Ltd's sourcing needs, ensuring compliance with Brazilian regulatory standards and facilitating a smooth importation process.
3Market Positioning
Based on its exclusive focus on the Advanced Oncology therapeutic category, SUN Farmaceutica DO Brazil Ltd serves a specialized segment of the Brazilian pharmaceutical market. The company's product mix positions it to cater to the needs of hospitals, oncology clinics, and specialized pharmacies that require high-quality oncology medications. By concentrating on this niche area, SUN Farmaceutica DO Brazil Ltd differentiates itself from broader pharmaceutical distributors, establishing expertise and a strong reputation within the oncology sector. This strategic positioning enables the company to effectively meet the specific demands of healthcare providers and patients seeking advanced cancer treatments.
Seller's Guide — How to Become a Supplier to M/s. SUN Farmaceutica DO Brazil Ltd
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
For new Indian suppliers, there is a realistic opportunity to engage with SUN Farmaceutica DO Brazil Ltd by offering complementary oncology products that align with the company's existing portfolio. Identifying
Frequently Asked Questions — M/s. SUN Farmaceutica DO Brazil Ltd
What products does M/s. SUN Farmaceutica DO Brazil Ltd import from India?
M/s. SUN Farmaceutica DO Brazil Ltd imports 4 pharmaceutical products across 1 categories. Top imports: Anastrozole ($587.3K), Capecitabine ($250.0K), Temozolomide ($250.0K), Bortezomib ($200.0K).
Who supplies pharmaceuticals to M/s. SUN Farmaceutica DO Brazil Ltd from India?
M/s. SUN Farmaceutica DO Brazil Ltd sources from 1 verified Indian suppliers. The primary supplier is Sun Pharmaceutical Industries Limited (100.0% of imports, $4.5M).
What is M/s. SUN Farmaceutica DO Brazil Ltd's total pharmaceutical import value?
M/s. SUN Farmaceutica DO Brazil Ltd's total pharmaceutical import value from India is $1.3M, based on 29 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does M/s. SUN Farmaceutica DO Brazil Ltd focus on?
M/s. SUN Farmaceutica DO Brazil Ltd imports across 1 categories. The largest: Advanced Oncology (100.0%).
Get Full M/s. SUN Farmaceutica DO Brazil Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: M/s. SUN Farmaceutica DO Brazil Ltd identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as M/s. SUN Farmaceutica DO Brazil Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 29 individual customs records matching M/s. SUN Farmaceutica DO Brazil Ltd.
- 5.Supplier Verification: M/s. SUN Farmaceutica DO Brazil Ltd sources from 1 verified Indian suppliers across 92 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.