Bells Healthcare
Pharmaceutical Importer · United Kingdom · Respiratory Focus · $11.9M Total Trade · DGFT Verified
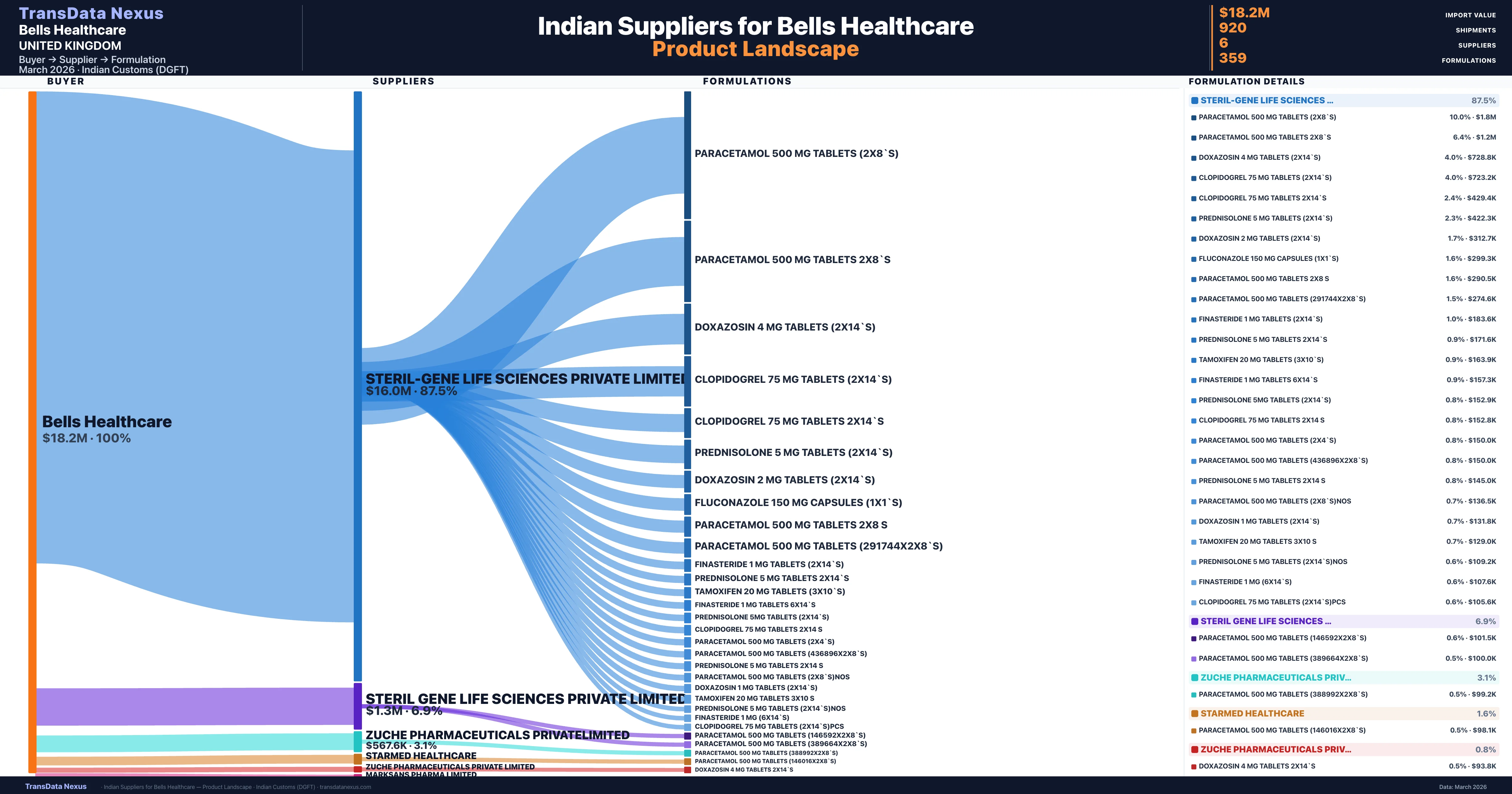
Bells Healthcare is a pharmaceutical importer based in United Kingdom with a total trade value of $11.9M across 4 products in 4 therapeutic categories. Based on 256 verified import shipments from Indian Customs (DGFT) records, Bells Healthcare is the #1 buyer in 1 product including Prednisolone. Bells Healthcare sources from 6 verified Indian suppliers, with Steril-gene Life Sciences Private Limited accounting for 87.5% of imports.
