Bpost International EMC
Pharmaceutical Importer · Belgium · Diabetes & Endocrine Focus · $202.9K Total Trade · DGFT Verified
Bpost International EMC is a pharmaceutical importer based in Belgium with a total trade value of $202.9K across 3 products in 2 therapeutic categories. Based on 320 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Bpost International EMC sources from 4 verified Indian suppliers, with Lawrence Walter accounting for 83.0% of imports.
Bpost International EMC — Import Portfolio & Supplier Network
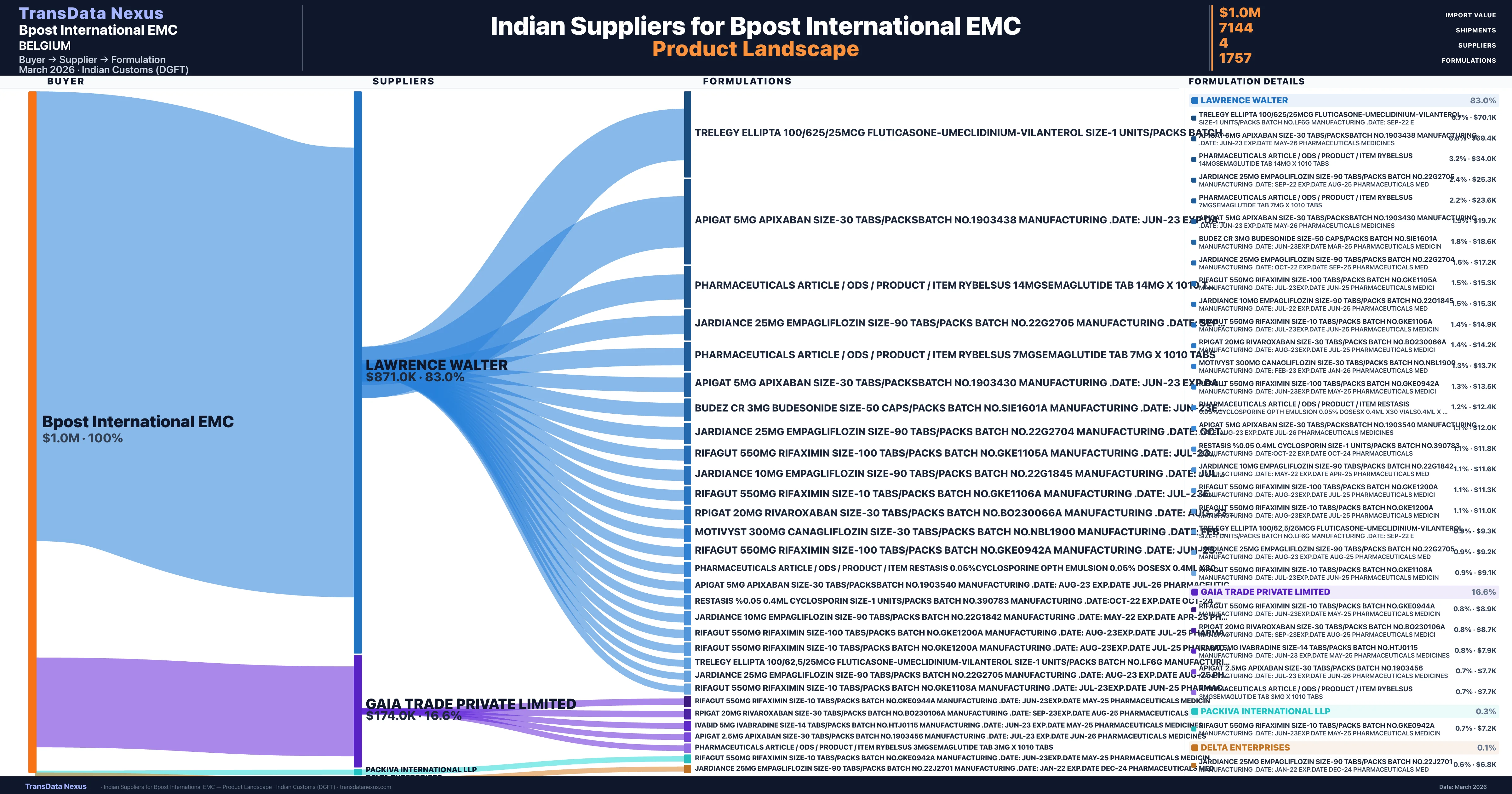
Who Are the Verified Indian Suppliers to Bpost International EMC?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Lawrence Walter | $871.0K | 5,051 | 83.0% |
| Gaia Trade Private Limited | $174.0K | 1,097 | 16.6% |
| Packiva International Llp | $2.9K | 609 | 0.3% |
| Delta Enterprises | $1.1K | 387 | 0.1% |
Bpost International EMC sources from 4 verified Indian suppliers across 1,757 distinct formulations. The sourcing is highly concentrated — Lawrence Walter accounts for 83.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Bpost International EMC Import?
