Krka, D.d., Novo Mesto
Pharmaceutical Importer · Slovenia · Analgesics & Antipyretics Focus · $14.3M Total Trade · DGFT Verified
Krka, D.d., Novo Mesto is a pharmaceutical importer based in Slovenia with a total trade value of $14.3M across 3 products in 3 therapeutic categories. Based on 287 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Krka, D.d., Novo Mesto sources from 6 verified Indian suppliers, with Unichem Laboratories Limited accounting for 57.2% of imports.
Krka, D.d., Novo Mesto — Import Portfolio & Supplier Network
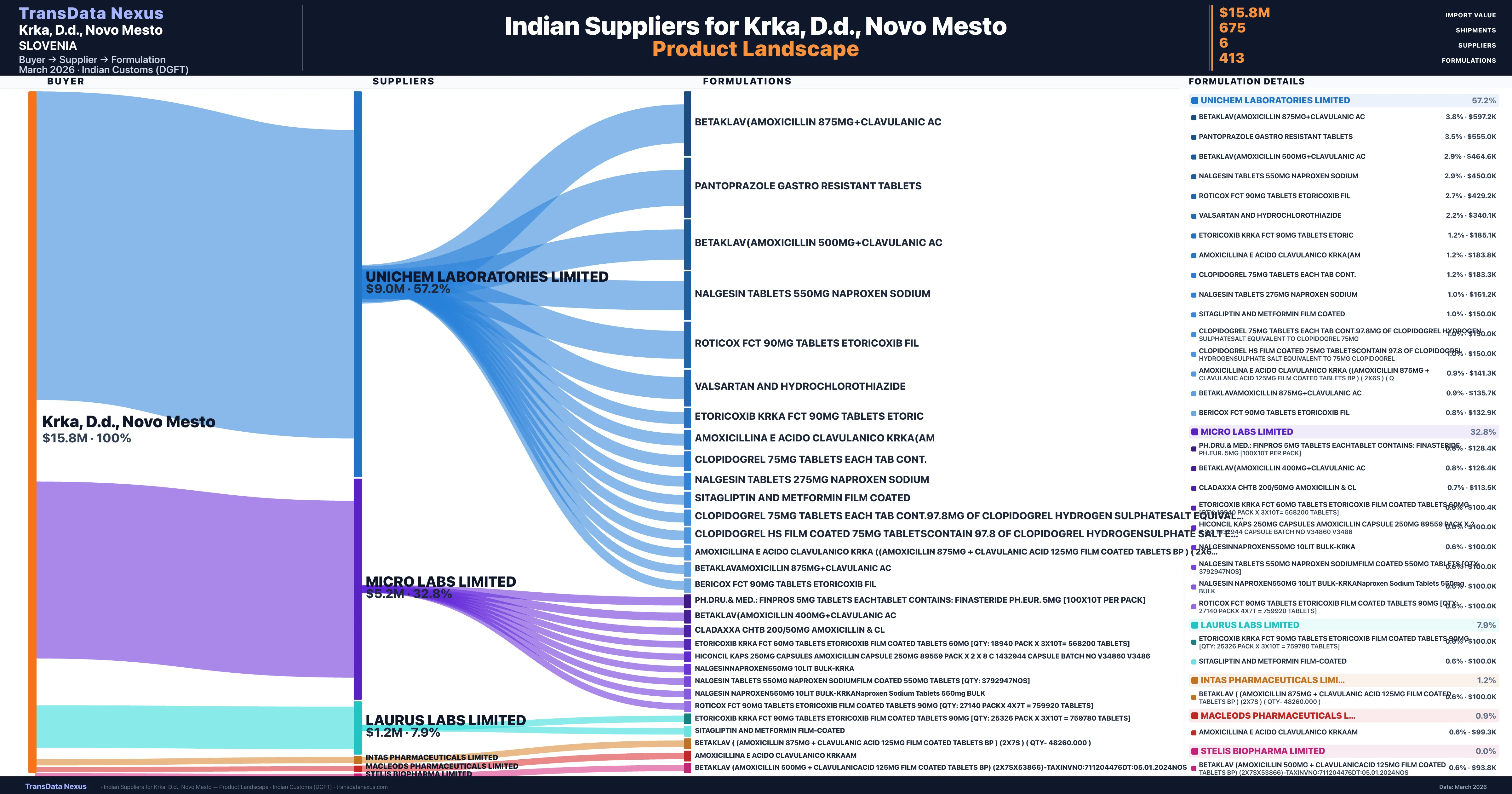
Who Are the Verified Indian Suppliers to Krka, D.d., Novo Mesto?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Unichem Laboratories Limited | $9.0M | 378 | 57.2% |
| Micro Labs Limited | $5.2M | 246 | 32.8% |
| Laurus Labs Limited | $1.2M | 26 | 7.9% |
| Intas Pharmaceuticals Limited | $183.2K | 11 | 1.2% |
| Macleods Pharmaceuticals Limited | $141.3K | 12 | 0.9% |
| Stelis Biopharma Limited | $99 | 2 | 0.0% |
Krka, D.d., Novo Mesto sources from 6 verified Indian suppliers across 413 distinct formulations. The supply base is diversified across 6 suppliers, reducing single-source dependency risk.