Bells Healthcare — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Bells Healthcare?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Steril-gene Life Sciences Private Limited | $16.0M | 797 | 87.5% |
| Steril Gene Life Sciences Private Limited | $1.3M | 42 | 6.9% |
| Zuche Pharmaceuticals Privatelimited | $567.6K | 45 | 3.1% |
| Starmed Healthcare | $293.6K | 29 | 1.6% |
| Zuche Pharmaceuticals Private Limited | $147.2K | 6 | 0.8% |
| Marksans Pharma Limited | $0 | 1 | 0.0% |
Bells Healthcare sources from 6 verified Indian suppliers across 359 distinct formulations. The sourcing is highly concentrated — Steril-gene Life Sciences Private Limited accounts for 87.5% of total imports, indicating a strategic single-source relationship.
What Formulations Does Bells Healthcare Import?
| Formulation | Value | Ships |
|---|---|---|
| Paracetamol 500 MG tablets (2x8`s) | $1.8M | 53 |
| Paracetamol 500 MG tablets | $1.2M | 40 |
| Doxazosin 4 MG tablets (2x14`s) | $728.8K | 42 |
| Clopidogrel 75 MG tablets (2x14`s) | $723.2K | 21 |
| Clopidogrel 75 MG tablets | $429.4K | 10 |
| Prednisolone 5 MG tablets (2x14`s) | $422.3K | 14 |
| Doxazosin 2 MG tablets (2x14`s) | $312.7K | 27 |
| Fluconazole 150 MG capsules (1x1`s) | $299.3K | 18 |
| Paracetamol 500 MG tablets 2x8 | $290.5K | 7 |
| Paracetamol 500 MG tablets (291744x2x8`s) | $274.6K | 9 |
| Finasteride 1 MG tablets (2x14`s) | $183.6K | 13 |
| Prednisolone 5 MG tablets | $171.6K | 6 |
| Tamoxifen 20 MG tablets (3x10`s) | $163.9K | 5 |
| Finasteride 1 MG tablets | $157.3K | 9 |
| Prednisolone 5MG tablets (2x14`s) | $152.9K | 4 |
Bells Healthcare imports 359 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Bells Healthcare Import?
Top Products by Import Value
Bells Healthcare Therapeutic Categories — 4 Specializations
Bells Healthcare imports across 4 therapeutic categories, with Respiratory (40.6%), Analgesics & Antipyretics (28.2%), Cardiovascular (27.6%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Respiratory
1 products · 40.6% · $4.8M
Analgesics & Antipyretics
1 products · 28.2% · $3.4M
Cardiovascular
1 products · 27.6% · $3.3M
Advanced Oncology
1 products · 3.5% · $416.4K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Prednisolone | Respiratory | $4.8M | 97 | 5.1% | 1 |
| 2 | Paracetamol | Analgesics & Antipyretics | $3.4M | 80 | 0.9% | 10 |
| 3 | Clopidogrel | Cardiovascular | $3.3M | 66 | 1.3% | 3 |
| 4 | Tamoxifen | Advanced Oncology | $416.4K | 13 | 2.8% | 5 |
Bells Healthcare imports 4 pharmaceutical products across 4 categories into United Kingdom totaling $11.9M. The company is the #1 buyer for 1 product: Prednisolone.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Bells Healthcare.
Request DemoBells Healthcare — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Bells Healthcare, officially known as Bells Sons & Co (Druggist) Ltd, is a prominent UK-based manufacturer specializing in over-the-counter pharmaceuticals. With a legacy spanning over 170 years, the company has established itself as a key player in the UK's pharmaceutical sector. Headquartered in Southport, England, Bells Healthcare operates a state-of-the-art manufacturing facility that adheres to Good Manufacturing Practice (GMP) and is approved by the Medicines and Healthcare products Regulatory Agency (MHRA). This facility boasts nine flexible production lines capable of producing over 20 million units annually, underscoring the company's capacity to meet diverse market demands.