| Formulation | Value | Ships |
|---|---|---|
| Trelegy ellipta 100/625/25mcg fluticasone-umeclidinium-vilanterol size-1 units/packs batch no.lf6g manufacturing .date: sep-22 | $70.1K | 19 |
| Apigat 5MG apixaban size-30 tabs/packsbatch no.1903438 manufacturing .date: jun-23 exp.date may-26 pharmaceuticals | $69.4K | 23 |
| Pharmaceuticals article / ods / product / item rybelsus 14mgsemaglutide TAB 14MG x 1010 | $34.0K | 16 |
| Jardiance 25MG empagliflozin size-90 tabs/packs batch no.22g2705 manufacturing .date: sep-22 exp.date aug-25 pharmaceuticals | $25.3K | 9 |
| Pharmaceuticals article / ods / product / item rybelsus 7mgsemaglutide TAB 7MG x 1010 | $23.6K | 24 |
| Apigat 5MG apixaban size-30 tabs/packsbatch no.1903430 manufacturing .date: jun-23 exp.date may-26 pharmaceuticals | $19.7K | 7 |
| Budez cr 3MG budesonide size-50 CAPS/packs batch no.sie1601a manufacturing .date: jun-23exp.date mar-25 pharmaceuticals | $18.6K | 30 |
| Jardiance 25MG empagliflozin size-90 tabs/packs batch no.22g2704 manufacturing .date: oct-22 exp.date sep-25 pharmaceuticals | $17.2K | 5 |
| Rifagut 550MG rifaximin size-100 tabs/packs batch no.gke1105a manufacturing .date: jul-23exp.date jun-25 pharmaceuticals | $15.3K | 9 |
| Jardiance 10MG empagliflozin size-90 tabs/packs batch no.22g1845 manufacturing .date: jul-22 exp.date jun-25 pharmaceuticals | $15.3K | 15 |
| Rifagut 550MG rifaximin size-10 tabs/packs batch no.gke1106a manufacturing .date: jul-23exp.date jun-25 pharmaceuticals | $14.9K | 9 |
| Rpigat 20MG rivaroxaban size-30 tabs/packs batch no.bo230066a manufacturing .date: aug-23exp.date jul-25 pharmaceuticals | $14.2K | 15 |
| Motivyst 300MG canagliflozin size-30 tabs/packs batch no.nbl1900 manufacturing .date: feb-23 exp.date jan-26 pharmaceuticals | $13.7K | 28 |
| Rifagut 550MG rifaximin size-100 tabs/packs batch no.gke0942a manufacturing .date: jun-23exp.date may-25 pharmaceuticals | $13.5K | 9 |
| Pharmaceuticals article / ods / product / item restasis 0.05%cyclosporine opth emulsion 0.05% dosesx 0.4ML x30 vials0.4ML x 30 | $12.4K | 15 |
Bpost International EMC imports 1,757 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Bpost International EMC Import?
Top Products by Import Value
Bpost International EMC Therapeutic Categories — 2 Specializations
Bpost International EMC imports across 2 therapeutic categories, with Diabetes & Endocrine (56.7%), Advanced Diabetes Medications (43.3%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Diabetes & Endocrine
1 products · 56.7% · $115.1K
Advanced Diabetes Medications
2 products · 43.3% · $87.7K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Empagliflozin | Diabetes & Endocrine | $115.1K | 184 | 1.7% | 9 |
| 2 | Semaglutide | Advanced Diabetes Medications | $70.5K | 73 | 1.4% | 13 |
| 3 | Canagliflozin | Advanced Diabetes Medications | $17.2K | 63 | 1.9% | 11 |
Bpost International EMC imports 3 pharmaceutical products across 2 categories into Belgium totaling $202.9K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Bpost International EMC.
Request DemoBpost International EMC — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Bpost International EMC is a pharmaceutical importer and buyer based in Belgium, specializing in the acquisition of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The company operates as a subsidiary of bpost, Belgium's leading postal operator, which has diversified its services to include logistics and supply chain solutions. Bpost International EMC plays a pivotal role in Belgium's pharmaceutical distribution network by sourcing and importing essential medications to meet domestic demand.
