Hawkary Pharmaceuitcal CO.
Pharmaceutical Importer · Iraq · Advanced Antibiotics Focus · $3.4M Total Trade · DGFT Verified
Hawkary Pharmaceuitcal CO. is a pharmaceutical importer based in Iraq with a total trade value of $3.4M across 5 products in 3 therapeutic categories. Based on 79 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Hawkary Pharmaceuitcal CO. sources from 1 verified Indian supplier, with Ajanta Pharma Limited accounting for 100.0% of imports.
Hawkary Pharmaceuitcal CO. — Import Portfolio & Supplier Network
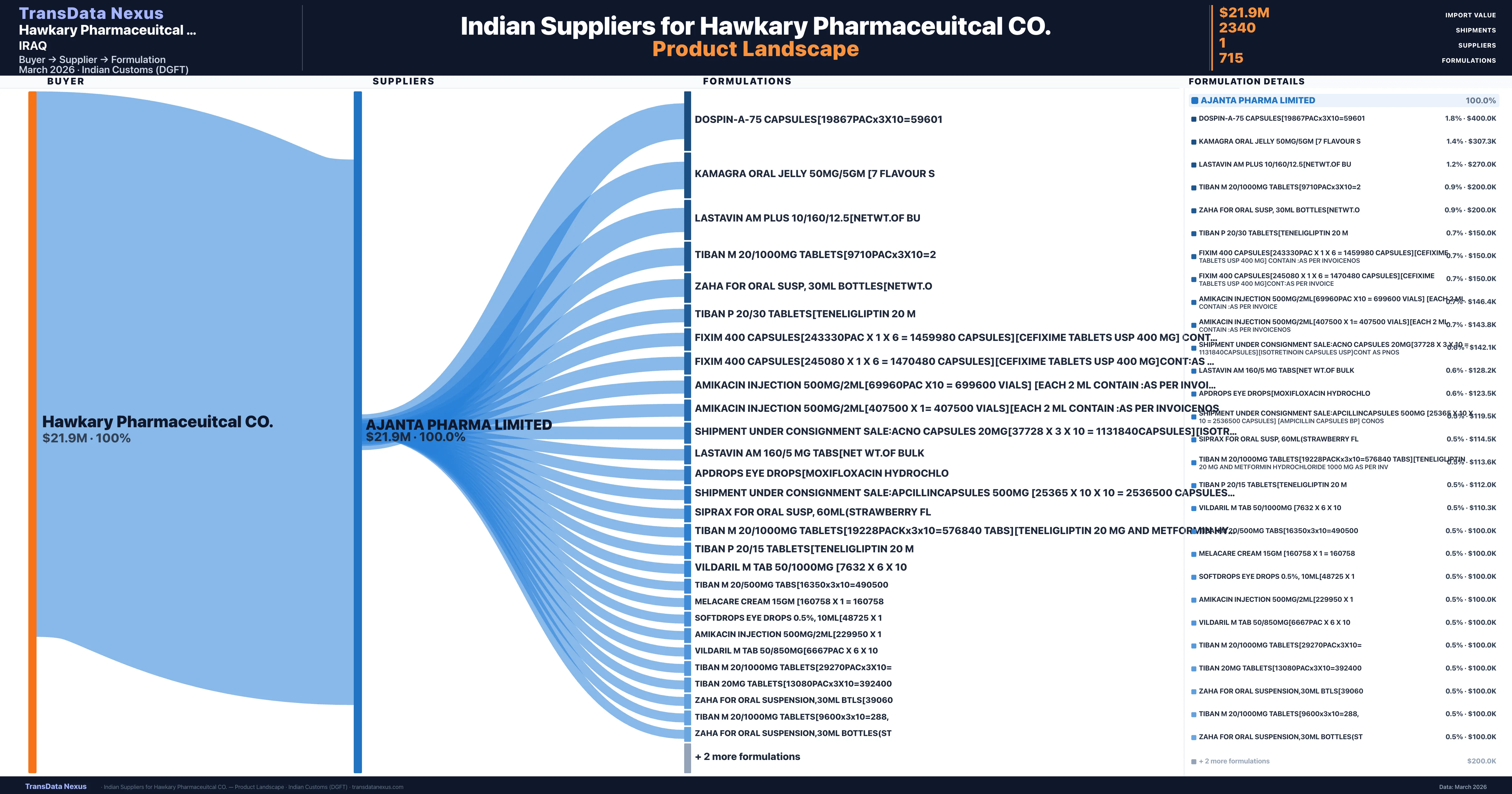
Who Are the Verified Indian Suppliers to Hawkary Pharmaceuitcal CO.?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Ajanta Pharma Limited | $21.9M | 2,340 | 100.0% |
Hawkary Pharmaceuitcal CO. sources from 1 verified Indian supplier across 715 distinct formulations. The sourcing is highly concentrated — Ajanta Pharma Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Hawkary Pharmaceuitcal CO. Import?
