Digecitss -ministerio DE Salud Publ
Pharmaceutical Importer · Dominican Republic · Antiviral & HIV Medications Focus · $400.0K Total Trade · DGFT Verified
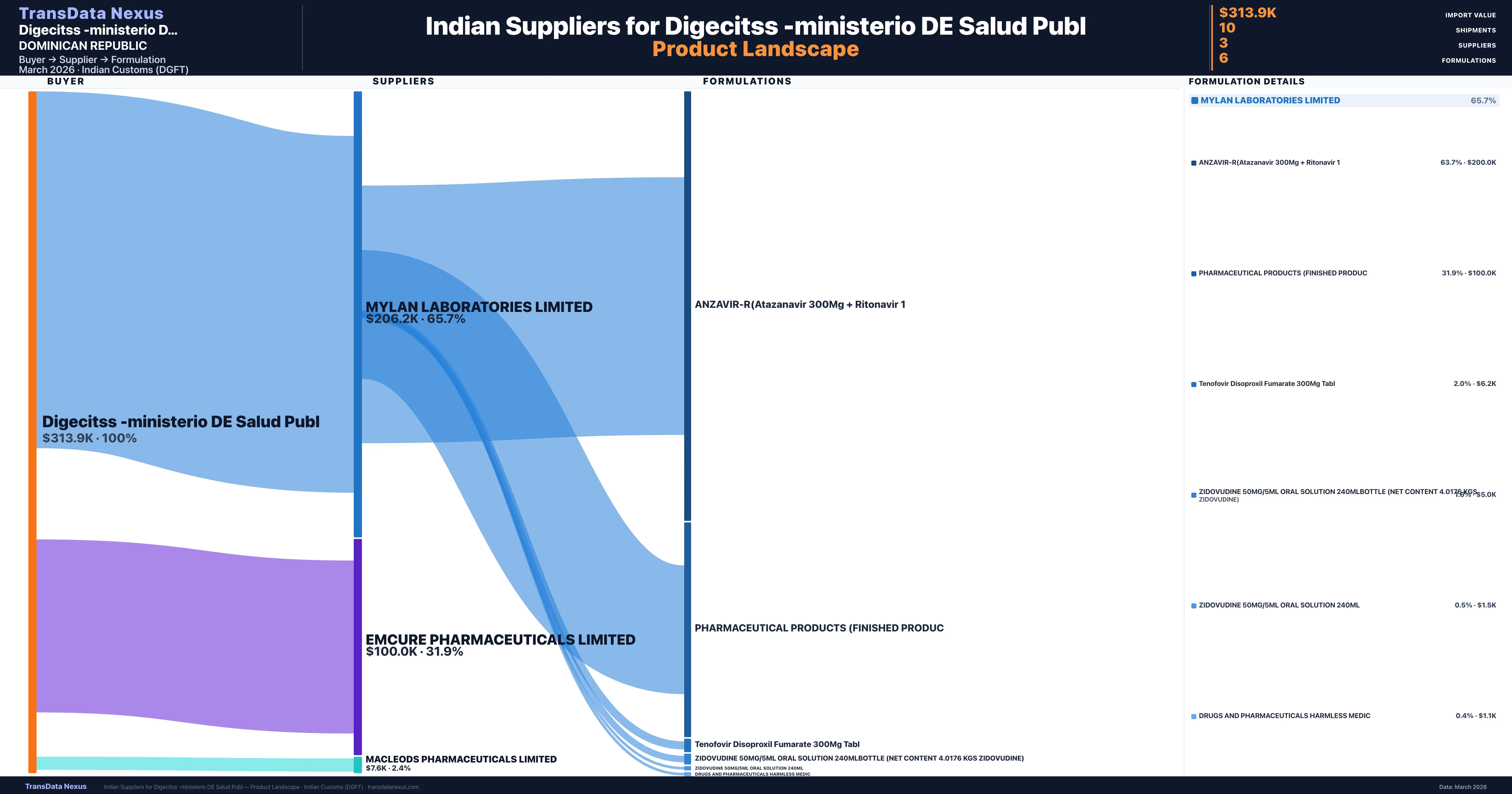
Digecitss -ministerio DE Salud Publ is a pharmaceutical importer based in Dominican Republic with a total trade value of $400.0K across 2 products in 1 therapeutic categories. Based on 8 verified import shipments from Indian Customs (DGFT) records, Digecitss -ministerio DE Salud Publ is the #1 buyer in 1 product including Atazanavir. Digecitss -ministerio DE Salud Publ sources from 3 verified Indian suppliers, with Mylan Laboratories Limited accounting for 65.7% of imports.
