H.essers & Zonen International Tran
Pharmaceutical Importer · Belgium · Antihistamines & Allergy Focus · $7.4M Total Trade · DGFT Verified
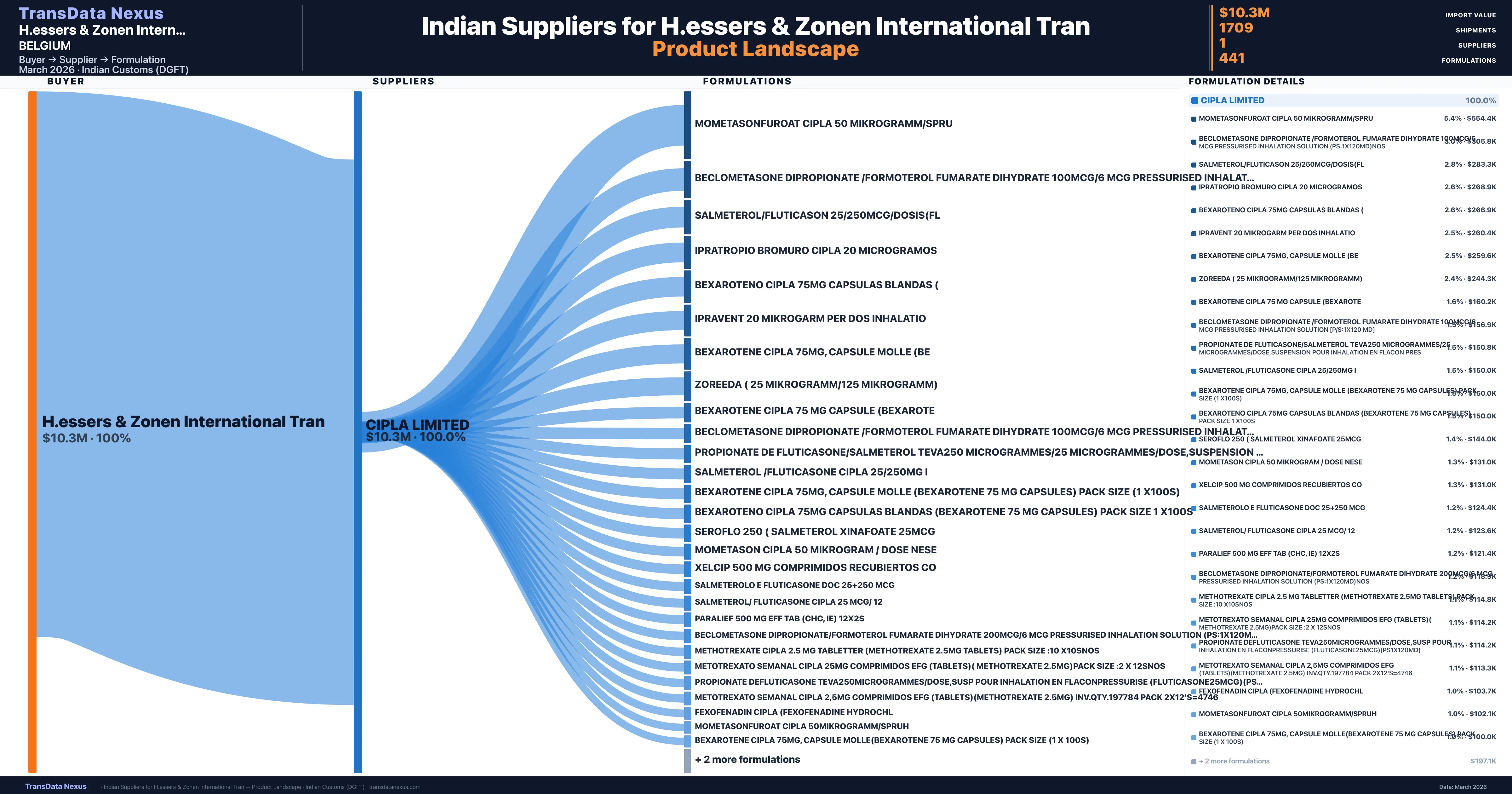
H.essers & Zonen International Tran is a pharmaceutical importer based in Belgium with a total trade value of $7.4M across 4 products in 3 therapeutic categories. Based on 413 verified import shipments from Indian Customs (DGFT) records, H.essers & Zonen International Tran is the #1 buyer in 1 product including Imatinib. H.essers & Zonen International Tran sources from 1 verified Indian supplier, with Cipla Limited accounting for 100.0% of imports.