What Formulations Does Krka, D.d., Novo Mesto Import?
| Formulation | Value | Ships |
|---|---|---|
| Betaklav(amoxicillin 875MG+clavulanic | $597.2K | 25 |
| Pantoprazole Gastro Resistant | $555.0K | 12 |
| Betaklav(amoxicillin 500MG+clavulanic | $464.6K | 17 |
| Nalgesin tablets 550MG naproxen | $450.0K | 9 |
| Roticox fct 90MG tablets etoricoxib | $429.2K | 16 |
| Valsartan and hydrochlorothiazide | $340.1K | 7 |
| Etoricoxib krka fct 90MG tablets | $185.1K | 6 |
| Amoxicillina e acido clavulanico | $183.8K | 4 |
| Clopidogrel 75MG tablets Each TAB cont. | $183.3K | 4 |
| Nalgesin tablets 275MG naproxen | $161.2K | 4 |
| Sitagliptin and metformin film | $150.0K | 3 |
| Clopidogrel 75MG tablets Each TAB cont.97.8MG of clopidogrel hydrogen sulphatesalt equivalent to clopidogrel 75MG | $150.0K | 3 |
| Clopidogrel hs film coated 75MG tabletscontain 97.8 of clopidogrel hydrogensulphate salt equivalent to 75MG | $150.0K | 3 |
| Amoxicillina e acido clavulanico krka ((amoxicillin 875MG + clavulanic acid 125MG film coated tablets BP ) ( 2x6s ) | $141.3K | 3 |
| Betaklavamoxicillin 875MG+clavulanic | $135.7K | 3 |
Krka, D.d., Novo Mesto imports 413 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Krka, D.d., Novo Mesto Import?
Top Products by Import Value
Krka, D.d., Novo Mesto Therapeutic Categories — 3 Specializations
Krka, D.d., Novo Mesto imports across 3 therapeutic categories, with Analgesics & Antipyretics (88.5%), Cardiovascular (9.1%), Diabetes & Endocrine (2.4%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Analgesics & Antipyretics
1 products · 88.5% · $12.7M
Cardiovascular
1 products · 9.1% · $1.3M
Diabetes & Endocrine
1 products · 2.4% · $350.0K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Naproxen | Analgesics & Antipyretics | $12.7M | 254 | 4.7% | 4 |
| 2 | Clopidogrel | Cardiovascular | $1.3M | 26 | 0.5% | 12 |
| 3 | Sitagliptin | Diabetes & Endocrine | $350.0K | 7 | 0.5% | 12 |
Krka, D.d., Novo Mesto imports 3 pharmaceutical products across 3 categories into Slovenia totaling $14.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Krka, D.d., Novo Mesto.
