Hemas Pharmaceuticals (pvt) Ltd
Pharmaceutical Importer · Sri Lanka · Nutritional Supplements Focus · $2.1M Total Trade · DGFT Verified
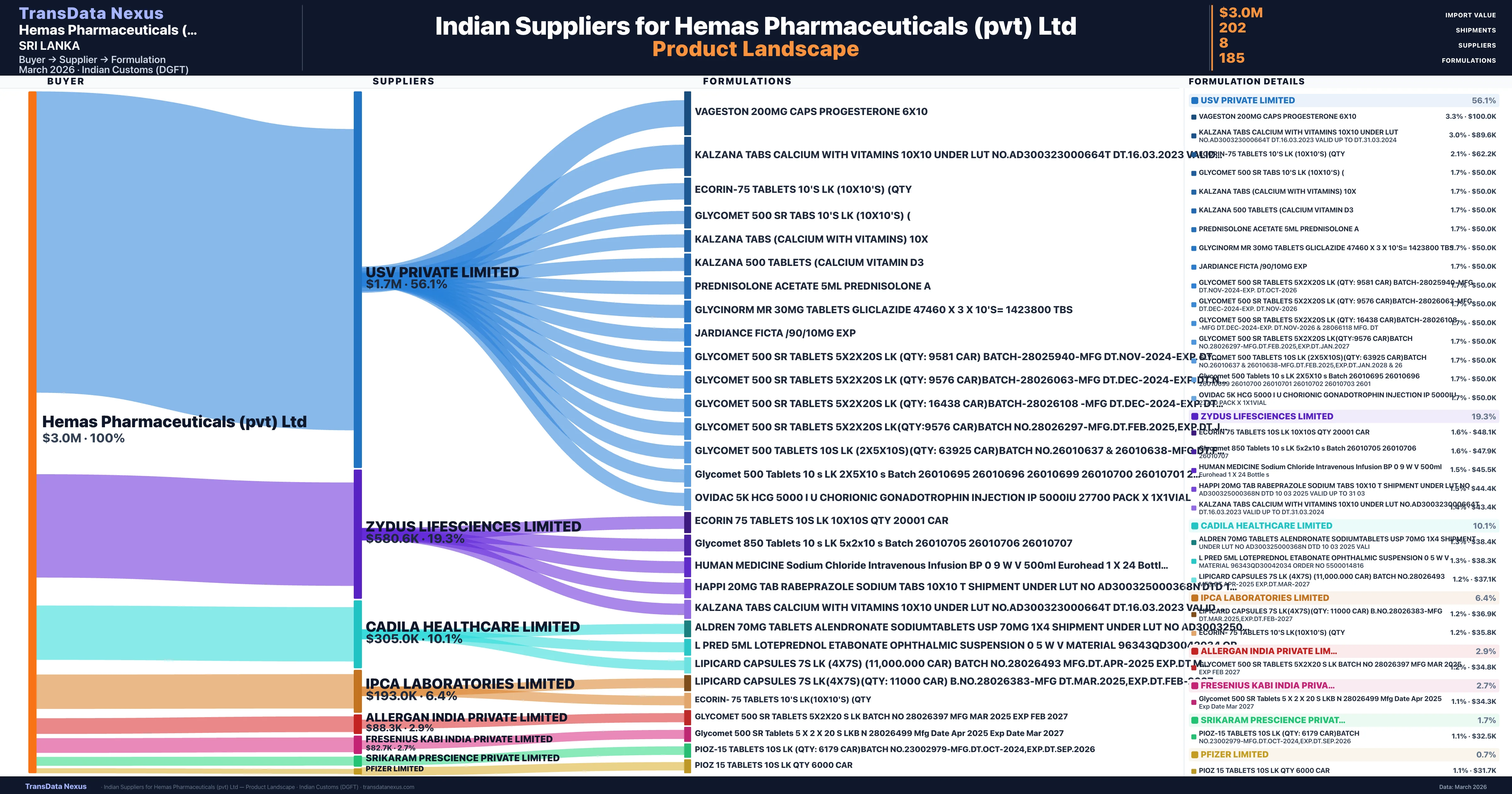
Hemas Pharmaceuticals (pvt) Ltd is a pharmaceutical importer based in Sri Lanka with a total trade value of $2.1M across 4 products in 4 therapeutic categories. Based on 59 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Hemas Pharmaceuticals (pvt) Ltd sources from 8 verified Indian suppliers, with Usv Private Limited accounting for 56.1% of imports.