H.essers & Zonen International Tran — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to H.essers & Zonen International Tran?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Cipla Limited | $10.3M | 1,709 | 100.0% |
H.essers & Zonen International Tran sources from 1 verified Indian supplier across 441 distinct formulations. The sourcing is highly concentrated — Cipla Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does H.essers & Zonen International Tran Import?
| Formulation | Value | Ships |
|---|---|---|
| Mometasonfuroat cipla 50 | $554.4K | 42 |
| Beclometasone dipropionate /formoterol fumarate dihydrate 100mcg/6 MCG pressurised inhalation solution (ps:1x120md | $305.8K | 24 |
| Salmeterol/fluticason | $283.3K | 14 |
| Ipratropio bromuro cipla 20 | $268.9K | 12 |
| Bexaroteno cipla 75MG capsulas blandas | $266.9K | 15 |
| Ipravent 20 mikrogarm per dos | $260.4K | 36 |
| Bexarotene cipla 75MG, capsule molle | $259.6K | 15 |
| Zoreeda ( 25 mikrogramm/125 mikrogramm) | $244.3K | 24 |
| Bexarotene cipla 75 MG capsule | $160.2K | 9 |
| Beclometasone dipropionate /formoterol fumarate dihydrate 100mcg/6 MCG pressurised inhalation solution [p/s:1x120 | $156.9K | 15 |
| Propionate de fluticasone/salmeterol teva250 microgrammes/25 microgrammes/dose,suspension pour inhalation en flacon | $150.8K | 5 |
| Salmeterol /fluticasone cipla 25/250MG | $150.0K | 24 |
| Bexarotene cipla 75MG, capsule molle (bexarotene 75 MG capsules) Pack size (1 x100s) | $150.0K | 3 |
| Bexaroteno cipla 75MG capsulas blandas (bexarotene 75 MG capsules) Pack size 1 x100s | $150.0K | 3 |
| Seroflo 250 ( salmeterol xinafoate 25mcg | $144.0K | 10 |
H.essers & Zonen International Tran imports 441 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does H.essers & Zonen International Tran Import?
Top Products by Import Value
H.essers & Zonen International Tran Therapeutic Categories — 3 Specializations
H.essers & Zonen International Tran imports across 3 therapeutic categories, with Antihistamines & Allergy (57.2%), Respiratory (38.8%), Advanced Oncology (4.1%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antihistamines & Allergy
1 products · 57.2% · $4.2M
Respiratory
2 products · 38.8% · $2.9M
Advanced Oncology
1 products · 4.1% · $300.0K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Fexofenadine | Antihistamines & Allergy | $4.2M | 166 | 3.9% | 7 |
| 2 | Salmeterol | Respiratory | $1.8M | 137 | 2.5% | 9 |
| 3 | Formoterol | Respiratory | $1.0M | 104 | 1.8% | 12 |
| 4 | Imatinib | Advanced Oncology | $300.0K | 6 | 1.0% | 1 |
H.essers & Zonen International Tran imports 4 pharmaceutical products across 3 categories into Belgium totaling $7.4M. The company is the #1 buyer for 1 product: Imatinib.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for H.essers & Zonen International Tran.
Request DemoH.essers & Zonen International Tran — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
H.essers & Zonen International Transport NV, established on January 1, 1968, is a prominent Belgian company specializing in international transport and logistics services. Headquartered in Genk, Belgium, the company operates as a subsidiary of the H.Essers Group, a leading European logistics provider. H.Essers Group offers comprehensive supply chain solutions, including warehousing, transport, and value-added services, with a strong emphasis on sectors such as chemicals and high-value goods.
Within the pharmaceutical sector, H.essers & Zonen International Transport NV plays a pivotal role in the distribution of finished pharmaceutical formulations. The company imports a diverse range of pharmaceutical products from India, serving as a key player in Belgium's pharmaceutical supply chain. Their operations encompass the importation, storage, and distribution of various pharmaceutical products, ensuring compliance with stringent regulatory standards and maintaining the integrity of the supply chain.
