Ethiopian Pharmaceuticals Supply
Pharmaceutical Importer · Ethiopia · Advanced Antibiotics Focus · $3.1M Total Trade · DGFT Verified
Ethiopian Pharmaceuticals Supply is a pharmaceutical importer based in Ethiopia with a total trade value of $3.1M across 6 products in 3 therapeutic categories. Based on 67 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Ethiopian Pharmaceuticals Supply sources from 34 verified Indian suppliers, with Fresenius Kabi India Private Limited accounting for 21.0% of imports.
Ethiopian Pharmaceuticals Supply — Import Portfolio & Supplier Network
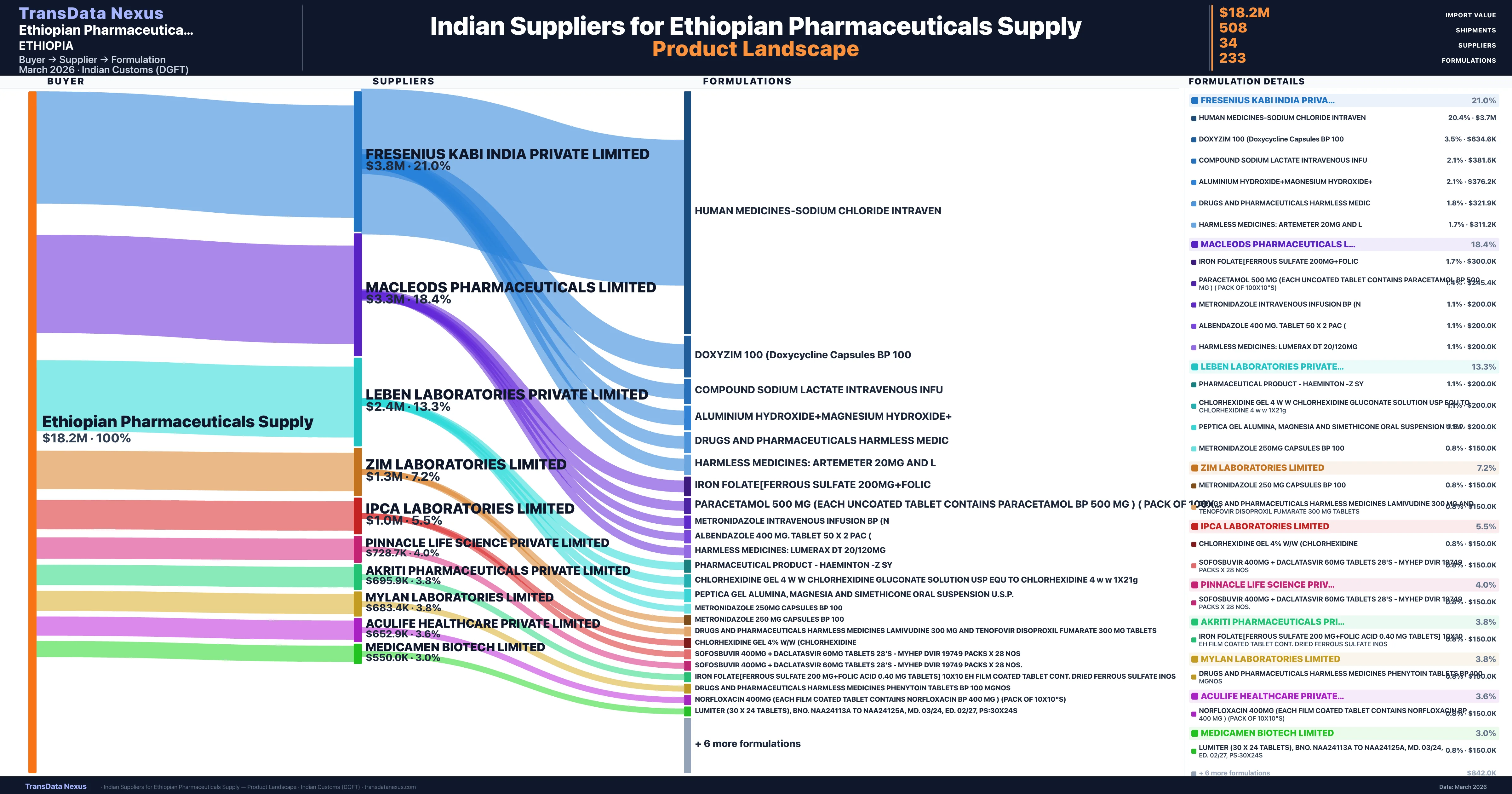
Who Are the Verified Indian Suppliers to Ethiopian Pharmaceuticals Supply?
