Atlanta Medicare CO. Ltd
Pharmaceutical Importer · Thailand · Advanced Oncology Focus · $575.1K Total Trade · DGFT Verified
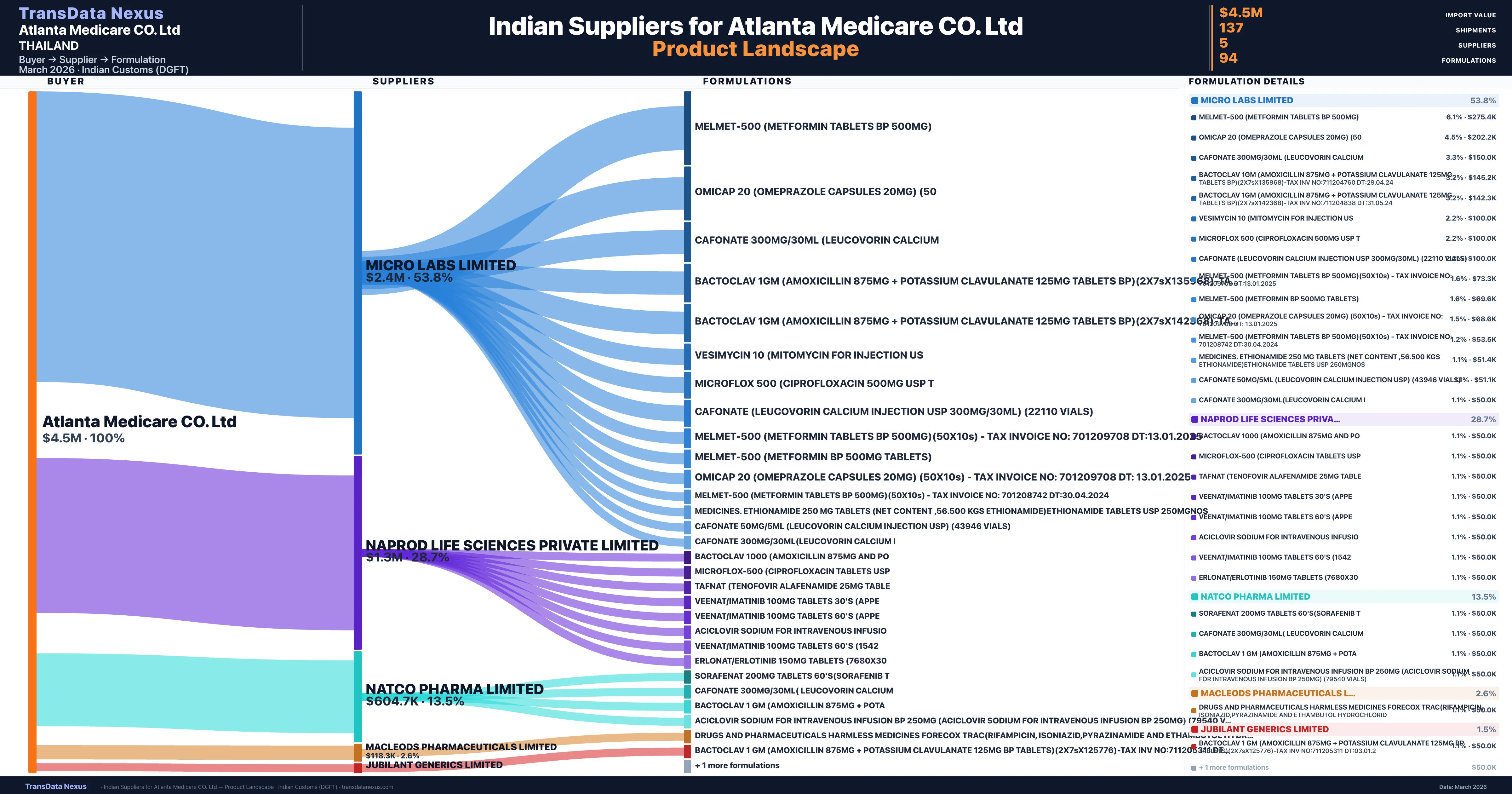
Atlanta Medicare CO. Ltd is a pharmaceutical importer based in Thailand with a total trade value of $575.1K across 3 products in 1 therapeutic categories. Based on 17 verified import shipments from Indian Customs (DGFT) records, Atlanta Medicare CO. Ltd is the #1 buyer in 1 product including Erlotinib. Atlanta Medicare CO. Ltd sources from 5 verified Indian suppliers, with Micro Labs Limited accounting for 53.8% of imports.
