CSP Mylan SAS AT Csp- Labo 274,
Pharmaceutical Importer · France · Diabetes & Endocrine Focus · $8.0M Total Trade · DGFT Verified
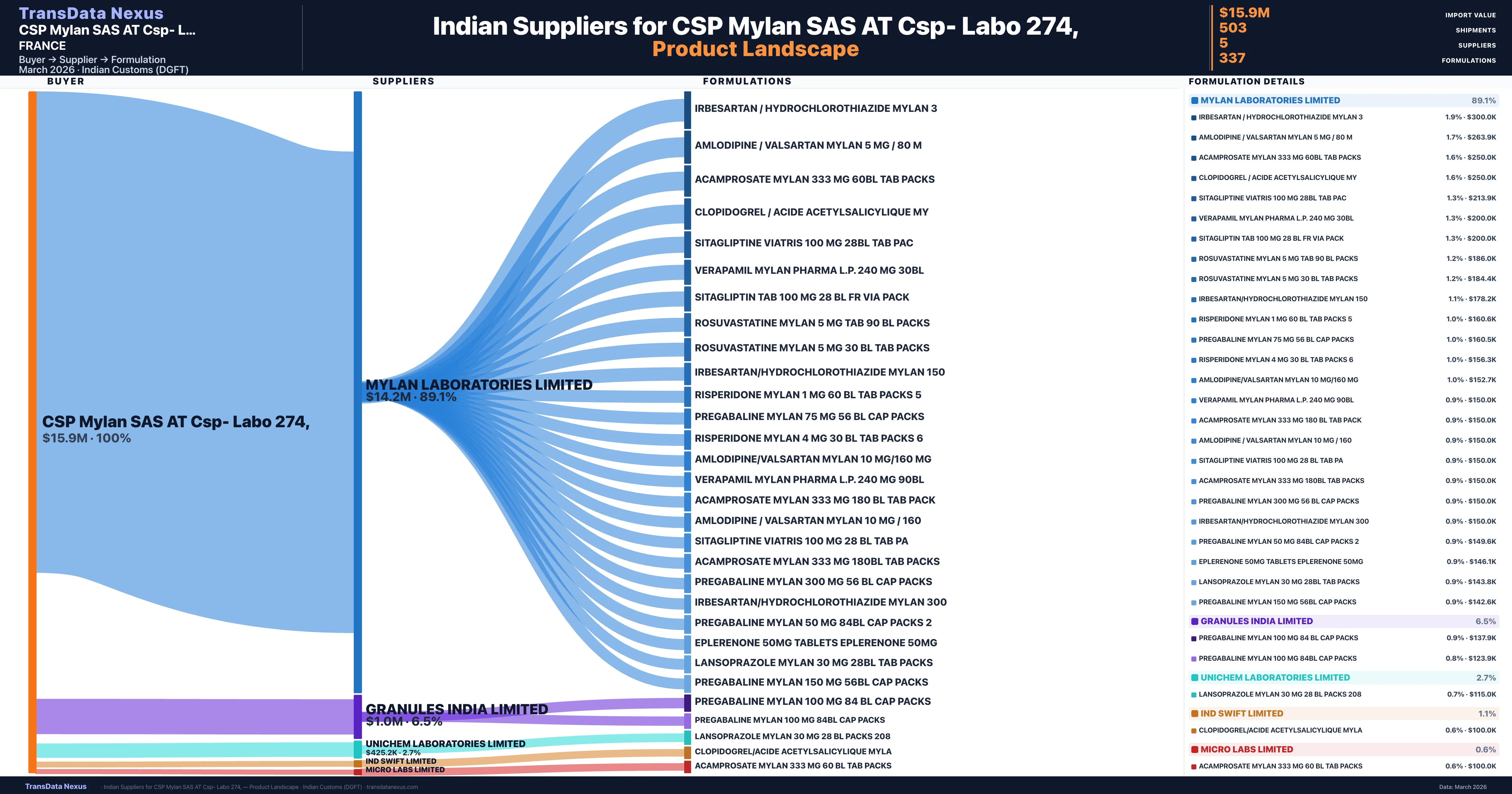
CSP Mylan SAS AT Csp- Labo 274, is a pharmaceutical importer based in France with a total trade value of $8.0M across 6 products in 5 therapeutic categories. Based on 160 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. CSP Mylan SAS AT Csp- Labo 274, sources from 5 verified Indian suppliers, with Mylan Laboratories Limited accounting for 89.1% of imports.
