Indira Gandhi Memorial Hospital
Pharmaceutical Importer · Maldives · Advanced Oncology Focus · $962.1K Total Trade · DGFT Verified
Indira Gandhi Memorial Hospital is a pharmaceutical importer based in Maldives with a total trade value of $962.1K across 5 products in 1 therapeutic categories. Based on 106 verified import shipments from Indian Customs (DGFT) records, Indira Gandhi Memorial Hospital is the #1 buyer in 2 products including Osimertinib, Pembrolizumab. Indira Gandhi Memorial Hospital sources from 1 verified Indian supplier, with Nu Hospitals Private Limited accounting for 100.0% of imports.
Indira Gandhi Memorial Hospital — Import Portfolio & Supplier Network
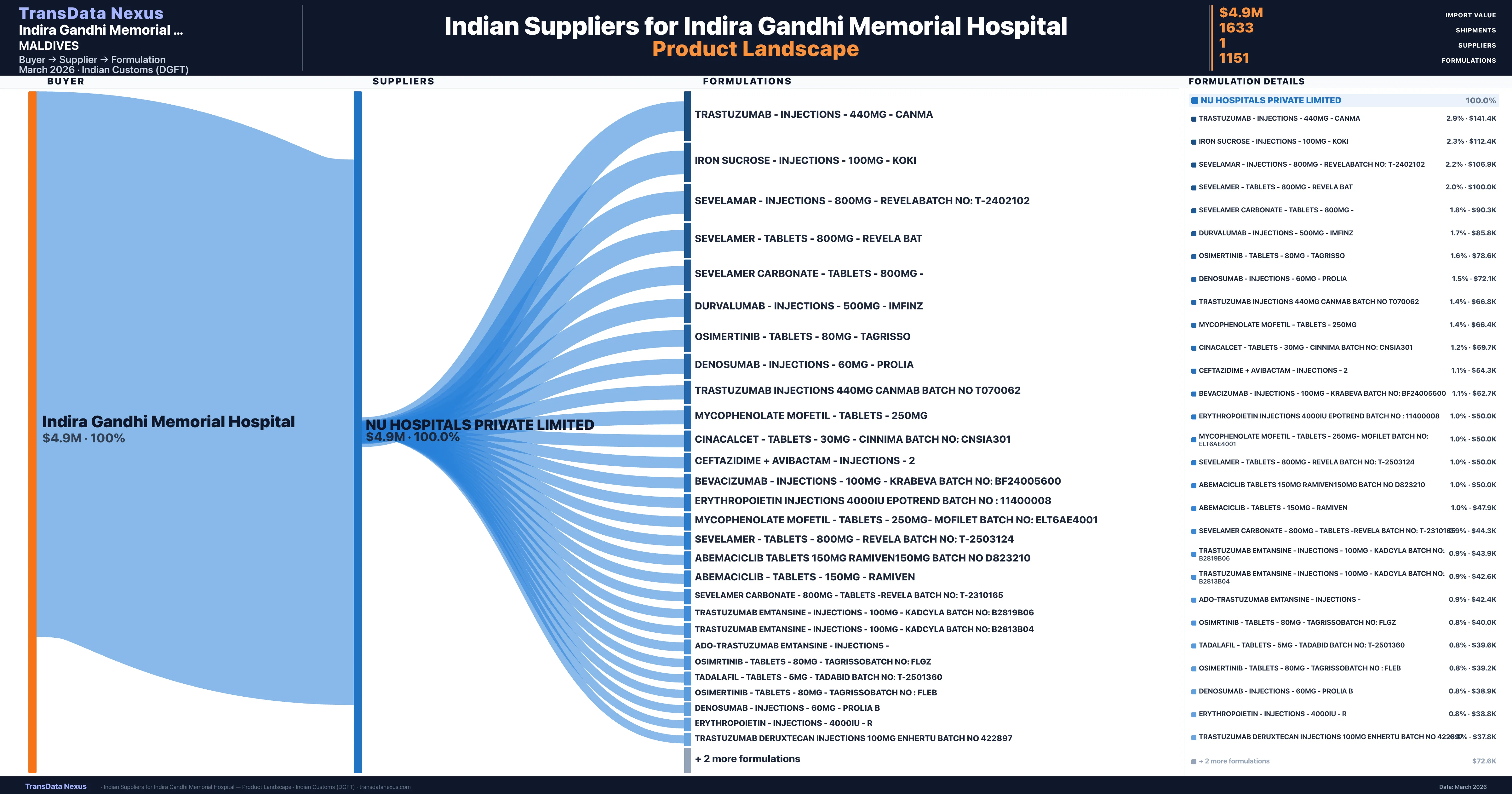
Who Are the Verified Indian Suppliers to Indira Gandhi Memorial Hospital?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Nu Hospitals Private Limited | $4.9M | 1,633 | 100.0% |
Indira Gandhi Memorial Hospital sources from 1 verified Indian supplier across 1,151 distinct formulations. The sourcing is highly concentrated — Nu Hospitals Private Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Indira Gandhi Memorial Hospital Import?