Customs-verified supplier relationships from Indian DGFT records
Ethiopian Pharmaceuticals Supply sources from 34 verified Indian suppliers across 233 distinct formulations. The supply base is diversified across 34 suppliers, reducing single-source dependency risk.
What Formulations Does Ethiopian Pharmaceuticals Supply Import?
| Formulation | Value | Ships |
|---|---|---|
| Human medicines-sodium chloride | $3.7M | 81 |
| Doxyzim 100 (doxycycline capsules BP | $634.6K | 13 |
| Compound sodium lactate intravenous | $381.5K | 8 |
| Aluminium hydroxide+magnesium | $376.2K | 10 |
| Drugs and pharmaceuticals harmless | $321.9K | 8 |
| Harmless medicines: artemeter 20MG and | $311.2K | 7 |
| Iron folate[ferrous sulfate | $300.0K | 6 |
| Paracetamol 500 MG (Each uncoated tablet contains paracetamol BP 500 MG ) ( Pack of 100x10"s) | $245.4K | 6 |
| Metronidazole intravenous infusion BP | $200.0K | 4 |
| Albendazole 400 MG. tablet 50 x 2 pac | $200.0K | 4 |
| Harmless medicines: lumerax dt 20/120MG | $200.0K | 4 |
| Pharmaceutical product - haeminton -z | $200.0K | 4 |
| Chlorhexidine gel 4 w w chlorhexidine gluconate solution USP equ to chlorhexidine 4 w w | $200.0K | 4 |
| Peptica gel alumina, magnesia and simethicone oral suspension u.s.p. | $200.0K | 4 |
| Metronidazole 250MG capsules BP | $150.0K | 3 |
Ethiopian Pharmaceuticals Supply imports 233 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Ethiopian Pharmaceuticals Supply Import?
Ethiopian Pharmaceuticals Supply Therapeutic Categories — 3 Specializations
Ethiopian Pharmaceuticals Supply imports across 3 therapeutic categories, with Advanced Antibiotics (54.8%), Antimalarial & Antiparasitic (40.4%), Antibiotics (4.8%) representing the largest segments. The portfolio is concentrated — top 5 products = 95% of total imports.
Advanced Antibiotics
3 products · 54.8% · $1.7M
Antimalarial & Antiparasitic
2 products · 40.4% · $1.3M
Antibiotics
1 products · 4.8% · $150.0K
Import Portfolio — Top 6 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Artemether | Antimalarial & Antiparasitic | $900.0K | 18 | 0.1% | 8 |
| 2 | Trimethoprim | Advanced Antibiotics | $844.4K | 22 | 1.3% | 7 |
| 3 | Metronidazole | Advanced Antibiotics | $650.0K | 13 | 0.3% | 10 |
| 4 | Albendazole | Antimalarial & Antiparasitic | $350.0K | 7 | 0.5% | 8 |
| 5 | Norfloxacin | Advanced Antibiotics | $200.0K | 4 | 2.4% | 2 |
| 6 | Tetracycline | Antibiotics | $150.0K | 3 | 0.9% | 5 |
Ethiopian Pharmaceuticals Supply imports 6 pharmaceutical products across 3 categories into Ethiopia totaling $3.1M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Ethiopian Pharmaceuticals Supply.
Request DemoEthiopian Pharmaceuticals Supply — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Ethiopian Pharmaceuticals Supply Agency (EPSA) is a government-established entity under the Ministry of Health of the Federal Democratic Republic of Ethiopia. Established in September 2007 by Proclamation No. 553/2007, EPSA's primary mission is to ensure the uninterrupted supply of quality-assured essential pharmaceuticals to public health institutions at affordable prices. (moh.gov.et)
Headquartered in Addis Ababa, EPSA operates a centralized procurement and distribution system, coordinating activities across nineteen branches and seven clusters. This structure enables the agency to supply pharmaceuticals, medical supplies, medical equipment, and laboratory reagents to over 4,000 medical facilities nationwide. (epss.gov.et)
As a government agency, EPSA plays a pivotal role in Ethiopia's pharmaceutical distribution network, acting as the primary wholesaler and distributor for public health institutions. Its operations are integral to the national health system, ensuring that essential medicines are accessible and affordable to the Ethiopian population.
