Lifestar Pharma Llc.
Pharmaceutical Importer · United States · Advanced Antibiotics Focus · $43.1M Total Trade · DGFT Verified
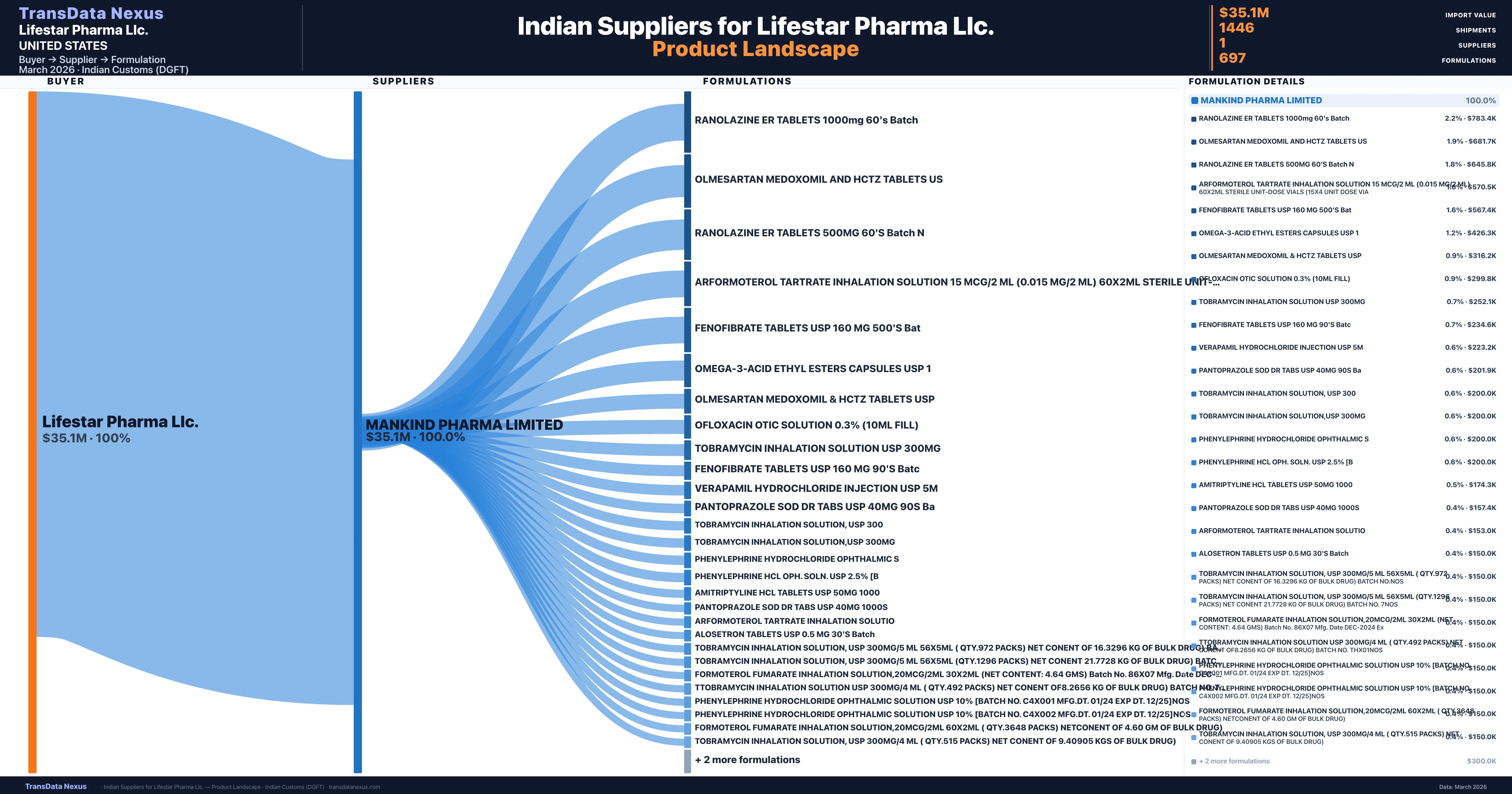
Lifestar Pharma Llc. is a pharmaceutical importer based in United States with a total trade value of $43.1M across 12 products in 9 therapeutic categories. Based on 1,217 verified import shipments from Indian Customs (DGFT) records, Lifestar Pharma Llc. is the #1 buyer in 5 products including Nitrofurantoin, Formoterol, Phenylephrine. Lifestar Pharma Llc. sources from 1 verified Indian supplier, with Mankind Pharma Limited accounting for 100.0% of imports.