Atlanta Medicare CO. Ltd — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Atlanta Medicare CO. Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Micro Labs Limited | $2.4M | 75 | 53.8% |
| Naprod Life Sciences Private Limited | $1.3M | 36 | 28.7% |
| Natco Pharma Limited | $604.7K | 17 | 13.5% |
| Macleods Pharmaceuticals Limited | $118.3K | 7 | 2.6% |
| Jubilant Generics Limited | $66.1K | 2 | 1.5% |
Atlanta Medicare CO. Ltd sources from 5 verified Indian suppliers across 94 distinct formulations. The supply base is diversified across 5 suppliers, reducing single-source dependency risk.
What Formulations Does Atlanta Medicare CO. Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Melmet-500 (metformin tablets BP 500MG) | $275.4K | 9 |
| Omicap 20 (omeprazole capsules 20MG) | $202.2K | 8 |
| Cafonate 300MG/30ML (leucovorin | $150.0K | 3 |
| Bactoclav 1GM (amoxicillin 875MG + potassium clavulanate 125MG tablets BP)(2x7sx135968)-tax inv no:711204760 | $145.2K | 3 |
| Bactoclav 1GM (amoxicillin 875MG + potassium clavulanate 125MG tablets BP)(2x7sx142368)-tax inv no:711204838 | $142.3K | 3 |
| Vesimycin 10 (mitomycin for injection | $100.0K | 2 |
| Microflox 500 (ciprofloxacin 500MG USP | $100.0K | 2 |
| Cafonate (leucovorin calcium injection USP 300MG/30ML) (22110 vials) | $100.0K | 2 |
| Melmet-500 (metformin tablets BP 500MG)(50x10s) - tax invoice no: 701209708 | $73.3K | 2 |
| Melmet-500 (metformin BP 500MG tablets) | $69.6K | 3 |
| Omicap 20 (omeprazole capsules 20MG) (50x10s) - tax invoice no: 701209708 dt | $68.6K | 2 |
| Melmet-500 (metformin tablets BP 500MG)(50x10s) - tax invoice no: 701208742 | $53.5K | 3 |
| Medicines. ethionamide 250 MG tablets (net content ,56.500 kgs ethionamide)ethionamide tablets USP | $51.4K | 3 |
| Cafonate 50MG/5ML (leucovorin calcium injection USP) (43946 vials) | $51.1K | 3 |
| Cafonate 300MG/30ML(leucovorin calcium | $50.0K | 1 |
Atlanta Medicare CO. Ltd imports 94 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Atlanta Medicare CO. Ltd Import?
Top Products by Import Value
Atlanta Medicare CO. Ltd Therapeutic Categories — 1 Specializations
Atlanta Medicare CO. Ltd imports across 1 therapeutic categories, with Advanced Oncology (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Oncology
3 products · 100.0% · $575.1K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Mitomycin | Advanced Oncology | $375.1K | 13 | 2.8% | 7 |
| 2 | Erlotinib | Advanced Oncology | $100.0K | 2 | 3.7% | 1 |
| 3 | Sorafenib | Advanced Oncology | $100.0K | 2 | 0.6% | 4 |
Atlanta Medicare CO. Ltd imports 3 pharmaceutical products across 1 categories into Thailand totaling $575.1K. The company is the #1 buyer for 1 product: Erlotinib.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Atlanta Medicare CO. Ltd.