Headquartered in Brussels, Belgium, Bpost International EMC leverages bpost's extensive logistics infrastructure to facilitate the efficient importation and distribution of pharmaceutical products. This strategic positioning enables the company to ensure timely delivery and maintain a steady supply of critical medications across the country. By integrating pharmaceutical importation with logistics expertise, Bpost International EMC contributes significantly to the healthcare sector's stability and responsiveness in Belgium.
2Distribution Network
Bpost International EMC utilizes bpost's extensive network of facilities and transportation services to manage its pharmaceutical distribution operations. While specific warehouse locations are not publicly disclosed, the company benefits from bpost's nationwide presence, ensuring comprehensive coverage across Belgium. This integration allows for efficient logistics capabilities, including secure storage, inventory management, and timely delivery of pharmaceutical products to various stakeholders within the healthcare system.
The geographic coverage extends beyond Belgium, facilitated by bpost's international logistics partnerships and infrastructure. This broader reach enables Bpost International EMC to source pharmaceutical products from a diverse range of suppliers, including those in India, and distribute them effectively within Belgium. The synergy between Bpost International EMC and bpost's logistics network underscores the company's commitment to maintaining a resilient and responsive pharmaceutical supply chain.
3Industry Role
Bpost International EMC functions as a pharmaceutical importer and distributor within Belgium's healthcare supply chain. Its primary role involves sourcing finished pharmaceutical formulations from international suppliers, such as those in India, and ensuring their availability in the Belgian market. By importing a range of medications, including those for diabetes and endocrine disorders, the company addresses specific therapeutic needs within the Belgian population.
While not a primary wholesaler or parallel importer, Bpost International EMC's activities are crucial for maintaining the diversity and availability of pharmaceutical products in Belgium. Its operations support the healthcare system by providing access to a broader spectrum of medications, thereby enhancing treatment options for patients. The company's strategic sourcing and distribution practices contribute to the overall resilience and adaptability of Belgium's pharmaceutical supply chain.
Supplier Relationship Intelligence — Bpost International EMC
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Bpost International EMC's sourcing strategy exhibits a high degree of concentration, with a significant majority of its pharmaceutical imports originating from a single supplier, LAWRENCE WALTER. This supplier accounted for $871K USD in imports, representing 83.0% of the total import value, and 5051 shipments, comprising 83.0% of the total shipments. Such a concentrated sourcing approach can present both strategic advantages and potential risks.
The reliance on a primary supplier may offer benefits such as streamlined logistics, consistent product quality, and favorable pricing agreements. However, this dependency also introduces vulnerabilities, particularly if the supplier faces operational disruptions, regulatory challenges, or geopolitical issues that could impact the supply chain. The absence of alternative suppliers for the majority of imports suggests a need for strategic diversification to mitigate potential risks associated with single-source dependency.
The shipment data indicates a stable relationship with LAWRENCE WALTER, given the high volume and frequency of transactions. Nonetheless, the lack of significant engagement with other suppliers, such as GAIA TRADE PRIVATE LIMITED, PACKIVA INTERNATIONAL LLP, and DELTA ENTERPRISES, highlights a potential area for strategic development. Exploring and establishing relationships with additional suppliers could enhance supply chain resilience and provide leverage in negotiations, thereby strengthening Bpost International EMC's position in the pharmaceutical import sector.
2Supply Chain Resilience
Bpost International EMC's supply chain resilience is closely tied to its sourcing practices, particularly the heavy reliance on LAWRENCE WALTER for the majority of its pharmaceutical imports. This concentrated sourcing strategy may pose challenges in maintaining a consistent and uninterrupted supply of medications, especially if the primary supplier encounters operational or regulatory issues.
The company's limited engagement with alternative suppliers suggests a need for strategic diversification to enhance supply chain robustness. By identifying and establishing relationships with additional suppliers, Bpost International EMC can mitigate risks associated with single-source dependency and ensure a more stable and reliable supply of pharmaceutical products. Diversifying the supplier base would also provide flexibility in responding to market fluctuations and regulatory changes, thereby strengthening the company's overall supply chain resilience.
3Strategic Implications
Bpost International EMC's concentrated sourcing strategy, primarily dependent on LAWRENCE WALTER, has significant strategic implications for its competitive positioning in the Belgian pharmaceutical import market. While this approach may offer operational efficiencies and cost benefits, it also exposes the company to potential risks associated with supplier disruptions, regulatory changes, or geopolitical factors affecting the primary supplier.