Hemas Pharmaceuticals (pvt) Ltd — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Hemas Pharmaceuticals (pvt) Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Usv Private Limited | $1.7M | 104 | 56.1% |
| Zydus Lifesciences Limited | $580.6K | 52 | 19.3% |
| Cadila Healthcare Limited | $305.0K | 25 | 10.1% |
| Ipca Laboratories Limited | $193.0K | 12 | 6.4% |
| Allergan India Private Limited | $88.3K | 2 | 2.9% |
| Fresenius Kabi India Private Limited | $82.7K | 4 | 2.7% |
| Srikaram Prescience Private Limited | $50.0K | 1 | 1.7% |
| Pfizer Limited | $22.1K | 2 | 0.7% |
Hemas Pharmaceuticals (pvt) Ltd sources from 8 verified Indian suppliers across 185 distinct formulations. The supply base is diversified across 8 suppliers, reducing single-source dependency risk.
What Formulations Does Hemas Pharmaceuticals (pvt) Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Vageston 200MG CAPS progesterone | $100.0K | 2 |
| Kalzana tabs calcium with vitamins 10x10 under lut no.ad300323000664t dt.16.03.2023 valid up to | $89.6K | 2 |
| Ecorin-75 tablets 10's lk (10x10's) | $62.2K | 2 |
| Glycomet 500 sr tabs 10's lk (10x10's) | $50.0K | 1 |
| Kalzana tabs (calcium with vitamins) | $50.0K | 1 |
| Kalzana 500 tablets (calcium vitamin | $50.0K | 1 |
| Prednisolone acetate 5ML prednisolone | $50.0K | 1 |
| Glycinorm mr 30MG tablets gliclazide 47460 x 3 x 10's= 1423800 | $50.0K | 1 |
| Jardiance ficta /90/10MG | $50.0K | 1 |
| Glycomet 500 sr tablets 5x2x20s lk (qty: 9581 car) batch-28025940-mfg dt.nov-2024-exp. | $50.0K | 1 |
| Glycomet 500 sr tablets 5x2x20s lk (qty: 9576 car)batch-28026063-mfg dt.dec-2024-exp. | $50.0K | 1 |
| Glycomet 500 sr tablets 5x2x20s lk (qty: 16438 car)batch-28026108 -mfg dt.dec-2024-exp. dt.nov-2026 & 28066118 mfg. | $50.0K | 1 |
| Glycomet 500 sr tablets 5x2x20s lk(qty:9576 car)batch no.28026297-mfg.dt.feb.2025 | $50.0K | 1 |
| Glycomet 500 tablets 10s lk (2x5x10s)(qty: 63925 car)batch no.26010637 & 26010638-mfg.dt.feb.2025,exp.dt.jan.2028 & | $50.0K | 1 |
| Glycomet 500 tablets 10 s lk 2x5x10 s batch 26010695 26010696 26010699 26010700 26010701 26010702 26010703 | $50.0K | 1 |
Hemas Pharmaceuticals (pvt) Ltd imports 185 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Hemas Pharmaceuticals (pvt) Ltd Import?
Top Products by Import Value
Hemas Pharmaceuticals (pvt) Ltd Therapeutic Categories — 4 Specializations
Hemas Pharmaceuticals (pvt) Ltd imports across 4 therapeutic categories, with Nutritional Supplements (59.9%), Immunosuppressants (20.9%), Advanced Oncology (13.3%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Nutritional Supplements
1 products · 59.9% · $1.3M
Immunosuppressants
1 products · 20.9% · $435.1K
Advanced Oncology
1 products · 13.3% · $277.4K
Gastrointestinal
1 products · 5.9% · $122.6K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Vitamin | Nutritional Supplements | $1.3M | 25 | 0.9% | 13 |
| 2 | Methotrexate | Immunosuppressants | $435.1K | 9 | 1.2% | 19 |
| 3 | Letrozole | Advanced Oncology | $277.4K | 10 | 0.9% | 11 |
| 4 | Bisacodyl | Gastrointestinal | $122.6K | 15 | 1.4% | 10 |
Hemas Pharmaceuticals (pvt) Ltd imports 4 pharmaceutical products across 4 categories into Sri Lanka totaling $2.1M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Hemas Pharmaceuticals (pvt) Ltd.