Request DemoAtlanta Medicare CO. Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Atlanta Medicare Company Limited, established in 2007, is a prominent pharmaceutical importer and distributor based in Bangkok, Thailand. The company specializes in the distribution and marketing of innovative and high-quality finished dosage formulations and medical devices, catering to both acute and chronic ailments prevalent among the Thai population. Their mission emphasizes delivering dependable services through teamwork, aiming to serve as the first-choice marketing partner for leading global pharmaceutical, biotech, medical device, and diagnostics companies seeking to connect with Thailand's medical and pharmaceutical community. (atlantamedicare.co.th)
Headquartered at 9 Soi Ramindra 19 Yeak 1, Anusawaree, Bangkhen, Bangkok 10220, Thailand, Atlanta Medicare operates with a vision to be an innovator in the Thai pharmaceutical industry. The company strives to provide the Thai population with the best available treatments worldwide, reflecting its commitment to enhancing healthcare standards in the region. (atlantamedicare.co.th)
2Distribution Network
Atlanta Medicare's distribution network is strategically designed to ensure efficient delivery of pharmaceutical products across Thailand. While specific warehouse locations are not publicly disclosed, the company's headquarters in Bangkok serves as a central hub for operations. The company emphasizes exceptional service and teamwork, which likely extends to its logistics capabilities, ensuring timely and reliable distribution of medical products throughout the country. (atlantamedicare.co.th)
3Industry Role
In Thailand's pharmaceutical supply chain, Atlanta Medicare Company Limited functions primarily as a pharmaceutical importer and distributor. By sourcing innovative and quality finished dosage formulations and medical devices from global manufacturers, the company plays a crucial role in introducing and supplying these products to the Thai market. This position enables Atlanta Medicare to bridge the gap between international pharmaceutical innovations and local healthcare needs, contributing significantly to the availability of advanced medical treatments in Thailand. (atlantamedicare.co.th)
Supplier Relationship Intelligence — Atlanta Medicare CO. Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Atlanta Medicare's sourcing strategy exhibits a high degree of concentration, with a portfolio focused on a limited number of products and suppliers. The company's total import value from India amounts to $575,000 USD across 17 shipments, encompassing three products within a single therapeutic category—Advanced Oncology. The top three products imported are Mitomycin ($375,000, 2.8% share), Erlotinib ($100,000, 3.7% share), and Sorafenib ($100,000, 0.6% share). This concentration indicates a strategic choice to specialize in a niche market segment, potentially allowing for deeper expertise and stronger relationships within the oncology domain.
The shipment data reveals a stable relationship with key Indian suppliers, including MICRO LABS LIMITED, NAPROD LIFE SCIENCES PRIVATE LIMITED, and NATCO PHARMA LIMITED. The consistent importation of specific products from these suppliers suggests a reliable and ongoing partnership, which is essential for maintaining a steady supply chain and ensuring product availability in the Thai market.
2Supply Chain Resilience
Atlanta Medicare's supply chain resilience is supported by its partnerships with established Indian pharmaceutical manufacturers. The company's focus on finished pharmaceutical formulations, rather than raw active pharmaceutical ingredients (APIs), indicates a preference for ready-to-market products, which can streamline the import process and reduce lead times. However, the high concentration of imports from a limited number of suppliers may expose the company to risks associated with supply chain disruptions, such as production delays or regulatory changes affecting these suppliers. To enhance resilience, Atlanta Medicare could consider diversifying its supplier base and exploring alternative sourcing options to mitigate potential risks.
3Strategic Implications
Atlanta Medicare's concentrated sourcing strategy positions the company as a specialized distributor in the oncology therapeutic area within Thailand. This focus allows for the development of deep expertise and strong relationships with key suppliers, potentially leading to favorable terms and priority access to new products. For Indian exporters, this presents an opportunity to establish or strengthen partnerships with Atlanta Medicare by offering high-quality oncology products that align with the company's portfolio. By understanding Atlanta Medicare's specific product needs and regulatory requirements, Indian exporters can tailor their offerings to meet these demands, thereby enhancing their competitive position in the Thai market.
Importing Pharmaceuticals into Thailand — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Thailand
1Regulatory Authority & Framework
In Thailand, the Food and Drug Administration (FDA) under the Ministry of Public Health is the primary regulatory authority overseeing the importation and distribution of pharmaceutical products. The Thai FDA requires that all pharmaceutical products, including finished dosage forms, be registered before they can be imported or marketed within the country. This registration process ensures that imported drugs meet the necessary safety, efficacy, and quality standards. (en.fda.moph.go.th)
The regulatory framework governing pharmaceutical imports in Thailand is comprehensive, encompassing various categories of drugs such as modern medicines, general medicines, traditional medicines, and veterinary medicines. Each category has distinct registration requirements, and the Thai FDA provides detailed guidelines to facilitate compliance. For instance, the registration process for general medicines involves submitting documentation in accordance with the ASEAN Common Technical Dossier (ACTD) or the ICH Common Technical Document (ICH CTD), ensuring harmonization with international standards. (en.fda.moph.go.th)
2Import Licensing & GMP
Importers of pharmaceutical products into Thailand must obtain a license from the Thai FDA. This license is mandatory for entities intending to import or order drugs from another country into Thailand. The application process involves submitting the necessary documentation and complying with the FDA's requirements, which may include demonstrating adherence to Good Manufacturing Practice (GMP) standards. GMP compliance is crucial, as it ensures that pharmaceutical products are consistently produced and controlled according to quality standards. (en.fda.moph.go.th)
The Thai FDA recognizes GMP certificates from various international bodies, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). This recognition facilitates the importation of pharmaceutical products from countries with established GMP standards, provided that the manufacturing facilities meet the FDA's criteria. Additionally, importers must ensure that their wholesale distribution operations are authorized, adhering to the FDA's regulations to maintain the integrity of the pharmaceutical supply chain. (en.fda.moph.go.th)
3Quality & Labeling
Imported pharmaceutical products in Thailand are subject to stringent quality control measures, including batch testing and stability assessments, to ensure their safety and efficacy. These tests are conducted in accordance with the Thai FDA's standards and guidelines, which may align with international practices. The labeling of pharmaceutical products must comply with the FDA's requirements, which typically include information such as the product name, active ingredients, dosage form, strength, batch number, manufacturing and expiry dates, storage conditions, and the manufacturer's details. Labels must be fully translated into the Thai language to ensure clear communication with healthcare professionals and patients. (en.sme.go.th)
Serialization mandates may be implemented to enhance traceability and prevent counterfeit products from entering the market. These mandates require that each unit of a pharmaceutical product be assigned a unique identifier, facilitating monitoring throughout the supply chain. Importers and distributors must stay informed about such requirements and ensure that their products are compliant to maintain market access and uphold public health standards.