For Indian pharmaceutical exporters seeking to become alternative suppliers to Bpost International EMC, there is a clear opportunity to diversify the company's supplier base. By offering competitive pricing, ensuring consistent product quality, and demonstrating reliability in meeting regulatory standards, Indian exporters can position themselves as viable partners. Establishing such partnerships would not only enhance Bpost International EMC's supply chain resilience but also provide Indian exporters with access to the Belgian market, thereby expanding their international footprint.
Importing Pharmaceuticals into Belgium — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Belgium
1Regulatory Authority & Framework
In Belgium, the Federal Agency for Medicines and Health Products (FAMHP) serves as the primary regulatory authority overseeing the importation, distribution, and marketing of medicinal products. The FAMHP is responsible for ensuring that all pharmaceutical products meet the required standards of quality, safety, and efficacy before they are authorized for use within the Belgian market.
The regulatory framework governing pharmaceutical imports into Belgium is primarily outlined in the Royal Decree of 14 December 2006 on medicinal products for human use. This decree establishes the procedures and requirements for the importation of medicinal products, including the necessity for import authorizations, compliance with Good Manufacturing Practice (GMP) standards, and adherence to labeling and packaging regulations. The FAMHP enforces these regulations to maintain the integrity of the pharmaceutical supply chain and protect public health.
2Import Licensing & GMP
Import licensing in Belgium is a critical component of the pharmaceutical regulatory framework. Under Articles 31 and 34 of the Royal Decree of 6 September 2017, any importation of medicinal products, including those from India, requires prior authorization from the FAMHP. Each import authorization is specific to the product and is valid for six months. This process ensures that all imported medicinal products comply with Belgian standards and are safe for patient use.
Compliance with Good Manufacturing Practice (GMP) is mandatory for all pharmaceutical products imported into Belgium. The FAMHP recognizes GMP certifications from the European Union, the World Health Organization (WHO), and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Indian pharmaceutical manufacturers seeking to export to Belgium must ensure that their facilities are GMP-compliant and that their products meet the quality standards set by these recognized bodies. Additionally, wholesalers must obtain a wholesale distribution authorization from the FAMHP, which involves demonstrating compliance with storage, handling, and distribution requirements.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality, safety, and efficacy. The FAMHP conducts these tests to ensure that all medicinal products meet the required standards before they are distributed within Belgium. Stability studies are also essential to confirm that products maintain their quality throughout their shelf life under various storage conditions.
Labeling requirements stipulate that all imported medicinal products must have packaging and labeling in one of Belgium's official languages—Dutch, French, or German. This ensures that patients and healthcare professionals can access accurate and comprehensible information about the product. Serialization mandates are in place to enhance traceability and prevent counterfeit products from entering the supply chain. These measures are part of the FAMHP's efforts to maintain the integrity and safety of the pharmaceutical market in Belgium.
4Recent Regulatory Changes
Between 2024 and 2026, Belgium implemented several legislative measures affecting the importation of medicinal products. Notably, on 1 November 2024, Article 105/1 of the Royal Decree of 14 December 2006 came into force. This provision allows wholesalers to import medicinal products not authorized in Belgium if they meet a special need due to critical unavailability. The FAMHP determines whether an unavailability is critical and adds it to the list of critical unavailabilities for which imports from other European Union/European Economic Area (EEA) member states are considered a possible solution. (faag.be)
Additionally, on 10 December 2025, a new Royal Decree was enacted, fundamentally changing the rules on parallel distribution and parallel import of medicinal products for human use in Belgium. Effective from 11 December 2025, this decree simplifies administrative steps and provides more transparency in the importation process. (faag.be)
Bpost International EMC — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Bpost International EMC's focus on importing medications for
Frequently Asked Questions — Bpost International EMC
What products does Bpost International EMC import from India?
Bpost International EMC imports 3 pharmaceutical products across 2 categories. Top imports: Empagliflozin ($115.1K), Semaglutide ($70.5K), Canagliflozin ($17.2K).
Who supplies pharmaceuticals to Bpost International EMC from India?
Bpost International EMC sources from 4 verified Indian suppliers. The primary supplier is Lawrence Walter (83.0% of imports, $871.0K).
What is Bpost International EMC's total pharmaceutical import value?
Bpost International EMC's total pharmaceutical import value from India is $202.9K, based on 320 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Bpost International EMC focus on?
Bpost International EMC imports across 2 categories. The largest: Diabetes & Endocrine (56.7%), Advanced Diabetes Medications (43.3%).
Get Full Bpost International EMC Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Bpost International EMC identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Bpost International EMC's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 320 individual customs records matching Bpost International EMC.
- 5.Supplier Verification: Bpost International EMC sources from 4 verified Indian suppliers across 1,757 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.