Digecitss -ministerio DE Salud Publ — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Digecitss -ministerio DE Salud Publ?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Mylan Laboratories Limited | $206.2K | 5 | 65.7% |
| Emcure Pharmaceuticals Limited | $100.0K | 2 | 31.9% |
| Macleods Pharmaceuticals Limited | $7.6K | 3 | 2.4% |
Digecitss -ministerio DE Salud Publ sources from 3 verified Indian suppliers across 6 distinct formulations. Total import value: $313.9K across 10 shipments.
What Formulations Does Digecitss -ministerio DE Salud Publ Import?
| Formulation | Value | Ships |
|---|---|---|
| Anzavir-r(atazanavir 300MG + ritonavir | $200.0K | 4 |
| Pharmaceutical products (finished | $100.0K | 2 |
| Tenofovir disoproxil fumarate 300MG | $6.2K | 1 |
| Zidovudine 50MG/5ML oral solution 240mlbottle (net content 4.0176 kgs zidovudine) | $5.0K | 1 |
| Zidovudine 50MG/5ML oral solution 240ML | $1.5K | 1 |
| Drugs and pharmaceuticals harmless | $1.1K | 1 |
Digecitss -ministerio DE Salud Publ imports 6 distinct pharmaceutical formulations. Showing top 6 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Digecitss -ministerio DE Salud Publ Import?
Top Products by Import Value
Digecitss -ministerio DE Salud Publ Therapeutic Categories — 1 Specializations
Digecitss -ministerio DE Salud Publ imports across 1 therapeutic categories, with Antiviral & HIV Medications (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antiviral & HIV Medications
2 products · 100.0% · $400.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Atazanavir | Antiviral & HIV Medications | $200.0K | 4 | 0.3% | 1 |
| 2 | Ritonavir | Antiviral & HIV Medications | $200.0K | 4 | 0.1% | 7 |
Digecitss -ministerio DE Salud Publ imports 2 pharmaceutical products across 1 categories into Dominican Republic totaling $400.0K. The company is the #1 buyer for 1 product: Atazanavir.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Digecitss -ministerio DE Salud Publ.
Request DemoDigecitss -ministerio DE Salud Publ — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Digecitss, officially known as the Dirección General de Control de las Infecciones de Transmisión Sexual y Sida, is a specialized division within the Dominican Republic's Ministry of Public Health and Social Assistance (MISPAS). Established to oversee the control and prevention of sexually transmitted infections (STIs) and HIV/AIDS, Digecitss plays a pivotal role in the nation's public health infrastructure. Its primary functions include the development and implementation of national strategies for STI and HIV prevention, education, and treatment, as well as the coordination of resources and policies aimed at mitigating the impact of these diseases. While Digecitss is not a pharmaceutical importer or buyer, it collaborates closely with various stakeholders, including pharmaceutical entities, to ensure the availability and distribution of essential medications for the treatment and prevention of HIV/AIDS and other STIs.
2Distribution Network
As a governmental health agency, Digecitss does not operate a traditional distribution network for pharmaceutical products. Instead, it partners with authorized pharmaceutical importers and distributors within the Dominican Republic to facilitate the procurement and distribution of medications necessary for the national response to HIV/AIDS and other STIs. These collaborations ensure that essential medicines are accessible to healthcare facilities and patients across the country.
3Industry Role
Digecitss serves as a central coordinating body within the Dominican Republic's pharmaceutical supply chain, particularly concerning the procurement and distribution of medications for the treatment and prevention of HIV/AIDS and other STIs. While it does not function as a pharmaceutical importer or distributor itself, Digecitss plays a critical role in ensuring that the necessary medications are available and accessible to the population. This involves working with various stakeholders, including pharmaceutical companies, importers, and healthcare providers, to facilitate the effective delivery of healthcare services related to STI and HIV prevention and treatment.