2Distribution Network
H.essers & Zonen International Transport NV leverages the extensive infrastructure of the H.Essers Group, which boasts 68 branches and 10 local operations across 15 countries. This expansive network enables the company to offer personalized and integrated logistics solutions, including warehousing and transport services, tailored to the specific needs of the pharmaceutical industry.
The company's logistics capabilities are further enhanced by its own fleet of 1,497 trucks and over 1,300,000 square meters of warehouse space. These resources facilitate efficient and secure distribution of pharmaceutical products both within Belgium and across Europe. H.essers & Zonen International Transport NV's strategic locations and robust logistics infrastructure ensure timely and reliable delivery of pharmaceutical goods to various stakeholders in the healthcare sector.
3Industry Role
H.essers & Zonen International Transport NV functions primarily as a logistics intermediary within Belgium's pharmaceutical supply chain. While the company does not hold a wholesale distribution authorization, it plays a crucial role in the importation, storage, and distribution of pharmaceutical products. By partnering with authorized wholesalers and pharmaceutical companies, H.essers & Zonen International Transport NV ensures that imported pharmaceutical formulations are efficiently and securely delivered to the Belgian market.
The company's operations are characterized by a strong commitment to compliance with Good Distribution Practice (GDP) and Good Manufacturing Practice (GMP) standards. This adherence guarantees the quality and safety of pharmaceutical products throughout the supply chain, from importation to final delivery. H.essers & Zonen International Transport NV's role as a logistics intermediary is integral to maintaining the integrity and reliability of Belgium's pharmaceutical distribution network.
Supplier Relationship Intelligence — H.essers & Zonen International Tran
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
H.essers & Zonen International Transport NV demonstrates a high degree of sourcing concentration, importing 100% of its pharmaceutical products from a single supplier, Cipla Limited. This exclusive sourcing strategy indicates a deep and strategic partnership between the two entities, likely fostering favorable terms and streamlined logistics processes.
However, this single-source dependency also introduces potential risks, such as supply chain disruptions due to unforeseen events affecting Cipla Limited. The absence of alternative suppliers could lead to challenges in maintaining a consistent supply of pharmaceutical products. To mitigate such risks, it would be prudent for H.essers & Zonen International Transport NV to consider diversifying its supplier base, thereby enhancing the resilience and flexibility of its supply chain.
The shipment data reveals a consistent and stable relationship between H.essers & Zonen International Transport NV and Cipla Limited, with a total of 1,709 shipments recorded. This stability suggests effective collaboration and mutual trust, which are essential for maintaining uninterrupted supply chain operations. Nonetheless, exploring additional supplier partnerships could further strengthen the company's position in the market and reduce potential vulnerabilities associated with sole sourcing.
2Supply Chain Resilience
The resilience of H.essers & Zonen International Transport NV's supply chain is closely tied to its exclusive partnership with Cipla Limited. While this relationship ensures a steady supply of pharmaceutical products, it also means that the company's supply chain is highly dependent on the operational stability and reliability of Cipla Limited.
Currently, there is no evidence of backup suppliers in place to mitigate potential disruptions. The lack of supplier diversification could pose challenges in the event of unforeseen circumstances affecting Cipla Limited's production or distribution capabilities. To enhance supply chain resilience, H.essers & Zonen International Transport NV should consider establishing relationships with additional suppliers, thereby reducing the impact of potential disruptions and ensuring a more robust and flexible supply chain.
Regarding formulation diversity, the company imports a total of 441 unique pharmaceutical formulations. This extensive range indicates a broad portfolio, which can help mitigate risks associated with dependency on a single product line. However, the lack of supplier diversification means that any supply chain disruptions could still impact the availability of these diverse formulations. Therefore, diversifying the supplier base remains a critical strategy for enhancing overall supply chain resilience.
3Strategic Implications
H.essers & Zonen International Transport NV's sourcing pattern, characterized by exclusive reliance on Cipla Limited, positions the company to benefit from strong supplier relationships and potentially favorable terms. This strategic choice may lead to cost efficiencies and streamlined logistics processes, enhancing the company's competitive position in the Belgian pharmaceutical market.
However, the single-source dependency also exposes the company to risks associated with supply chain disruptions. To maintain a competitive edge and ensure uninterrupted service to the Belgian market, it is advisable for H.essers & Zonen International Transport NV to explore partnerships with additional suppliers. This approach would not only mitigate potential risks but also provide access to a broader range of products and innovations, further strengthening the company's market position.