The company's product portfolio encompasses more than 150 lines, with nearly 100 MHRA-registered licenses, reflecting its commitment to quality and regulatory compliance. Bells Healthcare supplies both branded and own-brand products to leading UK supermarkets and exports to over 65 international markets, including regions in Africa, Asia, Europe, the Middle East, and Australia. This extensive reach highlights the company's significant role in the global pharmaceutical distribution network.
2Distribution Network
Bells Healthcare's distribution network is robust and expansive, ensuring efficient delivery of its products both domestically and internationally. The company's UK-based manufacturing facility in Southport serves as the central hub for production and distribution. From this location, products are dispatched to various UK wholesalers, international distributors, and own-brand providers. The facility's capacity to produce over 20 million units annually, coupled with nine dedicated production lines, enables Bells Healthcare to accommodate large-scale orders and tight lead times, ensuring dependable deliveries.
Internationally, Bells Healthcare exports to over 65 markets worldwide, including regions in Africa, Asia, Europe, the Middle East, and Australia. This global distribution network underscores the company's capability to meet diverse market demands and adapt to various regulatory environments. The company's commitment to quality and compliance with international standards has established it as a trusted partner in the global pharmaceutical supply chain.
3Industry Role
In the United Kingdom's pharmaceutical supply chain, Bells Healthcare functions primarily as a manufacturer and wholesaler. The company produces a wide range of over-the-counter pharmaceutical products, including analgesics, allergy relief, and children's medicines, which are then distributed to UK wholesalers, international distributors, and own-brand providers. This role positions Bells Healthcare as a key supplier in the UK's pharmaceutical market, catering to both domestic and international demand.
While Bells Healthcare does not operate as a parallel importer or hospital supplier, its extensive manufacturing capabilities and distribution network enable it to serve a broad spectrum of the pharmaceutical supply chain. The company's adherence to Good Manufacturing Practice (GMP) and MHRA regulations further solidifies its position as a reliable and compliant entity within the industry.
Supplier Relationship Intelligence — Bells Healthcare
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Bells Healthcare's sourcing strategy exhibits a high degree of concentration, with a significant portion of its pharmaceutical imports originating from India. Between 2022 and 2026, the company imported finished pharmaceutical formulations valued at $11.9 million USD from India, encompassing 256 shipments across four therapeutic categories. The top five imported products include Prednisolone ($4.8 million, 5.1% share), Paracetamol ($3.4 million, 0.9% share), Clopidogrel ($3.3 million, 1.3% share), and Tamoxifen ($416,000, 2.8% share). Notably, Bells Healthcare is the leading buyer of Prednisolone, indicating a strategic focus on this product.
The supplier base from India comprises six unique entities, with STERIL-GENE LIFE SCIENCES PRIVATE LIMITED accounting for 87.5% of the total import value. This heavy reliance on a single supplier for the majority of imports suggests a strategic partnership aimed at ensuring consistent quality and supply. However, such concentration also introduces potential risks, including supply chain disruptions due to geopolitical issues, regulatory changes, or operational challenges within the supplier's facilities.
The stability of the relationship with STERIL-GENE LIFE SCIENCES PRIVATE LIMITED is crucial for Bells Healthcare's operations. The consistent volume of shipments over the years indicates a stable and ongoing partnership. Nonetheless, the company may consider diversifying its supplier base to mitigate risks associated with over-reliance on a single source, thereby enhancing the resilience of its supply chain.
2Supply Chain Resilience
Bells Healthcare's supply chain resilience is closely tied to its sourcing strategy from India, particularly its dependence on STERIL-GENE LIFE SCIENCES PRIVATE LIMITED. While this supplier relationship has provided consistent supply, the concentration poses potential risks, including supply chain disruptions due to geopolitical issues, regulatory changes, or operational challenges within the supplier's facilities.
To enhance supply chain resilience, Bells Healthcare may consider diversifying its supplier base by engaging additional manufacturers from India or other countries. This approach would mitigate risks associated with over-reliance on a single source and provide alternative options in case of disruptions. Additionally, expanding the diversity of imported formulations beyond the current four therapeutic categories could further strengthen the supply chain by reducing dependency on specific product lines.
Ensuring that all suppliers comply with international regulatory standards, such as Good Manufacturing Practice (GMP) and MHRA regulations, is essential for maintaining product quality and safety. Regular audits and quality assessments of suppliers can help identify potential issues early and facilitate proactive measures to address them, thereby enhancing the overall resilience of Bells Healthcare's supply chain.