2Distribution Network
EPSA's distribution network is extensive, encompassing nineteen branches and seven clusters strategically located across Ethiopia. This widespread presence ensures that pharmaceuticals and medical supplies reach over 4,000 health facilities, including hospitals, clinics, and health centers, throughout the country. (epss.gov.et)
The agency's logistics capabilities are robust, featuring modern storage facilities equipped with advanced inventory management systems. These facilities are designed to maintain the quality and efficacy of pharmaceuticals, ensuring an uninterrupted supply chain. EPSA's transportation infrastructure is optimized for efficiency, facilitating timely deliveries to meet the demands of health institutions across Ethiopia.
3Industry Role
EPSA serves as the central wholesaler and distributor within Ethiopia's pharmaceutical supply chain. By procuring and distributing essential medicines, medical supplies, and equipment, the agency ensures that public health institutions have access to the necessary resources to provide quality healthcare services. Its operations are critical to the functioning of the national health system, supporting the delivery of healthcare services to the Ethiopian population.
Supplier Relationship Intelligence — Ethiopian Pharmaceuticals Supply
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
EPSA's sourcing strategy involves a diversified network of suppliers, including both local and international manufacturers. This approach mitigates the risks associated with single-source dependency, ensuring a stable and continuous supply of pharmaceuticals. The agency's procurement system emphasizes quality assurance and cost-effectiveness, aligning with its mission to provide affordable and quality-assured essential medicines to the public. (moh.gov.et)
2Supply Chain Resilience
EPSA's supply chain is designed for resilience, incorporating multiple suppliers to prevent disruptions. The agency's modern storage facilities and efficient transportation systems further enhance its ability to maintain an uninterrupted supply of pharmaceuticals. By establishing a robust supply chain, EPSA ensures that health institutions across Ethiopia have continuous access to essential medicines, even in the face of potential challenges.
3Strategic Implications
EPSA's diversified sourcing strategy strengthens its competitive position by reducing dependency on a single supplier and enhancing supply chain stability. For Indian pharmaceutical exporters, this approach presents opportunities to engage with EPSA by meeting the agency's quality standards and cost-effectiveness criteria. Establishing partnerships with EPSA can lead to increased market access and contribute to the agency's mission of providing affordable and quality-assured essential medicines to the Ethiopian public.
Importing Pharmaceuticals into Ethiopia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Ethiopia
1Regulatory Authority & Framework
The Ethiopian Food and Drug Authority (EFDA) is the primary regulatory body overseeing the pharmaceutical sector in Ethiopia. EFDA is responsible for ensuring the safety, efficacy, and quality of pharmaceuticals and medical supplies entering the Ethiopian market. Key legislation governing pharmaceutical imports includes the Proclamation No. 553/2007, which established EPSA, and various regulations set forth by EFDA. The marketing authorization pathway for Indian generics involves obtaining approval from EFDA, which includes submitting comprehensive documentation on product quality, safety, and efficacy.
2Import Licensing & GMP
Import licensing requirements for pharmaceuticals in Ethiopia are stringent, necessitating that all imported medicines and medical supplies be registered with EFDA. Manufacturers must provide evidence of Good Manufacturing Practice (GMP) certification, such as EU GMP, WHO GMP, or PIC/S, to demonstrate compliance with international quality standards. Additionally, importers must obtain wholesale distribution authorization from EFDA, ensuring that they adhere to the regulatory standards set for pharmaceutical distribution within the country.
3Quality & Labeling
Imported pharmaceuticals must undergo batch testing to confirm their quality and safety before distribution. Stability requirements are enforced to ensure that medicines maintain their efficacy throughout their shelf life. Labeling must be in the official language(s) of Ethiopia, providing clear information on dosage, usage, and storage conditions. Serialization mandates are implemented to enhance traceability and prevent counterfeit products from entering the market, thereby safeguarding public health.