| Formulation | Value | Ships |
|---|---|---|
| Dospin-a-75 capsules[19867pacx3x10=59601 | $400.0K | 8 |
| Kamagra oral jelly 50MG/5GM [7 flavour | $307.3K | 7 |
| Lastavin am plus 10/160/12.5[netwt.of | $270.0K | 6 |
| Tiban m 20/1000MG tablets[9710pacx3x10=2 | $200.0K | 4 |
| Zaha for oral SUSP, 30ML | $200.0K | 4 |
| Tiban p 20/30 tablets[teneligliptin 20 | $150.0K | 3 |
| Fixim 400 capsules[243330pac x 1 x 6 = 1459980 capsules][cefixime tablets USP 400 MG] contain :as per | $150.0K | 3 |
| Fixim 400 capsules[245080 x 1 x 6 = 1470480 capsules][cefixime tablets USP 400 MG]cont:as per | $150.0K | 3 |
| Amikacin injection 500MG/2ML[69960pac x10 = 699600 vials] [Each 2 ML contain :as per | $146.4K | 3 |
| Amikacin injection 500MG/2ML[407500 x 1= 407500 vials][Each 2 ML contain :as per | $143.8K | 3 |
| Shipment under consignment sale:acno capsules 20MG[37728 x 3 x 10 = 1131840capsules][isotretinoin capsules USP]cont as | $142.1K | 3 |
| Lastavin am 160/5 MG tabs[net wt.of | $128.2K | 3 |
| Apdrops eye drops[moxifloxacin | $123.5K | 7 |
| Shipment under consignment sale:apcillincapsules 500MG [25365 x 10 x 10 = 2536500 capsules] [ampicillin capsules BP] | $119.5K | 3 |
| Siprax for oral SUSP, 60ML(strawberry | $114.5K | 13 |
Hawkary Pharmaceuitcal CO. imports 715 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Hawkary Pharmaceuitcal CO. Import?
Top Products by Import Value
Hawkary Pharmaceuitcal CO. Therapeutic Categories — 3 Specializations
Hawkary Pharmaceuitcal CO. imports across 3 therapeutic categories, with Advanced Antibiotics (54.2%), Antibiotics (34.0%), Analgesics & Antipyretics (11.8%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
3 products · 54.2% · $1.8M
Antibiotics
1 products · 34.0% · $1.1M
Analgesics & Antipyretics
1 products · 11.8% · $400.0K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Amikacin | Advanced Antibiotics | $1.4M | 28 | 3.6% | 5 |
| 2 | Azithromycin | Antibiotics | $1.1M | 23 | 0.6% | 17 |
| 3 | Aspirin | Analgesics & Antipyretics | $400.0K | 8 | 1.1% | 12 |
| 4 | Moxifloxacin | Advanced Antibiotics | $335.4K | 18 | 1.0% | 14 |
| 5 | Gatifloxacin | Advanced Antibiotics | $100.0K | 2 | 12.5% | 2 |
Hawkary Pharmaceuitcal CO. imports 5 pharmaceutical products across 3 categories into Iraq totaling $3.4M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Hawkary Pharmaceuitcal CO..
Request DemoHawkary Pharmaceuitcal CO. — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Hawkary Pharmaceutical Co. is a leading pharmaceutical distribution company based in Erbil, Iraq. Established in 1992, it has rapidly ascended to become one of the top five fastest-growing local pharmaceutical distributors in the country, securing a significant market share. The company specializes in the distribution and marketing of pharmaceutical products, continually expanding its portfolio to meet the evolving needs of the Iraqi pharmaceutical market.
Under the leadership of Dr. Muhammed Ismail Seyan, who serves as the Chairman and Acting CEO, Hawkary has implemented advanced systems such as Enterprise Resource Planning (ERP) to enhance operational efficiency. This integration facilitates seamless information flow across business functions and regional offices, ensuring timely delivery of quality products.