| Formulation | Value | Ships |
|---|---|---|
| Trastuzumab - injections - 440MG - | $141.4K | 15 |
| Iron sucrose - injections - 100MG - | $112.4K | 11 |
| Sevelamar - injections - 800MG - revelabatch no | $106.9K | 3 |
| Sevelamer - tablets - 800MG - revela | $100.0K | 2 |
| Sevelamer carbonate - tablets - 800MG | $90.3K | 2 |
| Durvalumab - injections - 500MG - | $85.8K | 6 |
| Osimertinib - tablets - 80MG - | $78.6K | 2 |
| Denosumab - injections - 60MG - | $72.1K | 10 |
| Trastuzumab injections 440MG canmab batch no | $66.8K | 2 |
| Mycophenolate mofetil - tablets - 250MG | $66.4K | 6 |
| Cinacalcet - tablets - 30MG - cinnima batch no | $59.7K | 2 |
| Ceftazidime + avibactam - injections - | $54.3K | 7 |
| Bevacizumab - injections - 100MG - krabeva batch no | $52.7K | 2 |
| Erythropoietin injections 4000iu epotrend batch no | $50.0K | 1 |
| Mycophenolate mofetil - tablets - 250MG- mofilet batch no | $50.0K | 1 |
Indira Gandhi Memorial Hospital imports 1,151 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Indira Gandhi Memorial Hospital Import?
Top Products by Import Value
Indira Gandhi Memorial Hospital Therapeutic Categories — 1 Specializations
Indira Gandhi Memorial Hospital imports across 1 therapeutic categories, with Advanced Oncology (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Oncology
5 products · 100.0% · $962.1K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Trastuzumab | Advanced Oncology | $654.7K | 61 | 0.8% | 15 |
| 2 | Osimertinib | Advanced Oncology | $130.8K | 4 | 12.0% | 1 |
| 3 | Pembrolizumab | Advanced Oncology | $130.1K | 30 | 6.7% | 1 |
| 4 | Cetuximab | Advanced Oncology | $34.5K | 6 | 4.1% | 3 |
| 5 | Nivolumab | Advanced Oncology | $12.0K | 5 | 0.5% | 7 |
Indira Gandhi Memorial Hospital imports 5 pharmaceutical products across 1 categories into Maldives totaling $962.1K. The company is the #1 buyer for 2 products: Osimertinib, Pembrolizumab.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Indira Gandhi Memorial Hospital.
Request DemoIndira Gandhi Memorial Hospital — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Indira Gandhi Memorial Hospital (IGMH) is the largest government-owned multi-specialty tertiary care facility in the Maldives, located in the capital city of Malé. Established on April 15, 1995, IGMH replaced the previous Central Hospital, which was later demolished to make way for another hospital. The hospital is dedicated to Indira Gandhi and serves as a significant healthcare provider in the region.
As a state-owned entity, IGMH operates under the Ministry of Health and is fully covered by the Aasandha social healthcare insurance scheme. The hospital offers a wide range of services across various departments, including Anesthesiology, Cardiology, Dentistry, Dermatology, Dietetics & Nutrition, Emergency & Trauma, Endocrinology, ENT, Gastroenterology, Gynaecological Oncology, Internal Medicine, Laboratory Medicine, Nephrology, Neurology, Neurosurgery, Obstetrics & Gynecology, Oncology, Ophthalmology, Oral and Maxillofacial Surgery, Orthopedics, Paediatrics, Paediatric Cardiology, Paediatric Neurology, Physiotherapy, Plastic & Reconstructive Surgery, Psychiatry, Pulmonology, Radiology, Rheumatology, Surgery, Surgical Oncology, Speech Therapy, and Urology. (igmh.gov.mv)
2Distribution Network
IGMH's distribution network primarily focuses on the procurement and distribution of pharmaceutical products essential for its diverse medical services. The hospital's logistics capabilities are designed to ensure the timely and efficient delivery of medical supplies to various departments, maintaining a continuous supply chain to meet patient care needs. While specific details about warehouse locations and geographic coverage are not publicly disclosed, IGMH's central location in Malé facilitates access to international shipping routes, enabling the importation of medical supplies from global suppliers.