3Strategic Implications
Bells Healthcare's sourcing pattern, characterized by a high concentration of imports from India and a significant reliance on STERIL-GENE LIFE SCIENCES PRIVATE LIMITED, has several strategic implications. The company's position as the leading buyer of Prednisolone underscores its strong market presence and potential influence within this therapeutic segment. This dominance can be leveraged to negotiate favorable terms with suppliers and strengthen its competitive position in the UK market.
For Indian exporters, Bells Healthcare's sourcing strategy presents an opportunity to establish or deepen partnerships, particularly in the Prednisolone segment. However, the concentration of imports from a single supplier indicates that Bells Healthcare values stability and reliability in its supplier relationships. Prospective suppliers would need to demonstrate consistent product quality, adherence to regulatory standards, and the capacity to meet Bells Healthcare's volume requirements to be considered viable partners.
Importing Pharmaceuticals into United Kingdom — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United Kingdom
1Regulatory Authority & Framework
In the United Kingdom, the Medicines and Healthcare products Regulatory Agency (MHRA) serves as the primary regulatory authority overseeing the safety, efficacy, and quality of medicines and medical devices. The MHRA operates under the Human Medicines Regulations 2012, which implement European Union directives and establish the legal framework for the authorization, manufacture, and distribution of medicinal products in the UK. (gov.uk)
The MHRA's responsibilities include granting marketing authorizations for medicinal products, ensuring compliance with Good Manufacturing Practice (GMP), and conducting inspections of manufacturing and distribution facilities. The agency also monitors the safety of medicines through pharmacovigilance activities, including the collection and analysis of adverse drug reactions. For imported medicines, the MHRA requires that a Qualified Person (QP) certifies that the product has been manufactured and tested according to the marketing authorization and GMP standards before it can be released to the market. (gov.uk)
For Indian pharmaceutical exporters aiming to supply the UK market, understanding and complying with the MHRA's regulatory framework is essential. This includes ensuring that products meet the MHRA's quality standards, obtaining the necessary marketing authorizations, and adhering to GMP requirements. Additionally, staying informed about any updates or changes to MHRA guidelines, such as those related to sourcing medicines for the UK market, is crucial for maintaining compliance and facilitating successful market entry.
2Import Licensing & GMP
To import human medicines into the United Kingdom, entities must obtain specific licenses from the MHRA. The primary licenses required include:
- Wholesale Distribution Licence (WDA(H)): This license authorizes the holder to distribute medicinal products within the UK.
- Manufacturer’s Licence: This license permits the holder to manufacture medicinal products, including importation and distribution operations.
- Marketing Authorisation (MA): This authorization is required before a medicine can be sold in the UK, ensuring that the product meets the necessary safety, efficacy, and quality standards.
For medicines containing controlled substances, additional authorization from the Home Office is necessary. It's important to note that the MHRA has updated its guidance on sourcing medicines for the UK market, reflecting new UK-wide licensing arrangements that came into effect on 1 January 2025 as part of the Windsor Framework.
Regarding Good Manufacturing Practice (GMP), the MHRA requires that all manufacturers and importers of medicinal
Frequently Asked Questions — Bells Healthcare
What products does Bells Healthcare import from India?
Bells Healthcare imports 4 pharmaceutical products across 4 categories. Top imports: Prednisolone ($4.8M), Paracetamol ($3.4M), Clopidogrel ($3.3M), Tamoxifen ($416.4K).
Who supplies pharmaceuticals to Bells Healthcare from India?
Bells Healthcare sources from 6 verified Indian suppliers. The primary supplier is Steril-gene Life Sciences Private Limited (87.5% of imports, $16.0M).
What is Bells Healthcare's total pharmaceutical import value?
Bells Healthcare's total pharmaceutical import value from India is $11.9M, based on 256 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Bells Healthcare focus on?
Bells Healthcare imports across 4 categories. The largest: Respiratory (40.6%), Analgesics & Antipyretics (28.2%), Cardiovascular (27.6%).
Get Full Bells Healthcare Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Bells Healthcare identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Bells Healthcare's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 256 individual customs records matching Bells Healthcare.
- 5.Supplier Verification: Bells Healthcare sources from 6 verified Indian suppliers across 359 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.