4Recent Regulatory Changes
Between 2024 and 2026, Ethiopia has implemented several policy changes affecting pharmaceutical imports. These include stricter enforcement of GMP certification requirements, enhanced scrutiny of import licensing processes, and the introduction of more rigorous quality control measures. These regulatory changes aim to improve the safety and efficacy of pharmaceuticals available in the Ethiopian market, aligning with international standards and protecting public health.
Ethiopian Pharmaceuticals Supply — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
EPSA's product strategy focuses on essential pharmaceuticals that address the primary health concerns in Ethiopia. The agency imports a range of medicines, including antibiotics, antimalarials, and other critical treatments, to meet the diverse healthcare needs of the population. Market demand drives these imports, with a particular emphasis on products that are in high demand due to prevalent health issues.
2Sourcing Profile
EPSA's sourcing strategy for generic drugs involves procuring formulations that meet international quality standards and are cost-effective. India plays a significant role in this strategy, serving as a key supplier due to its robust pharmaceutical manufacturing sector and adherence to international quality norms. EPSA's procurement process emphasizes quality assurance, regulatory compliance, and affordability to ensure that imported medicines are accessible to the Ethiopian public.
3Market Positioning
Based on its product mix, EPSA serves the public healthcare sector in Ethiopia, supplying essential medicines to hospitals, clinics, and health centers. The agency's focus on affordability and quality ensures that it meets the needs of government health institutions and contributes to the overall health system's effectiveness. EPSA's role is crucial in providing equitable access to essential pharmaceuticals across the country.
Seller's Guide — How to Become a Supplier to Ethiopian Pharmaceuticals Supply
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with EPSA, provided they meet the agency's quality standards and regulatory requirements. Gaps in EPSA's current sourcing may exist in specific therapeutic areas or formulations that are in high demand but underrepresented in the current supply chain. Identifying these gaps and offering suitable products can lead to successful partnerships with EPSA.
2Requirements & Qualifications
Indian exporters seeking to supply EPSA must obtain GMP certification recognized by EFDA, such as EU GMP, WHO GMP, or PIC/S. They must also comply with EFDA's import licensing requirements, including product registration and adherence to labeling and packaging standards. Ensuring that products meet EFDA's quality and safety standards is essential for successful market entry.
3How to Approach
To build a relationship with EPSA, Indian exporters should initiate contact through formal channels, providing comprehensive product dossiers that include quality certifications, regulatory approvals, and detailed product information. Participating in tenders and responding to EPSA's procurement notices can also facilitate engagement. Understanding the regulatory filing process and preparing for the necessary documentation and timelines is crucial for successful market entry.
Frequently Asked Questions — Ethiopian Pharmaceuticals Supply
What products does Ethiopian Pharmaceuticals Supply import from India?
Ethiopian Pharmaceuticals Supply imports 6 pharmaceutical products across 3 categories. Top imports: Artemether ($900.0K), Trimethoprim ($844.4K), Metronidazole ($650.0K), Albendazole ($350.0K), Norfloxacin ($200.0K).
Who supplies pharmaceuticals to Ethiopian Pharmaceuticals Supply from India?
Ethiopian Pharmaceuticals Supply sources from 34 verified Indian suppliers. The primary supplier is Fresenius Kabi India Private Limited (21.0% of imports, $3.8M).
What is Ethiopian Pharmaceuticals Supply's total pharmaceutical import value?
Ethiopian Pharmaceuticals Supply's total pharmaceutical import value from India is $3.1M, based on 67 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Ethiopian Pharmaceuticals Supply focus on?
Ethiopian Pharmaceuticals Supply imports across 3 categories. The largest: Advanced Antibiotics (54.8%), Antimalarial & Antiparasitic (40.4%), Antibiotics (4.8%).
Get Full Ethiopian Pharmaceuticals Supply Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Ethiopian Pharmaceuticals Supply identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Ethiopian Pharmaceuticals Supply's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 67 individual customs records matching Ethiopian Pharmaceuticals Supply.
- 5.Supplier Verification: Ethiopian Pharmaceuticals Supply sources from 34 verified Indian suppliers across 233 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
6 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.