IP RIM
Pharmaceutical Importer · Kazakhstan · Ayurvedic & Herbal Products Focus · $1.1M Total Trade · DGFT Verified
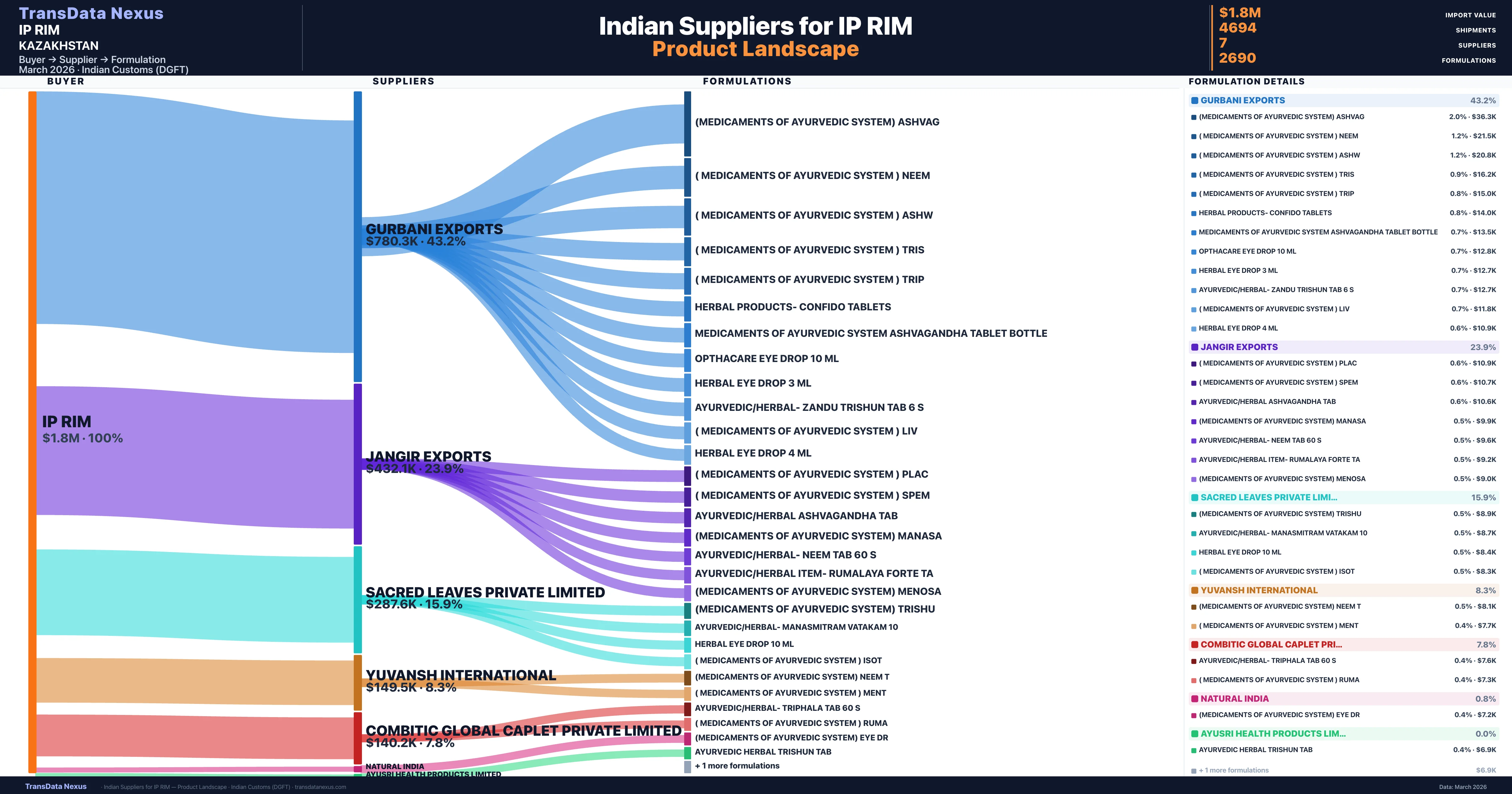
IP RIM is a pharmaceutical importer based in Kazakhstan with a total trade value of $1.1M across 3 products in 1 therapeutic categories. Based on 2,818 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. IP RIM sources from 7 verified Indian suppliers, with Gurbani Exports accounting for 43.2% of imports.
IP RIM — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to IP RIM?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Gurbani Exports | $780.3K | 1,840 | 43.2% |
| Jangir Exports | $432.1K | 545 | 23.9% |
| Sacred Leaves Private Limited | $287.6K | 1,603 | 15.9% |
| Yuvansh International | $149.5K | 430 | 8.3% |
| Combitic Global Caplet Private Limited | $140.2K | 173 | 7.8% |
| Natural India | $14.5K | 100 | 0.8% |
| Ayusri Health Products Limited | $580 | 3 | 0.0% |
IP RIM sources from 7 verified Indian suppliers across 2,690 distinct formulations. The supply base is diversified across 7 suppliers, reducing single-source dependency risk.