Lifestar Pharma Llc. — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Lifestar Pharma Llc.?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Mankind Pharma Limited | $35.1M | 1,446 | 100.0% |
Lifestar Pharma Llc. sources from 1 verified Indian supplier across 697 distinct formulations. The sourcing is highly concentrated — Mankind Pharma Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Lifestar Pharma Llc. Import?
| Formulation | Value | Ships |
|---|---|---|
| Ranolazine er tablets 1000MG 60's | $783.4K | 23 |
| Olmesartan medoxomil and hctz tablets | $681.7K | 38 |
| Ranolazine er tablets 500MG 60's batch | $645.8K | 23 |
| Arformoterol tartrate inhalation solution 15 MCG/2 ML (0.015 MG/2 ML) 60x2ml sterile unit-dose vials (15x4 unit dose | $570.5K | 12 |
| Fenofibrate tablets USP 160 MG 500's | $567.4K | 40 |
| Omega-3-acid ethyl esters capsules USP | $426.3K | 10 |
| Olmesartan medoxomil & hctz tablets | $316.2K | 17 |
| Ofloxacin otic solution 0.3% (10ML fill) | $299.8K | 18 |
| Tobramycin inhalation solution USP 300MG | $252.1K | 6 |
| Fenofibrate tablets USP 160 MG 90's | $234.6K | 17 |
| Verapamil hydrochloride injection USP | $223.2K | 6 |
| Pantoprazole sod dr tabs USP 40MG 90s | $201.9K | 6 |
| Tobramycin inhalation solution, USP | $200.0K | 4 |
| Tobramycin inhalation solution,USP 300MG | $200.0K | 4 |
| Phenylephrine hydrochloride ophthalmic | $200.0K | 4 |
Lifestar Pharma Llc. imports 697 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Lifestar Pharma Llc. Import?
Lifestar Pharma Llc. Therapeutic Categories — 9 Specializations
Lifestar Pharma Llc. imports across 9 therapeutic categories, with Advanced Antibiotics (35.3%), Respiratory (14.9%), Respiratory & OTC (14.8%) representing the largest segments. The portfolio is concentrated — top 5 products = 75% of total imports.
Advanced Antibiotics
3 products · 35.3% · $15.2M
Respiratory
1 products · 14.9% · $6.4M
Respiratory & OTC
1 products · 14.8% · $6.4M
Lipid & Metabolism
1 products · 10.8% · $4.7M
Ayurvedic & Herbal Products
1 products · 6.4% · $2.7M
Antibiotics
1 products · 6.0% · $2.6M
Nutritional Supplements
1 products · 5.8% · $2.5M
Advanced Oncology
1 products · 3.1% · $1.3M
Cardiovascular
2 products · 2.8% · $1.2M
Import Portfolio — Top 12 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Nitrofurantoin | Advanced Antibiotics | $9.1M | 190 | 25.8% | 1 |
| 2 | Formoterol | Respiratory | $6.4M | 129 | 8.2% | 1 |
| 3 | Phenylephrine | Respiratory & OTC | $6.4M | 128 | 5.7% | 1 |
| 4 | Ofloxacin | Advanced Antibiotics | $5.8M | 148 | 2.0% | 6 |
| 5 | Fenofibrate | Lipid & Metabolism | $4.7M | 293 | 6.3% | 3 |
| 6 | Vati | Ayurvedic & Herbal Products | $2.7M | 108 | 10.4% | 2 |
| 7 | Tobramycin | Antibiotics | $2.6M | 52 | 5.1% | 1 |
| 8 | Omega | Nutritional Supplements | $2.5M | 51 | 13.2% | 3 |
| 9 | Capecitabine | Advanced Oncology | $1.3M | 50 | 0.6% | 1 |
| 10 | Verapamil | Cardiovascular | $770.7K | 18 | 0.7% | 11 |
| 11 | Nitroglycerin | Cardiovascular | $435.7K | 42 | 4.5% | 6 |
| 12 | Moxifloxacin | Advanced Antibiotics | $314.5K | 8 | 1.0% | 16 |
Lifestar Pharma Llc. imports 12 pharmaceutical products across 9 categories into United States totaling $43.1M. The company is the #1 buyer for 5 products: Nitrofurantoin, Formoterol, Phenylephrine, Tobramycin, Capecitabine.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Lifestar Pharma Llc..
Request DemoLifestar Pharma Llc. — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Lifestar Pharma LLC is a U.S.-based pharmaceutical company specializing in the development, manufacturing, and distribution of affordable, high-quality specialty generic medicines. Established in 2016, the company operates as a subsidiary of Mankind Pharma Limited, one of India's leading pharmaceutical firms. Headquartered in Mahwah, New Jersey, Lifestar Pharma focuses on delivering complex generics across various dosage forms, including ophthalmics, injectables, inhalation solutions, and oral solids.