2Distribution Network
Hawkary's distribution network spans the entire Iraqi territory, encompassing major cities such as Erbil, Baghdad, Basra, Karbala, Tikrit, Sulaymaniyah, Duhok, Mosul, and Kirkuk. The company operates a substantial warehouse with a capacity of 25,000 pallets, adhering to European and U.S. FDA guidelines for Good Distribution Practice (GDP). This commitment ensures the maintenance of high-quality standards in the storage and distribution of medical products.
3Industry Role
In Iraq's pharmaceutical supply chain, Hawkary Pharmaceutical Co. functions primarily as a wholesaler and distributor. By importing finished pharmaceutical formulations from international suppliers, the company plays a pivotal role in ensuring the availability of a diverse range of medications across the nation. Its extensive distribution network and adherence to international standards underscore its significant position in the market.
Supplier Relationship Intelligence — Hawkary Pharmaceuitcal CO.
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Hawkary's sourcing strategy exhibits a high degree of concentration, with a total import value of $3.4 million USD from India over 79 shipments, all sourced from a single supplier, AJANTA PHARMA LIMITED. This exclusive partnership indicates a strategic choice to maintain a consistent supply chain and potentially negotiate favorable terms. However, such dependency also poses risks related to supply chain disruptions, pricing fluctuations, and regulatory changes affecting the sole supplier. The absence of alternative suppliers may limit Hawkary's ability to diversify its product offerings and mitigate potential supply chain vulnerabilities.
2Supply Chain Resilience
The resilience of Hawkary's Indian supply chain is contingent upon the stability and reliability of AJANTA PHARMA LIMITED. The lack of backup suppliers and limited formulation diversity within the current sourcing strategy heightens the company's exposure to potential disruptions. Factors such as geopolitical tensions, regulatory changes, or operational challenges within the supplier's facilities could adversely impact Hawkary's ability to meet market demand. Additionally, the reliance on a single supplier may affect the company's capacity to adapt to evolving market needs and introduce new products.
3Strategic Implications
Hawkary's concentrated sourcing pattern positions the company to leverage strong relationships with its sole supplier, potentially securing favorable pricing and consistent product quality. However, this strategy also exposes Hawkary to significant risks associated with supply chain disruptions and regulatory changes affecting the supplier. For Indian exporters seeking to become alternative suppliers, this presents an opportunity to diversify Hawkary's sourcing base, thereby enhancing supply chain resilience and reducing dependency on a single source. Engaging with Hawkary could lead to a mutually beneficial partnership, offering Indian exporters access to the Iraqi market while providing Hawkary with a more diversified and robust supply chain.
Importing Pharmaceuticals into Iraq — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Iraq
1Regulatory Authority & Framework
In Iraq, the Ministry of Health (MOH) through the Iraq Drug Regulatory Authority (IDRA) oversees the regulation of pharmaceutical imports. The IDRA is responsible for ensuring the safety, efficacy, and quality of pharmaceutical products entering the Iraqi market. This includes the approval of marketing authorizations, pricing oversight, pharmacovigilance, inspections, import permissions, and promotion rules. Quality and standards are further regulated by the Central Organization for Standardization and Quality Control (COSQC), which operates under the Ministry of Planning.
2Import Licensing & GMP
Pharmaceutical products imported into Iraq must be registered with the IDRA, which requires evidence of Good Manufacturing Practice (GMP) compliance. The registration process involves submitting documentation that demonstrates the safety, efficacy, and quality of the products. Marketing authorizations are typically time-limited, commonly for five years, and are renewable with updated data. Generics follow an abbreviated, bioequivalence-driven route, while biologics and biosimilars require comparability packages. Promotion of pharmaceutical products is tightly controlled, with strict rules on interactions with healthcare professionals.
3Quality & Labeling
Imported pharmaceutical products must comply with Iraq's National Drug System, which mandates comprehensive registration and tracking of all pharmaceutical products and manufacturers within a centralized digital platform. Each product is required to display an electronic label featuring essential drug information, official pricing, and a unique QR code for instant verification. This labeling system provides immediate access to critical product data, including drug names, expiry dates, manufacturer details, active ingredients, side effects, warnings, and precautions.