Supplier Relationship Intelligence — Digecitss -ministerio DE Salud Publ
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Digecitss's procurement strategy for HIV/AIDS and STI medications involves sourcing from a diverse range of pharmaceutical suppliers to ensure a stable and uninterrupted supply of essential medicines. By collaborating with multiple suppliers, Digecitss mitigates the risks associated with single-source dependency, such as supply chain disruptions or price fluctuations. This approach enhances the resilience and reliability of the pharmaceutical supply chain, ensuring that patients have continuous access to necessary treatments.
2Supply Chain Resilience
The resilience of Digecitss's pharmaceutical supply chain is bolstered by its strategic partnerships with multiple suppliers, including those from India. This diversification ensures that the supply chain is not overly reliant on a single source, reducing the risk of disruptions due to geopolitical issues, regulatory changes, or other unforeseen events. Additionally, by sourcing from suppliers with recognized Good Manufacturing Practices (GMP) certifications, Digecitss ensures that the medications meet international quality standards, further strengthening the integrity and reliability of the supply chain.
3Strategic Implications
For Digecitss, maintaining a diversified sourcing strategy is crucial for ensuring the continuous availability of high-quality HIV/AIDS and STI medications. This approach not only mitigates potential risks associated with supply chain disruptions but also fosters competitive pricing and innovation among suppliers. For Indian pharmaceutical exporters, the Dominican Republic represents a promising market, especially given the country's focus on HIV/AIDS and STI prevention and treatment. Establishing partnerships with Digecitss and other stakeholders can lead to increased market penetration and contribute to the global effort in combating these diseases.
Importing Pharmaceuticals into Dominican Republic — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Dominican Republic
1Regulatory Authority & Framework
The primary regulatory authority overseeing pharmaceutical imports in the Dominican Republic is the Ministry of Public Health and Social Assistance (MISPAS), specifically through the General Directorate of Medicines, Food, and Health Products (DIGEMAPS). This body is responsible for ensuring that all pharmaceutical products entering the country meet the required safety, efficacy, and quality standards. The regulatory framework governing pharmaceutical imports includes the General Health Law and Regulation No. 246-06 on Medicines, which outline the procedures and requirements for the registration, importation, and distribution of pharmaceutical products. These regulations are designed to protect public health by ensuring that only safe and effective medications are available in the Dominican market.
2Import Licensing & GMP
Importers of pharmaceutical products into the Dominican Republic must obtain a Sanitary Register from DIGEMAPS for each product they intend to import. This registration process involves submitting a comprehensive technical dossier that includes evidence of compliance with Good Manufacturing Practices (GMP), product specifications, labeling information, and other relevant documentation. The Sanitary Register is valid for five years and must be renewed upon expiration. Additionally, importers must adhere to customs regulations, including registration with the General Directorate of Customs (DGA) and compliance with import procedures. Working with licensed customs brokers is recommended to navigate the complexities of the import process effectively.
3Quality & Labeling
Pharmaceutical products imported into the Dominican Republic must meet specific quality standards, including compliance with GMP certifications recognized by international bodies such as the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Labeling requirements stipulate that all information be provided in Spanish and include details such as product name, active ingredients, dosage form, manufacturer information, batch number, expiration date, and storage conditions. Additionally, products must undergo batch testing to ensure quality and safety before being released into the market.
4Recent Regulatory Changes
Between 2024 and 2026, the Dominican Republic has implemented several regulatory changes affecting pharmaceutical imports. These include updates to the General Health Law and Regulation No. 246-06 on Medicines, aimed at enhancing the efficiency and transparency of the importation process. The changes also focus on strengthening the enforcement of GMP compliance and improving the monitoring of imported pharmaceutical products to ensure they meet the required safety and efficacy standards. These regulatory updates reflect the Dominican Republic's commitment to safeguarding public health and ensuring the availability of high-quality medications.
Digecitss -ministerio DE Salud Publ — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Digecitss's focus on HIV/AIDS and STI medications aligns with the significant public health challenges posed by these diseases in the Dominican Republic. The high prevalence of HIV/AIDS necessitates a robust response, including the availability of effective treatments and preventive measures. By prioritizing these therapeutic areas, Digecitss aims to reduce transmission rates, improve patient outcomes, and enhance the overall quality of life for individuals affected by these conditions. The strategic emphasis on HIV/AIDS and STI medications is driven by both domestic health needs and international commitments to combat these diseases.