Importing Pharmaceuticals into Belgium — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Belgium
1Regulatory Authority & Framework
In Belgium, the Federal Agency for Medicines and Health Products (FAMHP) serves as the primary regulatory authority overseeing the importation and distribution of pharmaceutical products. The FAMHP ensures that all medicinal products meet the required safety, efficacy, and quality standards before they are authorized for sale in the Belgian market.
The legal framework governing pharmaceutical imports is primarily established by the Royal Decree of 14 December 2006 on medicinal products for human use. This decree outlines the procedures for obtaining marketing authorizations, import licenses, and the conditions under which unlicensed medicines can be imported to address special needs or critical unavailability.
For Indian pharmaceutical exporters, understanding and complying with the FAMHP's regulations is essential for successful market entry. This includes obtaining the necessary marketing authorizations and import licenses, as well as adhering to the specific requirements for parallel imports and distribution. Staying informed about regulatory updates and maintaining open communication with the FAMHP can facilitate smoother import processes and ensure compliance with Belgian pharmaceutical laws.
2Import Licensing & GMP
Import licensing in Belgium is governed by the FAMHP, which requires wholesalers to obtain an import license to bring medicinal products into the country. This license ensures that the importer meets the necessary standards for handling and distributing pharmaceutical products. Additionally, wholesalers must possess a valid wholesale distribution authorization, which is granted by the FAMHP upon demonstrating compliance with Good Distribution Practice (GDP) and Good Manufacturing Practice (GMP) standards.
For Indian pharmaceutical exporters, it is crucial to ensure that their products are manufactured in facilities that comply with EU GMP standards. Recognition of WHO GMP or PIC/S certifications can also be beneficial, as these are often accepted by the FAMHP. Ensuring that products meet these standards is essential for obtaining the necessary import licenses and for the successful distribution of pharmaceutical products in Belgium.
3Quality & Labeling
Belgian regulations mandate that imported pharmaceutical products undergo batch testing to verify their quality, safety, and efficacy. This testing ensures that products meet the required standards before they are distributed within the Belgian market. Additionally, products must adhere to stability requirements to ensure their effectiveness throughout their shelf life.
Labeling requirements stipulate that all information on pharmaceutical products be provided in Dutch and French, the official languages of Belgium. This includes product names, usage instructions, and any necessary warnings or precautions. Serialization mandates are also in place to enhance traceability and prevent counterfeit products from entering the supply chain. Indian exporters must ensure that their products comply with these labeling and serialization requirements to facilitate smooth importation and distribution in Belgium.
4Recent Regulatory Changes
In September 2022, Belgium implemented a Royal Decree amending the Royal Decree of 19 April 2001 on the parallel import of medicines for human use. This amendment removed the condition that the medicinal product for which a parallel import license is sought must have "the same therapeutic indications" as the Belgian reference medicinal product. This change simplifies the process for obtaining parallel import licenses, potentially increasing the availability of imported medicines in Belgium.
Another significant regulatory change occurred in November 2024, with the entry into force of Article 105/1 of the Royal Decree of 14 December 2006. This provision allows wholesalers to import
Frequently Asked Questions — H.essers & Zonen International Tran
What products does H.essers & Zonen International Tran import from India?
H.essers & Zonen International Tran imports 4 pharmaceutical products across 3 categories. Top imports: Fexofenadine ($4.2M), Salmeterol ($1.8M), Formoterol ($1.0M), Imatinib ($300.0K).
Who supplies pharmaceuticals to H.essers & Zonen International Tran from India?
H.essers & Zonen International Tran sources from 1 verified Indian suppliers. The primary supplier is Cipla Limited (100.0% of imports, $10.3M).
What is H.essers & Zonen International Tran's total pharmaceutical import value?
H.essers & Zonen International Tran's total pharmaceutical import value from India is $7.4M, based on 413 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does H.essers & Zonen International Tran focus on?
H.essers & Zonen International Tran imports across 3 categories. The largest: Antihistamines & Allergy (57.2%), Respiratory (38.8%), Advanced Oncology (4.1%).
Get Full H.essers & Zonen International Tran Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: H.essers & Zonen International Tran identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as H.essers & Zonen International Tran's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 413 individual customs records matching H.essers & Zonen International Tran.
- 5.Supplier Verification: H.essers & Zonen International Tran sources from 1 verified Indian suppliers across 441 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.