Request DemoKrka, D.d., Novo Mesto — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Krka, d. d., Novo Mesto, established on April 23, 1954, is a Slovenian multinational pharmaceutical company headquartered in Novo Mesto, Slovenia. The company specializes in the development, production, and marketing of generic pharmaceuticals, non-prescription products, and animal health products. Its product portfolio spans various therapeutic areas, including cardiovascular, gastrointestinal, central nervous system, oncology, pain relief, and anti-infectives. Krka markets its products under numerous brand names, such as Atoris, Roswera, Ampril, Lorista, and Nolpaza. (krka.biz)
In 2023, Krka reported total sales of €1.806 billion, underscoring its significant presence in the global pharmaceutical market. The company employs over 12,500 individuals and operates in more than 70 countries worldwide. Its production facilities are located in Slovenia, with subsidiaries in the Russian Federation, Poland, Croatia, and Germany. Additionally, Krka has a joint venture in China, reflecting its expansive international footprint. (krka.biz)
2Distribution Network
Krka's distribution network is extensive, encompassing both domestic and international markets. Within Slovenia, the company operates multiple production sites, including those in Ločna and Bršljin (both in Novo Mesto), Krško, Šentjernej, and Ljutomer. These facilities are strategically located to facilitate efficient distribution across the country. Internationally, Krka's subsidiaries and joint ventures in the Russian Federation, Poland, Croatia, Germany, and China play a crucial role in the company's distribution strategy, ensuring a robust supply chain that caters to a diverse global clientele. (krka.biz)
3Industry Role
Krka, d. d., Novo Mesto holds a prominent position in Slovenia's pharmaceutical supply chain as a primary wholesaler and distributor. The company's extensive product range and established market presence enable it to serve a wide array of healthcare providers, including hospitals, clinics, and pharmacies. Krka's role extends beyond distribution, as it also engages in the development and manufacturing of pharmaceutical products, positioning itself as a key player in both the production and supply of medicines within Slovenia and internationally. (krka.biz)
Supplier Relationship Intelligence — Krka, D.d., Novo Mesto
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Krka's sourcing strategy exhibits a high degree of concentration, with a significant portion of its pharmaceutical imports originating from India. Between 2022 and 2026, the company imported finished pharmaceutical formulations valued at $14.3 million USD from India, encompassing 287 shipments and 413 unique formulations. The top five products imported include Naproxen ($12.7 million, 88.5% share), Clopidogrel ($1.3 million, 9.1% share), and Sitagliptin ($350,000, 2.4% share). This concentration indicates a strategic reliance on Indian suppliers for key therapeutic areas, particularly in analgesics and antipyretics.
The primary suppliers from India are UNICHEM LABORATORIES LIMITED, MICRO LABS LIMITED, and LAURUS LABS LIMITED, accounting for 57.2%, 32.8%, and 7.9% of the total import value, respectively. This supplier concentration suggests a stable and established relationship between Krka and these Indian manufacturers. However, the heavy reliance on a limited number of suppliers may pose risks related to supply chain disruptions, pricing fluctuations, and geopolitical factors affecting trade relations.
2Supply Chain Resilience
Krka's supply chain resilience concerning its Indian imports appears robust, given the established relationships with key suppliers such as UNICHEM LABORATORIES LIMITED, MICRO LABS LIMITED, and LAURUS LABS LIMITED. The diversity of formulations—413 unique products—indicates a broad sourcing strategy that mitigates risks associated with single-product dependencies. However, the concentration of imports in specific therapeutic categories, notably analgesics and antipyretics, suggests that any disruptions in these product lines could impact Krka's ability to meet market demand.
To enhance supply chain resilience, Krka may consider diversifying its supplier base further, exploring alternative sourcing options, and establishing contingency plans to address potential disruptions. Additionally, maintaining strong relationships with existing suppliers and monitoring geopolitical developments that could affect trade routes and regulations is crucial for sustaining a reliable supply chain.
3Strategic Implications
Krka's sourcing pattern, characterized by a high concentration of imports from Indian suppliers, positions the company competitively in the Slovenian pharmaceutical market, particularly in the analgesics and antipyretics segments. The established relationships with key Indian manufacturers likely contribute to favorable pricing and consistent product quality, enhancing Krka's market position.
For Indian exporters aiming to become alternative suppliers to Krka, understanding the company's specific product requirements, quality standards, and regulatory compliance expectations is essential. Demonstrating the ability to meet these criteria, along with offering competitive pricing and reliable delivery schedules, can facilitate entry into Krka's supply chain.
Importing Pharmaceuticals into Slovenia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Slovenia
1Regulatory Authority & Framework
In Slovenia, the Agency for Medicinal Products and Medical Devices (JAZMP) serves as the primary regulatory authority overseeing the pharmaceutical sector. JAZMP is responsible for the evaluation, approval, and monitoring of medicinal products, ensuring they meet the required safety, efficacy, and quality standards. The legislative framework governing pharmaceutical imports includes the Medicines Act, which outlines the procedures for marketing authorization, importation, and distribution of medicinal products.
For Indian generics to be marketed in Slovenia, they must undergo a centralized marketing authorization procedure through the European Medicines Agency (EMA). This process involves a comprehensive assessment of the medicinal product's quality, safety, and efficacy. Once approved by the EMA, the product can be marketed across all EU member states, including Slovenia. This centralized procedure streamlines the approval process for generics, facilitating their entry into the Slovenian market.