What Formulations Does IP RIM Import?
| Formulation | Value | Ships |
|---|---|---|
| (medicaments of ayurvedic system) | $36.3K | 4 |
| ( medicaments of ayurvedic system ) | $21.5K | 25 |
| ( medicaments of ayurvedic system ) | $20.8K | 23 |
| ( medicaments of ayurvedic system ) | $16.2K | 9 |
| ( medicaments of ayurvedic system ) | $15.0K | 34 |
| Herbal products- confido | $14.0K | 1 |
| Medicaments of ayurvedic system ashvagandha tablet | $13.5K | 1 |
| Opthacare eye drop 10 ML | $12.8K | 2 |
| Herbal eye drop 3 ML | $12.7K | 8 |
| Ayurvedic/herbal- zandu trishun TAB 6 | $12.7K | 1 |
| ( medicaments of ayurvedic system ) | $11.8K | 19 |
| Herbal eye drop 4 ML | $10.9K | 4 |
| ( medicaments of ayurvedic system ) | $10.9K | 12 |
| ( medicaments of ayurvedic system ) | $10.7K | 8 |
| Ayurvedic/herbal ashvagandha TAB | $10.6K | 2 |
IP RIM imports 2,690 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does IP RIM Import?
Top Products by Import Value
IP RIM Therapeutic Categories — 1 Specializations
IP RIM imports across 1 therapeutic categories, with Ayurvedic & Herbal Products (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Ayurvedic & Herbal Products
3 products · 100.0% · $1.1M
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ayurvedic | Ayurvedic & Herbal Products | $635.7K | 1,529 | 2.8% | 7 |
| 2 | Herbal | Ayurvedic & Herbal Products | $471.7K | 1,232 | 2.5% | 6 |
| 3 | Kwatha | Ayurvedic & Herbal Products | $14.0K | 57 | 2.6% | 11 |
IP RIM imports 3 pharmaceutical products across 1 categories into Kazakhstan totaling $1.1M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
IP RIM — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
IP RIM is a pharmaceutical importer and buyer based in Kazakhstan, specializing in the acquisition of finished pharmaceutical formulations from India. The company has engaged in 2,818 shipments, importing a total value of $1.1 million USD worth of products. Their portfolio comprises 2,690 unique formulations across three products within a single therapeutic category, indicating a focused approach to their product offerings. The top five products imported include Ayurvedic products ($636K), Herbal products ($472K), and Kwatha ($14K), with Ayurvedic and Herbal products accounting for 100% of their imports. This concentration suggests a strategic emphasis on these specific product lines.
2Distribution Network
While specific details about IP RIM's warehouse locations and logistics capabilities are not publicly available, their extensive import activities indicate a well-established distribution network within Kazakhstan. The company has engaged with seven unique Indian suppliers, including GURBANI EXPORTS, JANGIR EXPORTS, and SACRED LEAVES PRIVATE LIMITED, suggesting a robust supply chain capable of handling a diverse range of products. Their operations likely cover major urban centers and healthcare facilities across Kazakhstan, ensuring widespread availability of their imported pharmaceutical products.
3Industry Role
IP RIM functions primarily as a pharmaceutical importer and buyer, focusing on sourcing finished pharmaceutical formulations from India. Their role in Kazakhstan's pharmaceutical supply chain is that of a wholesaler, distributing imported products to various stakeholders within the healthcare sector. By concentrating on Ayurvedic and Herbal products, IP RIM caters to a niche market segment, addressing the demand for traditional and alternative medicine options among Kazakhstani consumers.
Supplier Relationship Intelligence — IP RIM
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
IP RIM's sourcing strategy exhibits a high degree of concentration, with the top five products accounting for 100% of their imports. This single-source dependency could pose risks, such as supply chain disruptions or price volatility. However, the company's engagement with multiple suppliers—seven unique Indian companies—indicates a diversified approach within their chosen product lines. The substantial number of shipments (2,818) and the total import value ($1.1 million USD) suggest stable and ongoing relationships with these suppliers, mitigating potential risks associated with sourcing concentration.
2Supply Chain Resilience
IP RIM's supply chain resilience appears robust, given their engagement with multiple suppliers and a diverse range of formulations. The company's focus on Ayurvedic and Herbal products aligns with global trends towards natural and alternative medicines, potentially enhancing the appeal and demand for their offerings. While specific details about backup suppliers and shipping routes are not available, the volume and frequency of shipments suggest a well-established and resilient supply chain capable of meeting market demands.
3Strategic Implications
IP RIM's concentrated sourcing strategy positions them as a specialized player in Kazakhstan's pharmaceutical market, catering to the growing demand for Ayurvedic and Herbal products. For Indian exporters, this focus presents an opportunity to establish long-term partnerships with a dedicated importer, potentially leading to increased market share in Kazakhstan. However, the high concentration of imports in these product categories also means that any shifts in market demand or regulatory changes could significantly impact IP RIM's business operations.