4Recent Regulatory Changes
Between 2024 and 2026, Thailand's regulatory landscape for pharmaceutical imports has undergone several changes aimed at enhancing efficiency and safety. The Thai FDA has streamlined the drug registration process, reducing approval times and simplifying documentation requirements to encourage the importation of innovative medicines. Additionally, there has been an increased emphasis on the adoption of international standards, such as EU GMP and PIC/S, to improve the quality of imported pharmaceutical products. These changes reflect Thailand's commitment to aligning its regulatory framework with global best practices, thereby improving the availability and quality of medicines in the country. (en.fda.moph.go.th)
Atlanta Medicare CO. Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Atlanta Medicare's product strategy focuses on the oncology therapeutic area, importing finished pharmaceutical formulations such as Mitomycin, Erlotinib, and Sorafenib. This strategic focus aligns with the growing demand for cancer treatments in Thailand, driven by an aging population and increasing awareness of cancer-related health issues. By concentrating on these specific products, Atlanta Medicare aims to address critical healthcare needs and establish a strong presence in the oncology market segment. (atlantamedicare.co.th)
The market demand for these oncology products is influenced by factors such as the prevalence of cancer, advancements in treatment options, and the availability of innovative therapies. Atlanta Medicare's imports of these products suggest a proactive approach to meeting the evolving needs of the Thai healthcare system and providing patients with access to effective cancer treatments.
2Sourcing Profile
Atlanta Medicare's sourcing strategy is centered on importing finished pharmaceutical formulations from India, focusing on products like Mitomycin, Erlotinib, and Sorafenib. This approach allows the company to offer ready-to-market products, reducing lead times and ensuring a consistent supply of essential medications. India's established pharmaceutical manufacturing industry, known for its adherence to international quality standards, makes it a reliable source for these products. By leveraging India's capabilities, Atlanta Medicare can enhance its product offerings and meet the demands of the Thai market effectively
Frequently Asked Questions — Atlanta Medicare CO. Ltd
What products does Atlanta Medicare CO. Ltd import from India?
Atlanta Medicare CO. Ltd imports 3 pharmaceutical products across 1 categories. Top imports: Mitomycin ($375.1K), Erlotinib ($100.0K), Sorafenib ($100.0K).
Who supplies pharmaceuticals to Atlanta Medicare CO. Ltd from India?
Atlanta Medicare CO. Ltd sources from 5 verified Indian suppliers. The primary supplier is Micro Labs Limited (53.8% of imports, $2.4M).
What is Atlanta Medicare CO. Ltd's total pharmaceutical import value?
Atlanta Medicare CO. Ltd's total pharmaceutical import value from India is $575.1K, based on 17 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Atlanta Medicare CO. Ltd focus on?
Atlanta Medicare CO. Ltd imports across 1 categories. The largest: Advanced Oncology (100.0%).
Get Full Atlanta Medicare CO. Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Atlanta Medicare CO. Ltd identified across shipments using consignee name normalization, aggregating 3 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Atlanta Medicare CO. Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 17 individual customs records matching Atlanta Medicare CO. Ltd.
- 5.Supplier Verification: Atlanta Medicare CO. Ltd sources from 5 verified Indian suppliers across 94 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 3 company name variants from customs records. For current shipment-level data, contact TransData Nexus.