Request DemoHemas Pharmaceuticals (pvt) Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Hemas Pharmaceuticals (Pvt) Ltd, established in 1948, is a leading importer and distributor of pharmaceutical products in Sri Lanka. As a member of the Hemas Group, one of the country's most respected conglomerates, Hemas Pharmaceuticals has over seven decades of experience in shaping Sri Lanka's healthcare industry through innovation and an extensive distribution network.
The company offers a comprehensive portfolio, including pharmaceutical, surgical, diagnostics, consumer health, and animal health products. With a market share exceeding 30%, Hemas Pharmaceuticals serves over 3,000 pharmacies and 7,000 healthcare practitioners across the island, demonstrating its significant role in the nation's pharmaceutical distribution.
2Distribution Network
Hemas Pharmaceuticals operates a robust distribution network with a presence in over 3,000 pharmacies and 7,000 healthcare practitioners across Sri Lanka. This extensive reach is supported by a world-class supply chain system, ensuring steady product availability and efficient logistics. The company collaborates with more than 50 distributors, enhancing its capacity to deliver pharmaceutical products nationwide.
3Industry Role
As a primary wholesaler and distributor, Hemas Pharmaceuticals plays a pivotal role in Sri Lanka's pharmaceutical supply chain. The company imports and distributes a wide range of pharmaceutical products, surgical and diagnostic items, consumer health products, and animal health products. Its extensive distribution network and partnerships with over 50 multinational companies underscore its significant position in the market.
Supplier Relationship Intelligence — Hemas Pharmaceuticals (pvt) Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Hemas Pharmaceuticals imports finished pharmaceutical formulations from eight verified Indian suppliers, with USV Private Limited being the largest contributor, accounting for 56.1% of the total import value. This concentration indicates a strategic choice to maintain strong relationships with key suppliers, ensuring consistent product quality and availability. The company's diversified portfolio, with 185 unique formulations, suggests a balanced approach to sourcing, mitigating risks associated with single-source dependency.
2Supply Chain Resilience
Hemas Pharmaceuticals' supply chain demonstrates resilience through its diversified sourcing from multiple Indian suppliers, reducing reliance on any single source. The company's importation of 185 unique formulations indicates a broad product base, enhancing its ability to adapt to market changes. While specific details on backup suppliers and shipping routes are not publicly disclosed, Hemas' established partnerships and extensive distribution network suggest a well-structured and resilient supply chain.
3Strategic Implications
Hemas Pharmaceuticals' sourcing strategy, characterized by strong relationships with key Indian suppliers and a diversified product portfolio, positions the company to maintain a competitive edge in Sri Lanka's pharmaceutical market. For Indian exporters, this approach presents opportunities to strengthen existing partnerships and explore new collaborations, particularly in therapeutic areas where Hemas has a significant market presence.
Importing Pharmaceuticals into Sri Lanka — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Sri Lanka
1Regulatory Authority & Framework
The National Medicines Regulatory Authority (NMRA) is the primary regulatory body overseeing pharmaceutical imports in Sri Lanka. Key legislation governing pharma imports includes the Medicines (Pricing Mechanism for Medicines) Regulations, which grant the NMRA authority to control medicine prices. Importers must register pharmaceutical products with the NMRA and obtain approval before distribution. As of February 2026, the NMRA has introduced new requirements for price submissions, particularly for import licenses expiring on or after 1st February 2026.