3Industry Role
In the Maldives' pharmaceutical supply chain, IGMH functions as a primary hospital supplier, directly importing and distributing pharmaceutical products necessary for its operations. The hospital's role is critical in ensuring the availability of essential medications and medical supplies, thereby supporting the healthcare needs of the population. IGMH's direct procurement from international suppliers underscores its pivotal position in the nation's healthcare infrastructure.
Supplier Relationship Intelligence — Indira Gandhi Memorial Hospital
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
IGMH's pharmaceutical sourcing exhibits a high degree of concentration, with a total import value of $962,000 USD from India across 106 shipments, focusing exclusively on five oncology products. This concentrated sourcing strategy indicates a strategic choice to streamline procurement processes and maintain consistency in product quality. The exclusive reliance on a single supplier, NU Hospitals Private Limited, for all shipments suggests a stable and long-term relationship, potentially reducing procurement complexities and ensuring reliable supply. However, this single-source dependency also poses risks, such as vulnerability to supply chain disruptions or changes in supplier terms.
2Supply Chain Resilience
The resilience of IGMH's Indian supply chain appears robust, given the consistent procurement of finished pharmaceutical formulations from a single supplier, NU Hospitals Private Limited. The hospital's focus on a limited number of oncology products may enhance supply chain stability by simplifying logistics and inventory management. However, the lack of backup suppliers and limited formulation diversity could expose IGMH to risks if the primary supplier faces operational challenges or regulatory issues. Additionally, the hospital's reliance on a single shipping route for imports may increase exposure to logistical disruptions.
3Strategic Implications
IGMH's concentrated sourcing strategy positions it to benefit from streamlined procurement processes and potentially favorable terms with its sole supplier. This approach may lead to cost efficiencies and consistent product quality. For Indian exporters, the hospital's exclusive relationship with NU Hospitals Private Limited presents an opportunity to explore alternative supply channels to IGMH, potentially diversifying the hospital's supplier base and mitigating risks associated with single-source dependency.
Importing Pharmaceuticals into Maldives — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Maldives
1Regulatory Authority & Framework
The Maldives Food and Drug Authority (MFDA) is the primary regulatory body overseeing the importation, manufacture, distribution, and sale of pharmaceuticals in the Maldives. The MFDA operates under the Ministry of Health and is responsible for ensuring that all medicinal products meet established safety, efficacy, and quality standards. The MFDA's regulations are designed to protect public health by regulating the pharmaceutical supply chain and ensuring that only approved and safe medications are available in the market. (maldivesvoice.mv)
2Import Licensing & GMP
Import licensing requirements in the Maldives stipulate that all medicine importers and sellers must be registered and licensed by the MFDA. Only drugs listed under the country's Approved Drug List are permitted for import and distribution. The MFDA recognizes Good Manufacturing Practice (GMP) certificates from reputable international standards, including EU GMP, WHO GMP, and PIC/S, ensuring that imported pharmaceuticals adhere to high-quality manufacturing standards. Additionally, wholesale distribution authorization is required for entities involved in the distribution of pharmaceutical products within the country. (maldivesvoice.mv)
3Quality & Labeling
Pharmaceutical products imported into the Maldives must undergo batch testing to ensure compliance with safety and efficacy standards. Stability requirements are enforced to guarantee that medications maintain their intended potency and safety throughout their shelf life. Labeling language requirements mandate that product information be provided in the local language, ensuring clear communication with consumers. Serialization mandates are implemented to enhance traceability and prevent counterfeit products from entering the market, thereby safeguarding public health.
