Elara Pharmaservices Limited
Pharmaceutical Importer · United Kingdom · Cardiovascular Focus · $2.5M Total Trade · DGFT Verified
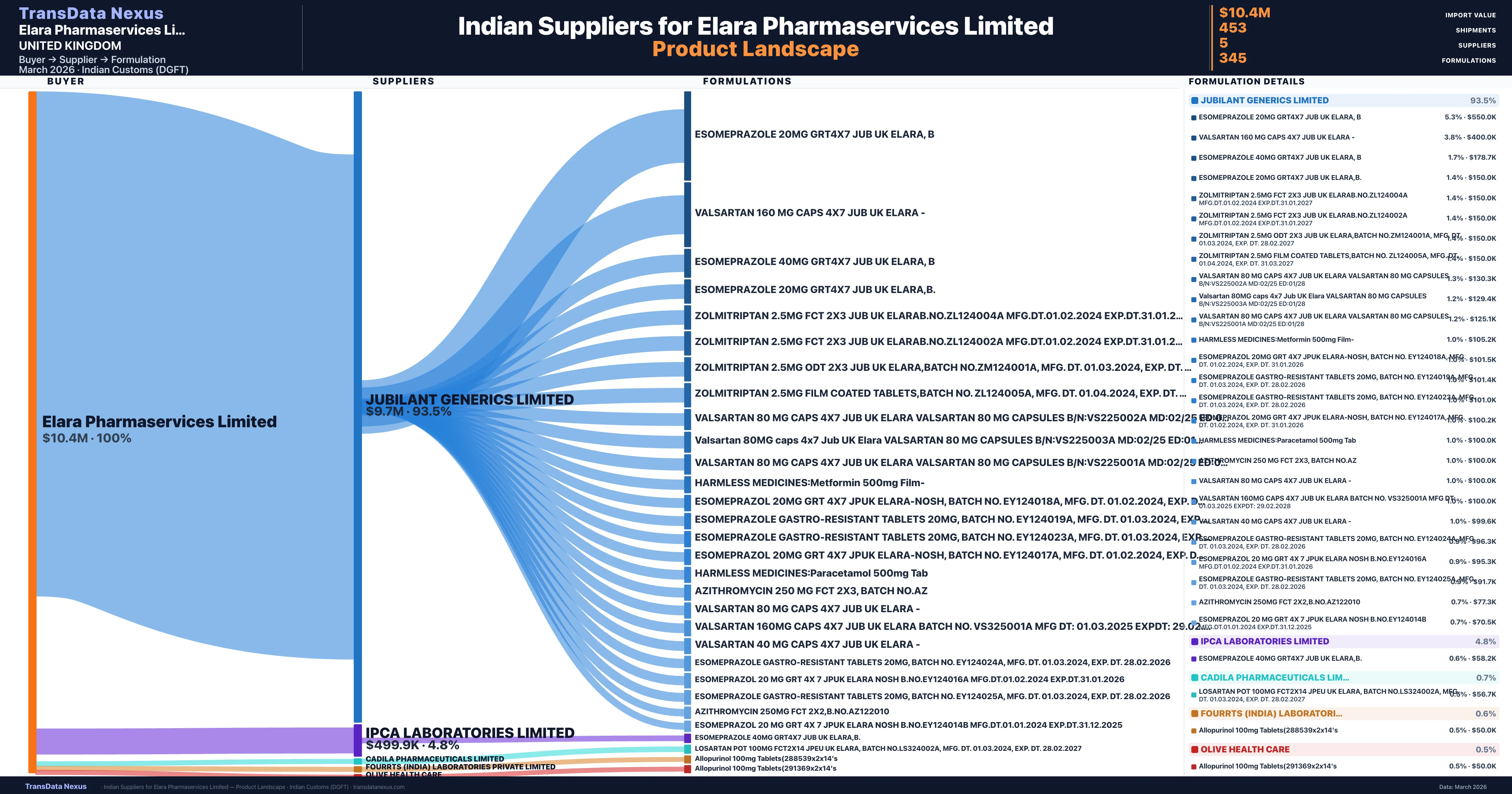
Elara Pharmaservices Limited is a pharmaceutical importer based in United Kingdom with a total trade value of $2.5M across 2 products in 2 therapeutic categories. Based on 50 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Elara Pharmaservices Limited sources from 5 verified Indian suppliers, with Jubilant Generics Limited accounting for 93.5% of imports.