CSP Mylan SAS AT Csp- Labo 274, — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to CSP Mylan SAS AT Csp- Labo 274,?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Mylan Laboratories Limited | $14.2M | 457 | 89.1% |
| Granules India Limited | $1.0M | 27 | 6.5% |
| Unichem Laboratories Limited | $425.2K | 10 | 2.7% |
| Ind Swift Limited | $171.5K | 7 | 1.1% |
| Micro Labs Limited | $97.5K | 2 | 0.6% |
CSP Mylan SAS AT Csp- Labo 274, sources from 5 verified Indian suppliers across 337 distinct formulations. The sourcing is highly concentrated — Mylan Laboratories Limited accounts for 89.1% of total imports, indicating a strategic single-source relationship.
What Formulations Does CSP Mylan SAS AT Csp- Labo 274, Import?
| Formulation | Value | Ships |
|---|---|---|
| Irbesartan / hydrochlorothiazide mylan | $300.0K | 6 |
| Amlodipine / valsartan mylan 5 MG / 80 | $263.9K | 9 |
| Acamprosate mylan 333 MG 60bl TAB | $250.0K | 5 |
| Clopidogrel / acide acetylsalicylique | $250.0K | 5 |
| Sitagliptine viatris 100 MG 28bl TAB | $213.9K | 5 |
| Verapamil mylan pharma l.p. 240 MG | $200.0K | 4 |
| Sitagliptin TAB 100 MG 28 bl fr via | $200.0K | 4 |
| Rosuvastatine mylan 5 MG TAB 90 bl | $186.0K | 6 |
| Rosuvastatine mylan 5 MG 30 bl TAB | $184.4K | 5 |
| Irbesartan/hydrochlorothiazide mylan | $178.2K | 4 |
| Risperidone mylan 1 MG 60 bl TAB packs | $160.6K | 4 |
| Pregabaline mylan 75 MG 56 bl cap | $160.5K | 4 |
| Risperidone mylan 4 MG 30 bl TAB packs | $156.3K | 4 |
| Amlodipine/valsartan mylan 10 MG/160 MG | $152.7K | 4 |
| Verapamil mylan pharma l.p. 240 MG | $150.0K | 3 |
CSP Mylan SAS AT Csp- Labo 274, imports 337 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does CSP Mylan SAS AT Csp- Labo 274, Import?
Top Products by Import Value
CSP Mylan SAS AT Csp- Labo 274, Therapeutic Categories — 5 Specializations
CSP Mylan SAS AT Csp- Labo 274, imports across 5 therapeutic categories, with Diabetes & Endocrine (46.9%), Cardiovascular (26.3%), Diuretics (16.3%) representing the largest segments. The portfolio is concentrated — top 5 products = 97% of total imports.
Diabetes & Endocrine
2 products · 46.9% · $3.8M
Cardiovascular
1 products · 26.3% · $2.1M
Diuretics
1 products · 16.3% · $1.3M
Lipid & Metabolism
1 products · 7.5% · $600.0K
Antifungals
1 products · 3.1% · $250.0K
Import Portfolio — Top 6 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Metformin | Diabetes & Endocrine | $3.3M | 65 | 0.7% | 16 |
| 2 | Irbesartan | Cardiovascular | $2.1M | 42 | 1.1% | 11 |
| 3 | Hydrochlorothiazide | Diuretics | $1.3M | 26 | 0.3% | 16 |
| 4 | Ezetimibe | Lipid & Metabolism | $600.0K | 12 | 0.1% | 14 |
| 5 | Sitagliptin | Diabetes & Endocrine | $500.0K | 10 | 0.7% | 18 |
| 6 | Terbinafine | Antifungals | $250.0K | 5 | 0.9% | 15 |
CSP Mylan SAS AT Csp- Labo 274, imports 6 pharmaceutical products across 5 categories into France totaling $8.0M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for CSP Mylan SAS AT Csp- Labo 274,.
Request DemoCSP Mylan SAS AT Csp- Labo 274, — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
CSP Mylan SAS AT Csp- Labo 274, based in Cournon-d'Auvergne, France, is a pharmaceutical importer specializing in the acquisition of finished pharmaceutical formulations from international suppliers. As part of the CSP Group, a family-operated company founded in 1951, it plays a significant role in the French pharmaceutical distribution network. The CSP Group is recognized as a leading pharmaceutical depository in France and Europe, serving a diverse clientele that includes pharmaceutical laboratories, wholesale distributors, pharmacies, healthcare establishments, and supermarkets.
CSP Mylan SAS AT Csp- Labo 274's operations are integral to the CSP Group's extensive logistics and distribution capabilities. The CSP Group manages a total of 200,000 m² of warehouse space across France and the Benelux region, with facilities in Cournon-d'Auvergne, Moussy-le-Neuf near Paris, and Belgium. This infrastructure supports the efficient importation and distribution of pharmaceutical products, ensuring timely delivery to various stakeholders in the healthcare sector.