4Recent Regulatory Changes
In January 2023, the Iraqi government issued a decision to reduce the import of 23 types of medicines that are produced locally and fulfill the need. This policy aims to encourage domestic production and reduce reliance on imports for certain pharmaceutical products. The decision underscores the government's commitment to enhancing local pharmaceutical manufacturing capabilities and ensuring the availability of essential medicines within the country. (ina.iq)
Hawkary Pharmaceuitcal CO. — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Hawkary Pharmaceutical Co. focuses on importing a diverse range of pharmaceutical products, with a notable emphasis on advanced antibiotics, antibiotics, and analgesics & antipyretics. This strategic focus aligns with the therapeutic needs of the Iraqi population, addressing prevalent health concerns and conditions. The company's product portfolio is designed to meet the growing demand for effective treatments in these categories, ensuring accessibility to essential medications.
2Sourcing Profile
Hawkary's sourcing strategy is centered on importing finished pharmaceutical formulations from a single supplier, AJANTA PHARMA LIMITED in India. This approach allows for streamlined operations and potentially favorable terms. However, the lack of diversification in sourcing may limit the company's ability to adapt to changing market demands and introduce new products. Exploring additional suppliers could enhance the diversity and resilience of Hawkary's product offerings.
3Market Positioning
Based on its product mix, Hawkary Pharmaceutical Co. serves a broad segment of the Iraqi market, including retail pharmacies, hospitals, government tenders, and wholesale distribution. The company's extensive distribution network and comprehensive product portfolio enable it to meet the diverse needs of healthcare providers and patients across the country.
Seller's Guide — How to Become a Supplier to Hawkary Pharmaceuitcal CO.
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Hawkary Pharmaceutical Co. by offering a diversified range of pharmaceutical products. By introducing alternative sources and formulations, Indian exporters can help mitigate Hawkary's current sourcing concentration, thereby enhancing supply chain resilience and reducing dependency on a single supplier. This collaboration could lead to a mutually beneficial partnership, expanding market reach and product offerings.
2Requirements & Qualifications
Indian exporters seeking to supply Hawkary Pharmaceutical Co. must ensure that their products are registered with the Iraq Drug Regulatory Authority (IDRA) and comply with Good Manufacturing Practice (GMP) standards. Products must be accompanied by electronic labeling featuring essential drug information and a unique QR code for verification. Additionally, exporters should be prepared to navigate Iraq's regulatory framework, including import licensing requirements and quality control standards.
3How to Approach
To establish a relationship with Hawkary Pharmaceutical Co., Indian exporters should initiate contact through formal channels, presenting their product portfolios and demonstrating compliance with Iraq's regulatory requirements. Participating in tenders and engaging in direct negotiations can facilitate partnership opportunities. Understanding the regulatory filing process and adhering to the necessary timelines are crucial for successful market entry. Building a strong relationship with Hawkary can lead to increased market presence and access to a significant distribution network within Iraq.
Frequently Asked Questions — Hawkary Pharmaceuitcal CO.
What products does Hawkary Pharmaceuitcal CO. import from India?
Hawkary Pharmaceuitcal CO. imports 5 pharmaceutical products across 3 categories. Top imports: Amikacin ($1.4M), Azithromycin ($1.1M), Aspirin ($400.0K), Moxifloxacin ($335.4K), Gatifloxacin ($100.0K).
Who supplies pharmaceuticals to Hawkary Pharmaceuitcal CO. from India?
Hawkary Pharmaceuitcal CO. sources from 1 verified Indian suppliers. The primary supplier is Ajanta Pharma Limited (100.0% of imports, $21.9M).
What is Hawkary Pharmaceuitcal CO.'s total pharmaceutical import value?
Hawkary Pharmaceuitcal CO.'s total pharmaceutical import value from India is $3.4M, based on 79 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Hawkary Pharmaceuitcal CO. focus on?
Hawkary Pharmaceuitcal CO. imports across 3 categories. The largest: Advanced Antibiotics (54.2%), Antibiotics (34.0%), Analgesics & Antipyretics (11.8%).
Get Full Hawkary Pharmaceuitcal CO. Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Hawkary Pharmaceuitcal CO. identified across shipments using consignee name normalization, aggregating 3 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Hawkary Pharmaceuitcal CO.'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 79 individual customs records matching Hawkary Pharmaceuitcal CO..
- 5.Supplier Verification: Hawkary Pharmaceuitcal CO. sources from 1 verified Indian suppliers across 715 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 3 company name variants from customs records. For current shipment-level data, contact TransData Nexus.