2Sourcing Profile
Digecitss's sourcing strategy for HIV/AIDS and STI medications involves procuring products from a diverse range of suppliers, including those from India. This approach ensures a stable and uninterrupted supply of essential medicines, mitigating risks associated with single-source dependency. The focus on finished pharmaceutical formulations, rather than raw active pharmaceutical ingredients (APIs), allows for more efficient distribution and ensures that medications are ready for patient use upon arrival. By sourcing from suppliers with recognized GMP certifications, Digecitss ensures that the imported products meet international quality standards, thereby safeguarding public health.
3Market Positioning
Digecitss serves a critical role in the Dominican Republic's healthcare system by ensuring the availability and distribution of HIV/AIDS and STI medications. Its focus on these therapeutic areas positions it as a key player in the public health sector, particularly in the treatment and prevention of infectious diseases. By collaborating with various stakeholders, including pharmaceutical companies, healthcare providers, and international organizations, Digecitss contributes to the national effort to combat HIV/AIDS and other STIs, thereby enhancing the overall health and well-being of the population.
Seller's Guide — How to Become a Supplier to Digecitss -ministerio DE Salud Publ
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
For Indian pharmaceutical exporters, the Dominican Republic presents a promising market for HIV/AIDS and STI medications. Digecitss's diversified sourcing strategy indicates an openness to engaging with new suppliers who can meet the required quality standards and regulatory requirements. Establishing partnerships with Digecitss and other stakeholders can lead to increased market penetration and contribute to the global effort in combating these diseases. However, potential suppliers must be prepared to navigate the regulatory complexities of the Dominican market and demonstrate compliance with GMP standards recognized by Dominican authorities.
2Requirements & Qualifications
Indian exporters seeking to supply pharmaceutical products to the Dominican Republic must ensure that their products are registered with DIGEMAPS and comply with the Sanitary Register requirements. This includes providing a comprehensive technical dossier, evidence of GMP compliance, and labeling information in Spanish. Additionally, exporters must adhere to customs regulations, including registration with the DGA and compliance with import procedures. Working with licensed customs brokers and regulatory experts is recommended to navigate the complexities of the import process effectively.
3How to Approach
To establish a successful partnership with Digecitss and other stakeholders in the Dominican Republic, Indian pharmaceutical exporters should focus on building relationships through direct engagement and participation in relevant industry events and forums. Understanding and complying with the regulatory requirements, including obtaining the necessary registrations and certifications, is crucial. Developing a clear strategy for market entry, including pricing, distribution, and promotional activities, will enhance the prospects of success. Patience and persistence are essential, as navigating the regulatory landscape and establishing trust with local partners can be time-consuming.
Frequently Asked Questions — Digecitss -ministerio DE Salud Publ
What products does Digecitss -ministerio DE Salud Publ import from India?
Digecitss -ministerio DE Salud Publ imports 2 pharmaceutical products across 1 categories. Top imports: Atazanavir ($200.0K), Ritonavir ($200.0K).
Who supplies pharmaceuticals to Digecitss -ministerio DE Salud Publ from India?
Digecitss -ministerio DE Salud Publ sources from 3 verified Indian suppliers. The primary supplier is Mylan Laboratories Limited (65.7% of imports, $206.2K).
What is Digecitss -ministerio DE Salud Publ's total pharmaceutical import value?
Digecitss -ministerio DE Salud Publ's total pharmaceutical import value from India is $400.0K, based on 8 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Digecitss -ministerio DE Salud Publ focus on?
Digecitss -ministerio DE Salud Publ imports across 1 categories. The largest: Antiviral & HIV Medications (100.0%).
Get Full Digecitss -ministerio DE Salud Publ Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Digecitss -ministerio DE Salud Publ identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Digecitss -ministerio DE Salud Publ's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 8 individual customs records matching Digecitss -ministerio DE Salud Publ.
- 5.Supplier Verification: Digecitss -ministerio DE Salud Publ sources from 3 verified Indian suppliers across 6 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.