As a key player in the U.S. pharmaceutical distribution network, Lifestar Pharma serves as a primary wholesaler, sourcing a diverse range of finished pharmaceutical formulations from international suppliers. The company's strategic partnership with Mankind Pharma Limited enables it to leverage global expertise and a robust supply chain, ensuring the availability of essential medicines to meet the needs of the U.S. healthcare sector.
2Distribution Network
Lifestar Pharma LLC's distribution network is centered around its headquarters in Mahwah, New Jersey. While specific details about additional warehouse locations and logistics capabilities are not publicly disclosed, the company's strategic position in New Jersey provides advantageous access to major U.S. markets. This location facilitates efficient distribution and timely delivery of pharmaceutical products across the United States. The company's partnership with Mankind Pharma Limited further enhances its logistics capabilities, ensuring a steady supply of high-quality generics to meet market demand.
3Industry Role
In the U.S. pharmaceutical supply chain, Lifestar Pharma LLC functions primarily as a wholesaler, sourcing finished pharmaceutical formulations from international suppliers, particularly from India. The company's focus on high-quality, affordable generics positions it as a significant contributor to the U.S. healthcare sector, addressing the growing demand for cost-effective medications. By leveraging its relationship with Mankind Pharma Limited, Lifestar Pharma ensures a diverse and reliable product portfolio, enhancing its role in the distribution of specialty generics within the United States.
Supplier Relationship Intelligence — Lifestar Pharma Llc.
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Lifestar Pharma LLC's sourcing strategy exhibits a high degree of concentration, with a total import value of $43.1 million USD from India, accounting for 100% of its pharmaceutical imports. This exclusive reliance on a single supplier, Mankind Pharma Limited, suggests a strategic choice to maintain a consistent and reliable supply chain. The substantial volume of imports, totaling 1,217 shipments, indicates a stable and long-term relationship between Lifestar Pharma and Mankind Pharma. However, this single-source dependency also presents potential risks, such as exposure to supply chain disruptions or regulatory changes affecting the supplier. The concentration of imports in a few therapeutic categories, notably Advanced Antibiotics (35.3%), Respiratory (14.9%), and Respiratory & OTC (14.8%), reflects Lifestar Pharma's strategic focus on high-demand areas within the U.S. market. The top five products—Nitrofurantoin, Formoterol, Phenylephrine, Ofloxacin, and Fenofibrate—comprise 75.2% of the total import value, highlighting a targeted approach to product selection.
2Supply Chain Resilience
Lifestar Pharma LLC's supply chain resilience is closely tied to its exclusive partnership with Mankind Pharma Limited. While this relationship ensures a consistent supply of high-quality generics, it also means that the company's supply chain is vulnerable to any disruptions affecting Mankind Pharma. The lack of publicly available information regarding backup suppliers or alternative sourcing strategies suggests a potential area for improvement in diversifying the supply chain to mitigate risks. The focus on a limited number of therapeutic categories and products may also impact the company's ability to adapt to changing market demands or regulatory environments. Ensuring compliance with U.S. Food and Drug Administration (FDA) regulations is crucial for maintaining the integrity and reliability of the supply chain.
3Strategic Implications
Lifestar Pharma LLC's concentrated sourcing strategy positions it as a significant player in the U.S. market for specific therapeutic categories, particularly in advanced antibiotics and respiratory treatments. This focus allows the company to leverage economies of scale and establish strong market presence in these segments. For Indian exporters, the exclusive relationship with Mankind Pharma Limited presents both opportunities and challenges. While the partnership offers a pathway to the U.S. market, it also means that alternative suppliers may face difficulties in penetrating this distribution channel. To become alternative suppliers to Lifestar Pharma, Indian exporters would need to demonstrate compliance with FDA regulations, establish robust quality assurance processes, and potentially seek partnerships or distribution agreements that align with Lifestar Pharma's strategic objectives.
Importing Pharmaceuticals into United States — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United States
1Regulatory Authority & Framework
In the United States, the Food and Drug Administration (FDA) serves as the primary regulatory authority overseeing the importation and distribution of pharmaceutical products. The FDA enforces the Federal Food, Drug, and Cosmetic Act (FD&C Act), which sets forth requirements for the safety, efficacy, and quality of drugs imported into the U.S. Pharmaceutical imports must comply with FDA standards, including proper labeling, manufacturing practices, and approval processes. The FDA reviews shipments of imported drugs to determine their admissibility, ensuring they meet the necessary criteria for entry into the U.S. market.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products entering the United States are governed by the FDA. Importers must file an entry notice and an entry bond with U.S. Customs, notifying the FDA of the product's arrival. The FDA then assesses the product's compliance with applicable regulations. Good Manufacturing Practice (GMP) certificates, such as those from the European Union (EU GMP), World Health Organization (WHO GMP), or Pharmaceutical Inspection Co-operation Scheme (PIC/S), are recognized by the FDA. These certifications demonstrate that the manufacturing facility adheres to internationally accepted quality standards. Importers must ensure that their suppliers possess valid GMP certifications to facilitate the importation process.