Elara Pharmaservices Limited — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Elara Pharmaservices Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Jubilant Generics Limited | $9.7M | 428 | 93.5% |
| Ipca Laboratories Limited | $499.9K | 12 | 4.8% |
| Cadila Pharmaceuticals Limited | $69.2K | 7 | 0.7% |
| Fourrts (india) Laboratories Private Limited | $61.9K | 5 | 0.6% |
| Olive Health Care | $50.0K | 1 | 0.5% |
Elara Pharmaservices Limited sources from 5 verified Indian suppliers across 345 distinct formulations. The sourcing is highly concentrated — Jubilant Generics Limited accounts for 93.5% of total imports, indicating a strategic single-source relationship.
What Formulations Does Elara Pharmaservices Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Esomeprazole 20MG grt4x7 jub uk elara | $550.0K | 11 |
| Valsartan 160 MG CAPS 4x7 jub uk elara | $400.0K | 8 |
| Esomeprazole 40MG grt4x7 jub uk elara | $178.7K | 6 |
| Esomeprazole 20MG grt4x7 jub uk elara,b. | $150.0K | 3 |
| Zolmitriptan 2.5MG fct 2x3 jub uk elarab.no.zl124004a mfg.dt.01.02.2024 | $150.0K | 3 |
| Zolmitriptan 2.5MG fct 2x3 jub uk elarab.no.zl124002a mfg.dt.01.02.2024 | $150.0K | 3 |
| Zolmitriptan 2.5MG odt 2x3 jub uk elara,batch no.zm124001a, mfg. dt. 01.03.2024, exp. dt. | $150.0K | 3 |
| Zolmitriptan 2.5MG film coated tablets,batch no. zl124005a, mfg. dt. 01.04.2024, exp. dt. | $150.0K | 3 |
| Valsartan 80 MG CAPS 4x7 jub uk elara valsartan 80 MG capsules b/n:vs225002a md:02/25 | $130.3K | 3 |
| Valsartan 80MG CAPS 4x7 jub uk elara valsartan 80 MG capsules b/n:vs225003a md:02/25 | $129.4K | 3 |
| Valsartan 80 MG CAPS 4x7 jub uk elara valsartan 80 MG capsules b/n:vs225001a md:02/25 | $125.1K | 3 |
| Harmless medicines:metformin 500MG | $105.2K | 3 |
| Esomeprazol 20MG grt 4x7 jpuk elara-nosh, batch no. ey124018a, mfg. dt. 01.02.2024, exp. dt. | $101.5K | 3 |
| Esomeprazole Gastro-Resistant tablets 20MG, batch no. ey124019a, mfg. dt. 01.03.2024, exp. dt. | $101.4K | 3 |
| Esomeprazole Gastro-Resistant tablets 20MG, batch no. ey124023a, mfg. dt. 01.03.2024, exp. dt. | $101.0K | 3 |
Elara Pharmaservices Limited imports 345 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Elara Pharmaservices Limited Import?
Top Products by Import Value
Elara Pharmaservices Limited Therapeutic Categories — 2 Specializations
Elara Pharmaservices Limited imports across 2 therapeutic categories, with Cardiovascular (76.0%), Immunosuppressants (24.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Cardiovascular
1 products · 76.0% · $1.9M
Immunosuppressants
1 products · 24.0% · $600.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Valsartan | Cardiovascular | $1.9M | 38 | 1.0% | 13 |
| 2 | Azathioprine | Immunosuppressants | $600.0K | 12 | 1.8% | 10 |
Elara Pharmaservices Limited imports 2 pharmaceutical products across 2 categories into United Kingdom totaling $2.5M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Elara Pharmaservices Limited.
Request DemoElara Pharmaservices Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Elara Pharmaservices Limited is a private limited company incorporated on 17 April 2012, headquartered at The Barn Holly Berry House, Rough Park, Hamstall Ridware, Rugeley, Staffordshire, WS15 3SQ, United Kingdom. (find-and-update.company-information.service.gov.uk) The company specializes in management consultancy activities, particularly focusing on the pharmaceutical sector. It offers contract Qualified Person (QP) and technical support services, facilitating the release of non-sterile human and veterinary medicines to the market.