Importing Pharmaceuticals into Kazakhstan — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Kazakhstan
1Regulatory Authority & Framework
The primary regulatory body overseeing pharmaceutical imports in Kazakhstan is the Ministry of Health. The Code of the Republic of Kazakhstan on People's Health and Healthcare System outlines the procedures for the importation of medicines, medical devices, and medical equipment. According to this code, entities involved in the circulation of pharmaceutical products and medical devices must possess a license for wholesale trading of pharmaceutical products or be included in the register of entities of public health services administering wholesale trading of medical products. (wipolex-res.wipo.int)
2Import Licensing & GMP
Import licensing in Kazakhstan requires entities to hold a license for wholesale trading of pharmaceutical products or to be included in the register of entities of public health services administering wholesale trading of medical products. Additionally, the Ministry of Health may require a site inspection if the manufacturing site does not hold a valid EAEU GMP certificate. (wipolex-res.wipo.int)
3Quality & Labeling
Imported pharmaceutical products must comply with Kazakhstan's quality standards, which include proper production practices and quality control measures. Batch testing and stability requirements are enforced to ensure product safety and efficacy. Labeling must be in the Kazakh and Russian languages, providing clear information on usage, dosage, and storage. Serialization mandates are in place to track and trace products throughout the supply chain, enhancing transparency and combating counterfeit products.
4Recent Regulatory Changes
In June 2023, the Rules on Wholesale and Retail Sale of Medicines and Medical Devices were amended by the Order of the Minister of Health of the Republic of Kazakhstan No. 98. These amendments introduced provisions on the return of medicines and a ban on selling medicines to children. Additionally, the sale of bulk products of medicines or medical devices was prohibited.
IP RIM — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
IP RIM's focus on Ayurvedic and Herbal products aligns with the growing global interest in natural and alternative medicines. This strategic choice caters to a segment of the Kazakhstani market seeking traditional remedies and wellness solutions. The market demand for such products is driven by consumer preferences for holistic health approaches and the cultural significance of traditional medicine in Kazakhstan.
2Sourcing Profile
IP RIM's sourcing strategy is centered on importing finished pharmaceutical formulations from India, particularly Ayurvedic and Herbal products. This approach leverages India's rich heritage in traditional medicine and its established manufacturing capabilities. By focusing on finished products, IP RIM ensures quality control and compliance with Kazakhstan's regulatory standards, facilitating smoother market entry and distribution.
3Market Positioning
With a product mix concentrated on Ayurvedic and Herbal formulations, IP RIM serves a niche segment within Kazakhstan's pharmaceutical market. Their offerings are likely distributed through retail pharmacies, private clinics, and hospitals, catering to consumers seeking alternative and traditional medicine options. The company's strategic focus positions them as a specialized wholesaler in this segment, differentiating them from competitors in the broader pharmaceutical market.
Seller's Guide — How to Become a Supplier to IP RIM
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with IP RIM, given their focused product strategy and established distribution network. Suppliers offering high-quality Ayurvedic and Herbal products that meet Kazakhstan's regulatory standards could find a receptive partner in IP RIM. However, potential suppliers should be aware of the competitive landscape and the need to align with IP RIM's specific product focus to effectively penetrate the market.
2Requirements & Qualifications
Indian exporters seeking to supply IP RIM and the Kazakhstan market must ensure their products are registered with the Ministry of Health and comply with Good Manufacturing Practice (GMP) standards. Possessing a valid EAEU GMP certificate can facilitate the import process, as it may exempt the manufacturer from local inspections. Additionally, products must meet Kazakhstan's quality standards, including proper production practices, batch testing, stability requirements, and labeling in Kazakh and Russian languages.
3How to Approach
To establish a partnership with IP RIM, Indian exporters should initiate contact by providing a comprehensive product portfolio and demonstrating compliance with Kazakhstan's regulatory requirements. Engaging in discussions about product registration processes, including dossier preparation and submission, is crucial. Participating in government tenders and understanding the wholesale distribution authorization process will enhance the partnership's success. Setting clear timelines for regulatory approvals and shipments will facilitate a smooth collaboration.
Frequently Asked Questions — IP RIM
What products does IP RIM import from India?
IP RIM imports 3 pharmaceutical products across 1 categories. Top imports: Ayurvedic ($635.7K), Herbal ($471.7K), Kwatha ($14.0K).
Who supplies pharmaceuticals to IP RIM from India?
IP RIM sources from 7 verified Indian suppliers. The primary supplier is Gurbani Exports (43.2% of imports, $780.3K).
What is IP RIM's total pharmaceutical import value?
IP RIM's total pharmaceutical import value from India is $1.1M, based on 2,818 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does IP RIM focus on?
IP RIM imports across 1 categories. The largest: Ayurvedic & Herbal Products (100.0%).
Get Full IP RIM Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: IP RIM identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as IP RIM's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 2,818 individual customs records matching IP RIM.
- 5.Supplier Verification: IP RIM sources from 7 verified Indian suppliers across 2,690 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.