2Distribution Network
CSP Mylan SAS AT Csp- Labo 274 operates within the CSP Group's extensive distribution network, which encompasses over 200,000 m² of warehouse space across France and the Benelux region. Key facilities include the 82,000 m² site in Cournon-d'Auvergne near Clermont-Ferrand, the 83,000 m² warehouse in Moussy-le-Neuf near Paris, and a 15,000 m² facility in Belgium. These strategically located warehouses enable CSP to efficiently manage the importation, storage, and distribution of pharmaceutical products throughout France and neighboring countries.
3Industry Role
CSP Mylan SAS AT Csp- Labo 274 functions as a pharmaceutical importer within the French supply chain, sourcing finished pharmaceutical formulations from international suppliers, particularly from India. Its role is pivotal in ensuring the availability of a wide range of pharmaceutical products in the French market, thereby supporting the operations of pharmacies, hospitals, and other healthcare providers. By leveraging the CSP Group's extensive logistics and distribution network, the company contributes to the efficient delivery of medications to various stakeholders in the healthcare sector.
Supplier Relationship Intelligence — CSP Mylan SAS AT Csp- Labo 274,
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
CSP Mylan SAS AT Csp- Labo 274 demonstrates a high degree of sourcing concentration, with a total import value of $8.0 million USD from India over 160 shipments, primarily consisting of six products across five therapeutic categories. The top five products—Metformin, Irbesartan, Hydrochlorothiazide, Ezetimibe, and Sitagliptin—account for 96.9% of the total import value, indicating a focused procurement strategy. This concentration suggests a strategic choice to streamline operations and maintain strong supplier relationships, potentially enhancing negotiation leverage and ensuring consistent product quality. However, such dependency also exposes the company to risks associated with supply chain disruptions, price fluctuations, and regulatory changes affecting these specific products.
The shipment data reveals a stable relationship with key suppliers, notably Mylan Laboratories Limited, which accounts for 89.1% of the total import value. This long-term partnership likely contributes to operational efficiency and product consistency. Nonetheless, the limited number of suppliers and the high concentration of imports in a few products may pose challenges in mitigating supply chain risks and adapting to market changes.
2Supply Chain Resilience
CSP Mylan SAS AT Csp- Labo 274's supply chain resilience is closely tied to its sourcing strategy from India. The company's reliance on a limited number of suppliers, particularly Mylan Laboratories Limited, indicates a focused procurement approach. While this can lead to operational efficiencies and strong supplier relationships, it also presents challenges in terms of supply chain diversification. The lack of backup suppliers for key products may expose the company to risks associated with supply disruptions, price volatility, and regulatory changes affecting these suppliers. Additionally, the concentration of imports in specific formulations and therapeutic categories may limit the company's ability to adapt to shifts in market demand or regulatory requirements.
3Strategic Implications
The sourcing pattern of CSP Mylan SAS AT Csp- Labo 274 reflects a strategic focus on a narrow range of products and suppliers, which can lead to operational efficiencies and strong supplier relationships. However, this concentration also exposes the company to risks associated with supply chain disruptions, price fluctuations, and regulatory changes affecting these specific products and suppliers. For Indian exporters, understanding this sourcing strategy is crucial for positioning themselves as potential suppliers. Offering a diverse product portfolio and ensuring compliance with French regulatory standards can enhance the attractiveness of their offerings to CSP Mylan SAS AT Csp- Labo 274. Additionally, demonstrating the ability to provide consistent product quality and reliable delivery schedules can strengthen the partnership potential.
Importing Pharmaceuticals into France — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for France
1Regulatory Authority & Framework
In France, the primary regulatory authority overseeing pharmaceutical imports is the Agence Nationale de Sécurité du Médicament et des Produits de Santé (ANSM). ANSM is responsible for ensuring the safety, efficacy, and quality of medicines and health products available in the French market. Key legislation governing pharmaceutical imports includes the French Public Health Code, which outlines the requirements for marketing authorization, importation, and distribution of medicinal products. The European Medicines Agency (EMA) also plays a significant role in harmonizing regulatory standards across EU member states, including France, facilitating the marketing authorization process for medicinal products.