Elara Pharmaservices Limited holds a Manufacturer's Authorisation (ManA) from the Medicines and Healthcare products Regulatory Agency (MHRA), authorisation number UK MIA 41203, granted on 18 March 2025. (cms.mhra.gov.uk) This authorisation permits the company to import medicinal products, including both human and veterinary medicines, into the United Kingdom. The company's operations are supported by a team of directors, including Mr. Kafait Ali, Mr. Massimo Cunico, and Mrs. Joanne Lindsey Bunyan.
2Distribution Network
Elara Pharmaservices Limited operates multiple facilities to support its distribution network. These include sites at Iron Farm, 7 Grimesgate, Diseworth, Derby, DE74 2QD, and 5 Garden Court, Lockington Hall, Main Street, Lockington, Derby, DE74 2RH. (cms.mhra.gov.uk) While specific details about warehouse locations and logistics capabilities are not publicly disclosed, the company's authorisations indicate a structured approach to the importation and distribution of medicinal products within the United Kingdom.
3Industry Role
Elara Pharmaservices Limited functions as an importer and distributor within the United Kingdom's pharmaceutical supply chain. By holding a Manufacturer's Authorisation (ManA) and a Manufacturer's Import Authorisation (MIA), the company is authorised to import and distribute medicinal products, including both human and veterinary medicines. (cms.mhra.gov.uk) This authorisation underscores its role in ensuring the availability of imported pharmaceutical products in the UK market.
Supplier Relationship Intelligence — Elara Pharmaservices Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Elara Pharmaservices Limited's sourcing strategy exhibits a high degree of concentration, with a total import value of $2.5 million USD from India across 50 shipments. The top five products imported are Valsartan ($1.9 million, 76% of total imports) and Azathioprine ($600,000, 24% of total imports). This concentration indicates a strategic focus on specific therapeutic areas, particularly cardiovascular and immunosuppressant medications.
The company's reliance on a limited number of products suggests a targeted approach to meet specific market demands. However, this concentration also implies potential risks associated with supply chain disruptions or regulatory changes affecting these products. The stability of the relationship with Indian suppliers, such as Jubilant Generics Limited, which accounts for 93.5% of the total import value, is crucial for maintaining a consistent supply chain.
2Supply Chain Resilience
Elara Pharmaservices Limited's supply chain resilience is influenced by its sourcing strategy, which is heavily concentrated on a few products and suppliers. The company's reliance on Jubilant Generics Limited for the majority of its imports indicates a strong partnership but also exposes it to risks if this supplier faces operational challenges. The limited number of unique formulations imported (345) suggests a focused product range, which may impact the company's ability to adapt to changing market demands.
To enhance supply chain resilience, Elara Pharmaservices Limited could consider diversifying its supplier base and expanding its product portfolio. Engaging with additional suppliers and incorporating a broader range of formulations could mitigate risks associated with supplier dependency and market fluctuations.
3Strategic Implications
Elara Pharmaservices Limited's concentrated sourcing pattern positions it as a specialized importer focusing on specific therapeutic areas, particularly cardiovascular and immunosuppressant medications. This strategic focus allows the company to develop expertise and potentially negotiate better terms with suppliers. However, the high dependency on a limited number of products and suppliers may expose the company to risks related to supply chain disruptions, regulatory changes, or shifts in market demand.
For Indian exporters, this concentration presents an opportunity to become key suppliers for Elara Pharmaservices Limited by offering high-quality products in the cardiovascular and immunosuppressant categories. Establishing strong relationships with the company could lead to a stable and long-term partnership, provided they can meet the required quality standards and regulatory compliance.