2Import Licensing & GMP
Importers must maintain active import licenses and renew them regularly. The NMRA requires that all pharmaceutical products imported into Sri Lanka be registered and approved before distribution. Importers are also required to submit import data in a specified format to the NMRA. Regarding Good Manufacturing Practice (GMP) certifications, the NMRA recognizes international standards such as EU GMP, WHO GMP, and PIC/S. Importers must ensure that their suppliers hold valid GMP certifications to comply with regulatory requirements. (nmra.gov.lk)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure quality and safety. Stability requirements are enforced to guarantee product efficacy throughout its shelf life. Labeling must be in English and Sinhala, providing clear information on usage, dosage, and storage. Serialization mandates are in place to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
In February 2026, the NMRA introduced new requirements for price submissions, particularly affecting import licenses expiring on or after 1st February 2026. This policy change aims to enhance transparency and control over pharmaceutical pricing in Sri Lanka.
Hemas Pharmaceuticals (pvt) Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Hemas Pharmaceuticals focuses on importing products in therapeutic areas such as nutritional supplements, immunosuppressants, and advanced oncology. The significant import value in these categories indicates a strategic alignment with market demand and the company's expertise. The emphasis on nutritional supplements (59.9%) and immunosuppressants (20.9%) suggests a commitment to addressing prevalent health concerns in Sri Lanka.
2Sourcing Profile
Hemas Pharmaceuticals' sourcing strategy emphasizes importing finished pharmaceutical formulations from reputable suppliers, primarily from India. The company's diversified product portfolio, with 185 unique formulations, reflects a comprehensive approach to meeting the diverse healthcare needs of the Sri Lankan market.
3Market Positioning
Hemas Pharmaceuticals serves a broad segment of the Sri Lankan market, including retail pharmacies, hospitals, government tenders, and wholesale distribution. Its extensive distribution network and diverse product portfolio enable the company to cater to various healthcare providers and consumers across the country.
Seller's Guide — How to Become a Supplier to Hemas Pharmaceuticals (pvt) Ltd
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Hemas Pharmaceuticals, particularly in therapeutic areas where the company has a significant market presence. Identifying gaps in Hemas' current sourcing, such as emerging therapeutic categories or innovative formulations, could present avenues for new partnerships.
2Requirements & Qualifications
Indian exporters seeking to supply Hemas Pharmaceuticals must ensure their products are registered with the NMRA and comply with Sri Lankan import regulations. Suppliers must hold valid GMP certifications recognized by the NMRA, such as EU GMP, WHO GMP, or PIC/S. Adherence to quality standards, including batch testing, stability requirements, and labeling mandates, is essential.
3How to Approach
Indian exporters should initiate contact with Hemas Pharmaceuticals by presenting their product portfolios and demonstrating compliance with NMRA regulations. Participating in tenders and engaging in discussions about regulatory filing strategies can facilitate collaboration. Understanding the regulatory framework and aligning with Hemas' product strategy will enhance the potential for a successful partnership.
Frequently Asked Questions — Hemas Pharmaceuticals (pvt) Ltd
What products does Hemas Pharmaceuticals (pvt) Ltd import from India?
Hemas Pharmaceuticals (pvt) Ltd imports 4 pharmaceutical products across 4 categories. Top imports: Vitamin ($1.3M), Methotrexate ($435.1K), Letrozole ($277.4K), Bisacodyl ($122.6K).
Who supplies pharmaceuticals to Hemas Pharmaceuticals (pvt) Ltd from India?
Hemas Pharmaceuticals (pvt) Ltd sources from 8 verified Indian suppliers. The primary supplier is Usv Private Limited (56.1% of imports, $1.7M).
What is Hemas Pharmaceuticals (pvt) Ltd's total pharmaceutical import value?
Hemas Pharmaceuticals (pvt) Ltd's total pharmaceutical import value from India is $2.1M, based on 59 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Hemas Pharmaceuticals (pvt) Ltd focus on?
Hemas Pharmaceuticals (pvt) Ltd imports across 4 categories. The largest: Nutritional Supplements (59.9%), Immunosuppressants (20.9%), Advanced Oncology (13.3%).
Get Full Hemas Pharmaceuticals (pvt) Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Hemas Pharmaceuticals (pvt) Ltd identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Hemas Pharmaceuticals (pvt) Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 59 individual customs records matching Hemas Pharmaceuticals (pvt) Ltd.
- 5.Supplier Verification: Hemas Pharmaceuticals (pvt) Ltd sources from 8 verified Indian suppliers across 185 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.