For Indian generics to enter the French market, they must obtain marketing authorization from ANSM. This process involves submitting a comprehensive dossier that includes data on the quality, safety, and efficacy of the product. The dossier must demonstrate that the generic product is bioequivalent to the reference product, ensuring that it performs in the same manner as the original. The bioequivalence study should adhere to the guidelines set forth by ANSM and EMA, which may include requirements for clinical trials and other supporting data. Once approved, the product is granted a marketing authorization, allowing it to be imported and distributed within France.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products in France are stringent, ensuring that all imported medicines meet the necessary quality and safety standards. Importers must hold a wholesale distribution authorization issued by ANSM, which permits the importation and distribution of medicinal products within France. This authorization requires compliance with Good Distribution Practice (GDP) guidelines, ensuring that the storage and transportation of medicines maintain their quality and integrity.
Regarding Good Manufacturing Practice (GMP), France recognizes certifications from various international bodies, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Indian manufacturers seeking to export to France must ensure that their facilities are GMP-compliant and hold valid certifications from recognized authorities. This compliance is crucial for obtaining marketing authorization and facilitating the importation process.
3Quality & Labeling
Imported pharmaceutical products in France are subject to rigorous quality control measures. Batch testing is mandatory to verify the quality, safety, and efficacy of each batch before it is released into the market. This testing ensures that the products meet the specifications outlined in the marketing authorization and comply with French and EU regulations.
Stability requirements are also stringent, with products needing to demonstrate stability under specified conditions to ensure their shelf-life and effectiveness. Labeling must be in French and include all necessary information, such as the product name, active ingredients, dosage form, strength, batch number, expiry date, storage conditions, and instructions for use. Serialization mandates require that each unit of the product be uniquely identified, facilitating traceability and enhancing patient safety.
4Recent Regulatory Changes
Between 2024 and 2026, several policy changes have impacted the importation of Indian pharmaceutical products into France. The French government has implemented stricter regulations concerning the bioequivalence studies required for generic drugs, aligning with EU standards to ensure the therapeutic equivalence of generics to their reference products. Additionally, there has been an increased emphasis on the traceability of pharmaceutical products, with enhanced serialization requirements to combat counterfeit medicines and ensure patient safety. These changes necessitate that Indian exporters adapt their manufacturing and documentation processes to comply with the updated French regulatory framework.
CSP Mylan SAS AT Csp- Labo 274, — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
CSP Mylan SAS AT Csp- Labo 274's product category focus—Diabetes & Endocrine (46.9%), Cardiovascular (26.3%), and Diuretics (16.3%)—aligns with prevalent health concerns in France. The high import values of Metformin, Irbesartan, and Hydrochlorothiazide indicate a strong market demand for these medications. This demand is driven by the significant prevalence of diabetes, hypertension, and related cardiovascular conditions among the French population. By focusing on these therapeutic areas, CSP Mylan SAS AT Csp- Labo 274 addresses critical healthcare needs, ensuring the availability of essential treatments for chronic conditions.
2Sourcing Profile
CSP Mylan SAS AT Csp- Labo 274's sourcing strategy emphasizes the procurement of generic drugs, particularly from India, which is known
Frequently Asked Questions — CSP Mylan SAS AT Csp- Labo 274,
What products does CSP Mylan SAS AT Csp- Labo 274, import from India?
CSP Mylan SAS AT Csp- Labo 274, imports 6 pharmaceutical products across 5 categories. Top imports: Metformin ($3.3M), Irbesartan ($2.1M), Hydrochlorothiazide ($1.3M), Ezetimibe ($600.0K), Sitagliptin ($500.0K).
Who supplies pharmaceuticals to CSP Mylan SAS AT Csp- Labo 274, from India?
CSP Mylan SAS AT Csp- Labo 274, sources from 5 verified Indian suppliers. The primary supplier is Mylan Laboratories Limited (89.1% of imports, $14.2M).
What is CSP Mylan SAS AT Csp- Labo 274,'s total pharmaceutical import value?
CSP Mylan SAS AT Csp- Labo 274,'s total pharmaceutical import value from India is $8.0M, based on 160 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does CSP Mylan SAS AT Csp- Labo 274, focus on?
CSP Mylan SAS AT Csp- Labo 274, imports across 5 categories. The largest: Diabetes & Endocrine (46.9%), Cardiovascular (26.3%), Diuretics (16.3%).
Get Full CSP Mylan SAS AT Csp- Labo 274, Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: CSP Mylan SAS AT Csp- Labo 274, identified across shipments using consignee name normalization, aggregating 3 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as CSP Mylan SAS AT Csp- Labo 274,'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 160 individual customs records matching CSP Mylan SAS AT Csp- Labo 274,.
- 5.Supplier Verification: CSP Mylan SAS AT Csp- Labo 274, sources from 5 verified Indian suppliers across 337 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
6 Products Tracked
5 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 3 company name variants from customs records. For current shipment-level data, contact TransData Nexus.