Importing Pharmaceuticals into United Kingdom — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United Kingdom
1Regulatory Authority & Framework
The Medicines and Healthcare products Regulatory Agency (MHRA) is the regulatory authority overseeing pharmaceutical imports into the United Kingdom. The MHRA is responsible for ensuring that medicines and medical devices meet appropriate standards of safety, quality, and efficacy. (cms.mhra.gov.uk)
Key legislation governing pharmaceutical imports includes the Human Medicines Regulations 2012 (SI 2012/1916), which consolidates and updates previous regulations. This legislation outlines the requirements for the importation, distribution, and sale of medicinal products in the UK. (cms.mhra.gov.uk)
The marketing authorisation pathway for Indian generics involves obtaining a UK marketing authorisation, which can be achieved through the European Medicines Agency (EMA) or the MHRA. The process includes demonstrating that the generic product is bioequivalent to the reference product and complies with Good Manufacturing Practice (GMP) standards.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products in the UK are governed by the Human Medicines Regulations 2012. Importers must hold a valid Manufacturer's Import Authorisation (MIA) issued by the MHRA. Elara Pharmaservices Limited holds MIA 41203, authorising it to import medicinal products into the UK. (cms.mhra.gov.uk)
Good Manufacturing Practice (GMP) certificates are essential for ensuring the quality and safety of imported medicines. The MHRA recognises GMP certificates from countries with equivalent standards, including those issued by the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Elara Pharmaservices Limited's authorisation indicates compliance with GMP standards, as evidenced by the MHRA's inspection and certification. (cms.mhra.gov.uk)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure they meet the required quality standards. The MHRA conducts inspections to verify compliance with GMP and other regulatory requirements. (cms.mhra.gov.uk)
Stability requirements for medicinal products are outlined in the British Pharmacopoeia (BP), which provides specifications for the quality of medicines. Labeling language requirements stipulate that labels must be in English, ensuring clear communication with healthcare professionals and patients. Serialization mandates are part of the EU Falsified Medicines Directive, which aims to prevent counterfeit medicines from entering the supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, the UK has implemented several regulatory changes affecting pharmaceutical imports. Post-Brexit, the UK has established its own regulatory framework, with the MHRA taking over responsibilities previously held by the EMA. This includes the introduction of the UK Conformity Assessed (UKCA) mark, replacing the CE mark for medical devices. Additionally, the UK has updated its import licensing requirements and GMP standards to align with international best practices.
Elara Pharmaservices Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Elara Pharmaservices Limited's focus on importing cardiovascular and immunosuppressant medications aligns with the therapeutic areas of Valsartan and Azathioprine, which constitute the majority of its imports. This strategic focus suggests an in-depth understanding of these therapeutic areas and a commitment to meeting the specific needs of the UK market.
The market demand for these products is driven by the prevalence of cardiovascular diseases and conditions requiring immunosuppression in the UK. By concentrating on these areas, Elara Pharmaservices Limited can leverage its expertise to provide high-quality, essential medicines to healthcare providers and patients.
2Sourcing Profile
Elara Pharmaservices Limited's sourcing strategy is centred on importing finished pharmaceutical formulations from India, focusing on specific therapeutic categories. This approach allows the company to offer a targeted product range that meets the needs of the UK market.
India's well-established pharmaceutical industry, known for its adherence to international quality standards, makes it a reliable source for these products. By importing from India, Elara Pharmaservices Limited can ensure a consistent supply of high-quality medicines while benefiting from competitive pricing.
3Market Positioning
Based on its product mix, Elara Pharmaservices Limited serves the wholesale distribution segment of the UK pharmaceutical market. By importing and distributing essential medicines, the company plays a crucial role in ensuring the availability of these products to healthcare providers and patients across the country.
Frequently Asked Questions — Elara Pharmaservices Limited
What products does Elara Pharmaservices Limited import from India?
Elara Pharmaservices Limited imports 2 pharmaceutical products across 2 categories. Top imports: Valsartan ($1.9M), Azathioprine ($600.0K).
Who supplies pharmaceuticals to Elara Pharmaservices Limited from India?
Elara Pharmaservices Limited sources from 5 verified Indian suppliers. The primary supplier is Jubilant Generics Limited (93.5% of imports, $9.7M).
What is Elara Pharmaservices Limited's total pharmaceutical import value?
Elara Pharmaservices Limited's total pharmaceutical import value from India is $2.5M, based on 50 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Elara Pharmaservices Limited focus on?
Elara Pharmaservices Limited imports across 2 categories. The largest: Cardiovascular (76.0%), Immunosuppressants (24.0%).
Get Full Elara Pharmaservices Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Elara Pharmaservices Limited identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Elara Pharmaservices Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 50 individual customs records matching Elara Pharmaservices Limited.
- 5.Supplier Verification: Elara Pharmaservices Limited sources from 5 verified Indian suppliers across 345 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.