2Import Licensing & GMP
Import licensing in Slovenia is regulated by JAZMP, which ensures that all imported medicinal products comply with Slovenian and EU regulations. Importers must hold a valid wholesale distribution authorization issued by JAZMP, demonstrating their capability to handle pharmaceutical products responsibly. Additionally, Good Manufacturing Practice (GMP) certification is mandatory for all imported medicinal products. The EU recognizes GMP certificates from countries with equivalent standards, such as those issued by the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Therefore, Indian manufacturers seeking to export to Slovenia must ensure their facilities are GMP-compliant and hold the necessary certifications recognized by the EU.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality, safety, and efficacy. Stability studies are also required to ensure that products maintain their intended quality throughout their shelf life. Labeling requirements stipulate that product information be provided in Slovenian, including details such as the product name, active ingredients, dosage form, strength, and instructions for use. Serialization mandates are in place to prevent counterfeit products, requiring unique identifiers on packaging to track and trace products throughout the supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, Slovenia has implemented several regulatory changes affecting pharmaceutical imports. These include stricter requirements for GMP certification, enhanced batch testing protocols, and updated labeling standards to improve patient safety and product traceability. Additionally, Slovenia has aligned its import regulations more closely with EU directives, emphasizing the need for compliance with EU-wide standards for medicinal products. These changes aim to strengthen the quality assurance processes and ensure that imported pharmaceuticals meet the highest standards of safety and efficacy.
Krka, D.d., Novo Mesto — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Krka's product strategy focuses on therapeutic areas with high market demand, particularly in analgesics and antipyretics, as evidenced by the significant import value of Naproxen. The company's emphasis on cardiovascular and diabetes medications aligns with global health trends, addressing prevalent conditions in the aging population. By concentrating on these therapeutic categories, Krka aims to meet the needs of a broad patient base, ensuring the availability of essential medicines in the Slovenian market.
2Sourcing Profile
Krka's sourcing strategy for generic drugs involves importing finished pharmaceutical formulations from established manufacturers in India. The company's preference for specific formulations, such as Naproxen, Clopidogrel, and Sitagliptin, indicates a targeted approach to procurement, focusing on high-demand products. India's well-established pharmaceutical industry, characterized by its compliance with international quality standards and competitive pricing, makes it a strategic partner for Krka's sourcing needs.
3Market Positioning
Krka serves multiple segments of the Slovenian pharmaceutical market, including retail pharmacies, hospitals, and government tenders. The company's diverse product portfolio enables it to cater to various healthcare providers, ensuring the availability of essential medicines across different channels. Krka's role as a primary wholesaler and distributor further solidifies its position in the Slovenian market, facilitating efficient supply chain management and product accessibility.
Seller's Guide — How to Become a Supplier to Krka, D.d., Novo Mesto
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to enter Krka's supply chain, particularly
Frequently Asked Questions — Krka, D.d., Novo Mesto
What products does Krka, D.d., Novo Mesto import from India?
Krka, D.d., Novo Mesto imports 3 pharmaceutical products across 3 categories. Top imports: Naproxen ($12.7M), Clopidogrel ($1.3M), Sitagliptin ($350.0K).
Who supplies pharmaceuticals to Krka, D.d., Novo Mesto from India?
Krka, D.d., Novo Mesto sources from 6 verified Indian suppliers. The primary supplier is Unichem Laboratories Limited (57.2% of imports, $9.0M).
What is Krka, D.d., Novo Mesto's total pharmaceutical import value?
Krka, D.d., Novo Mesto's total pharmaceutical import value from India is $14.3M, based on 287 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Krka, D.d., Novo Mesto focus on?
Krka, D.d., Novo Mesto imports across 3 categories. The largest: Analgesics & Antipyretics (88.5%), Cardiovascular (9.1%), Diabetes & Endocrine (2.4%).
Get Full Krka, D.d., Novo Mesto Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Krka, D.d., Novo Mesto identified across shipments using consignee name normalization, aggregating 4 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Krka, D.d., Novo Mesto's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 287 individual customs records matching Krka, D.d., Novo Mesto.
- 5.Supplier Verification: Krka, D.d., Novo Mesto sources from 6 verified Indian suppliers across 413 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 4 company name variants from customs records. For current shipment-level data, contact TransData Nexus.