3Quality & Labeling
All pharmaceutical products offered for importation into the United States are subject to FDA labeling requirements. The FDA reviews drug labeling at the time the product is offered for import to verify compliance with regulations. Specific labeling requirements depend on the type of drug product, including over-the-counter drugs, prescription drugs, and investigational drugs. Additionally, imported drugs must meet FDA standards for quality, safety, and effectiveness. Batch testing and stability requirements are part of the FDA's evaluation process to ensure that imported drugs are safe and effective for consumer use.
4Recent Regulatory Changes
Between 2024 and 2026, the FDA has implemented several policy changes affecting pharmaceutical imports into the United States. These changes include updates to import procedures, enhanced scrutiny of foreign manufacturing facilities, and revisions to labeling and documentation requirements. The FDA has also introduced new pathways for the importation of certain prescription drugs originally intended for foreign markets, aiming to increase access to affordable medications. Importers must stay informed about these regulatory changes to ensure compliance and maintain uninterrupted access to the U.S. market.
Lifestar Pharma Llc. — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Lifestar Pharma LLC's product strategy focuses on high-barrier generics, targeting therapeutic areas with significant market demand and complexity. The company's emphasis on advanced antibiotics, respiratory treatments, and over-the-counter (OTC) products aligns with the growing need for affordable and effective medications in these categories. By concentrating on complex generics, Lifestar Pharma aims to address unmet medical needs and differentiate itself in the competitive U.S. pharmaceutical market. The selection of specific therapeutic areas is driven by market demand, regulatory pathways, and the company's expertise in developing challenging formulations.
2Sourcing Profile
Lifestar Pharma LLC's sourcing strategy is centered on importing finished pharmaceutical formulations from India, primarily from its parent company, Mankind Pharma Limited. This approach allows the company to leverage Mankind Pharma's manufacturing capabilities and established quality standards. The focus on finished formulations, rather than active pharmaceutical ingredients (APIs) or bulk drugs, enables Lifestar Pharma to streamline its supply chain and ensure consistent product quality. India's robust pharmaceutical manufacturing sector, with its adherence to international quality standards, makes it a strategic sourcing partner for Lifestar Pharma.
3Market Positioning
Based on its product mix, Lifestar Pharma LLC serves the U.S. pharmaceutical market by supplying specialty generics to various segments, including retail pharmacies, hospitals, and government tenders. The company's focus on high-barrier generics positions it to meet the needs of healthcare providers seeking cost-effective alternatives to branded medications. By offering a diverse portfolio of complex generics, Lifestar Pharma aims to enhance patient access to essential medicines across different healthcare settings.
Seller's Guide — How to Become a Supplier to Lifestar Pharma Llc.
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to enter the U.S. market by partnering with Lifestar Pharma
Frequently Asked Questions — Lifestar Pharma Llc.
What products does Lifestar Pharma Llc. import from India?
Lifestar Pharma Llc. imports 12 pharmaceutical products across 9 categories. Top imports: Nitrofurantoin ($9.1M), Formoterol ($6.4M), Phenylephrine ($6.4M), Ofloxacin ($5.8M), Fenofibrate ($4.7M).
Who supplies pharmaceuticals to Lifestar Pharma Llc. from India?
Lifestar Pharma Llc. sources from 1 verified Indian suppliers. The primary supplier is Mankind Pharma Limited (100.0% of imports, $35.1M).
What is Lifestar Pharma Llc.'s total pharmaceutical import value?
Lifestar Pharma Llc.'s total pharmaceutical import value from India is $43.1M, based on 1,217 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Lifestar Pharma Llc. focus on?
Lifestar Pharma Llc. imports across 9 categories. The largest: Advanced Antibiotics (35.3%), Respiratory (14.9%), Respiratory & OTC (14.8%).
Get Full Lifestar Pharma Llc. Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Lifestar Pharma Llc. identified across shipments using consignee name normalization, aggregating 6 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Lifestar Pharma Llc.'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 1,217 individual customs records matching Lifestar Pharma Llc..
- 5.Supplier Verification: Lifestar Pharma Llc. sources from 1 verified Indian suppliers across 697 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
12 Products Tracked
9 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 6 company name variants from customs records. For current shipment-level data, contact TransData Nexus.