4Recent Regulatory Changes
Between 2024 and 2026, the Maldives Food and Drug Authority (MFDA) has intensified its regulatory oversight, particularly concerning the importation and sale of pharmaceuticals. The MFDA has launched investigations into unauthorized advertising and sale of medicines on social media platforms, emphasizing the need for all medicine importers and sellers to be registered and licensed. These measures aim to ensure that only approved and safe medications are available in the market, thereby protecting public health. (maldivesvoice.mv)
Indira Gandhi Memorial Hospital — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
IGMH's focus on importing oncology products, specifically trastuzumab, osimertinib, pembrolizumab, cetuximab, and nivolumab, aligns with the hospital's strategic emphasis on providing comprehensive cancer care services. The high demand for these medications in the Maldives, driven by the prevalence of cancer cases and the hospital's commitment to offering advanced treatment options, necessitates a reliable supply of these critical drugs. The importation of these specific therapeutic areas underscores IGMH's dedication to enhancing patient outcomes through access to cutting-edge oncology treatments.
2Sourcing Profile
IGMH's sourcing strategy is centered on procuring finished pharmaceutical formulations, particularly in the oncology therapeutic area, from India. This approach ensures access to high-quality medications that meet international standards. The hospital's exclusive reliance on a single supplier, NU Hospitals Private Limited, reflects a strategic decision to maintain consistency in product quality and streamline procurement processes. However, this strategy also highlights the importance of establishing contingency plans to mitigate potential risks associated with single-source dependency.
3Market Positioning
Based on its product mix, IGMH serves the hospital segment of the Maldives market, focusing on providing specialized medical services, particularly in oncology, to both domestic and international patients. The hospital's strategic sourcing and procurement decisions are tailored to meet the specific needs of its patient population, ensuring the availability of essential medications and medical supplies to support its comprehensive healthcare services.
Seller's Guide — How to Become a Supplier to Indira Gandhi Memorial Hospital
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with IGMH, especially in the oncology therapeutic area. While the hospital currently relies on a single supplier, the high demand for specialized medications and the potential benefits of diversifying its supplier base present avenues for collaboration. Indian exporters can explore these opportunities by demonstrating compliance with MFDA regulations, ensuring product quality, and establishing reliable supply chains to meet IGMH's procurement standards.
2Requirements & Qualifications
Indian exporters seeking to supply pharmaceuticals to IGMH and the Maldives market must obtain the necessary import licenses and wholesale distribution authorizations from the Maldives Food and Drug Authority (MFDA). Products must adhere to recognized Good Manufacturing Practice (GMP) standards, such as EU GMP, WHO GMP, or PIC/S. Additionally, compliance with batch testing, stability requirements, labeling language mandates, and serialization regulations is essential to meet the MFDA's quality and safety standards.
3How to Approach
To establish a relationship with IGMH, Indian exporters should initiate contact with the hospital's procurement department to understand specific product requirements and procurement processes. Participating in tenders and responding to Requests for Proposals (RFPs) issued by IGMH can provide opportunities for collaboration. Developing a comprehensive regulatory filing strategy that aligns with MFDA requirements, including obtaining necessary licenses and certifications, is crucial. Setting realistic timelines for product registration, quality assessments, and logistics planning will facilitate a smooth entry into the Maldives pharmaceutical market.
Frequently Asked Questions — Indira Gandhi Memorial Hospital
What products does Indira Gandhi Memorial Hospital import from India?
Indira Gandhi Memorial Hospital imports 5 pharmaceutical products across 1 categories. Top imports: Trastuzumab ($654.7K), Osimertinib ($130.8K), Pembrolizumab ($130.1K), Cetuximab ($34.5K), Nivolumab ($12.0K).
Who supplies pharmaceuticals to Indira Gandhi Memorial Hospital from India?
Indira Gandhi Memorial Hospital sources from 1 verified Indian suppliers. The primary supplier is Nu Hospitals Private Limited (100.0% of imports, $4.9M).
What is Indira Gandhi Memorial Hospital's total pharmaceutical import value?
Indira Gandhi Memorial Hospital's total pharmaceutical import value from India is $962.1K, based on 106 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Indira Gandhi Memorial Hospital focus on?
Indira Gandhi Memorial Hospital imports across 1 categories. The largest: Advanced Oncology (100.0%).
Get Full Indira Gandhi Memorial Hospital Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Indira Gandhi Memorial Hospital identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Indira Gandhi Memorial Hospital's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 106 individual customs records matching Indira Gandhi Memorial Hospital.
- 5.Supplier Verification: Indira Gandhi Memorial Hospital sources from 1 verified Indian suppliers across 1,151 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.