ACE Health Care LDA
Pharmaceutical Importer · Mozambique · Antibiotics Focus · $1.4M Total Trade · DGFT Verified
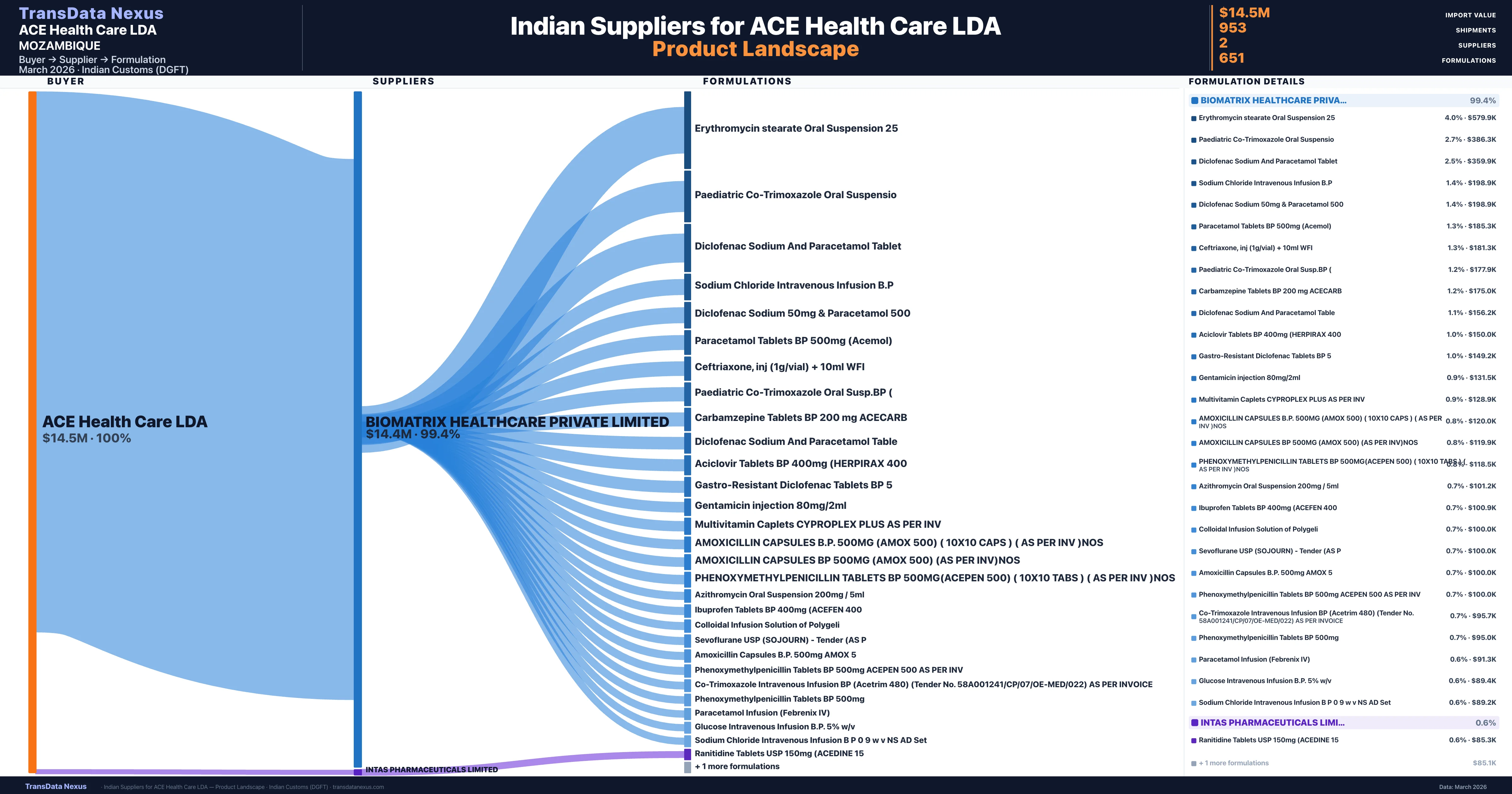
ACE Health Care LDA is a pharmaceutical importer based in Mozambique with a total trade value of $1.4M across 3 products in 3 therapeutic categories. Based on 51 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. ACE Health Care LDA sources from 2 verified Indian suppliers, with Biomatrix Healthcare Private Limited accounting for 99.4% of imports.
ACE Health Care LDA — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to ACE Health Care LDA?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Biomatrix Healthcare Private Limited | $14.4M | 947 | 99.4% |
| Intas Pharmaceuticals Limited | $85.0K | 6 | 0.6% |
ACE Health Care LDA sources from 2 verified Indian suppliers across 651 distinct formulations. The sourcing is highly concentrated — Biomatrix Healthcare Private Limited accounts for 99.4% of total imports, indicating a strategic single-source relationship.
What Formulations Does ACE Health Care LDA Import?
| Formulation | Value | Ships |
|---|---|---|
| Erythromycin stearate oral suspension | $579.9K | 14 |
| Paediatric co-trimoxazole oral | $386.3K | 15 |
| Diclofenac sodium and paracetamol | $359.9K | 8 |
| Sodium chloride intravenous infusion | $198.9K | 7 |
| Diclofenac sodium 50MG & paracetamol | $198.9K | 6 |
| Paracetamol tablets BP 500MG (acemol) | $185.3K | 8 |
| Ceftriaxone, INJ (1g/vial) + 10ML | $181.3K | 4 |
| Paediatric co-trimoxazole oral SUSP.BP | $177.9K | 5 |
| Carbamzepine tablets BP 200 MG | $175.0K | 4 |
| Diclofenac sodium and paracetamol | $156.2K | 4 |
| Aciclovir tablets BP 400MG (herpirax | $150.0K | 3 |
| Gastro-resistant diclofenac tablets BP | $149.2K | 14 |
| Gentamicin injection 80MG/2ML | $131.5K | 6 |
| Multivitamin caplets cyproplex plus as per | $128.9K | 5 |
| Amoxicillin capsules b.p. 500MG (amox 500) ( 10x10 CAPS ) ( as per inv | $120.0K | 3 |
ACE Health Care LDA imports 651 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does ACE Health Care LDA Import?
Top Products by Import Value
ACE Health Care LDA Therapeutic Categories — 3 Specializations
ACE Health Care LDA imports across 3 therapeutic categories, with Antibiotics (87.8%), Gastrointestinal (8.8%), Combination Drugs (3.4%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antibiotics
1 products · 87.8% · $1.2M
Gastrointestinal
1 products · 8.8% · $125.2K
Combination Drugs
1 products · 3.4% · $47.8K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Erythromycin | Antibiotics | $1.2M | 38 | 1.6% | 4 |
| 2 | Ranitidine | Gastrointestinal | $125.2K | 11 | 1.0% | 17 |
| 3 | Ibuprofen Paracetamol | Combination Drugs | $47.8K | 2 | 2.5% | 6 |
ACE Health Care LDA imports 3 pharmaceutical products across 3 categories into Mozambique totaling $1.4M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for ACE Health Care LDA.
Request DemoACE Health Care LDA — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
ACE Health Care LDA is a prominent pharmaceutical importer and distributor based in Maputo, Mozambique. Established in March 2014, the company has rapidly expanded its operations to include branches in Beira and Nampula, thereby enhancing its national reach. ACE Health Care LDA specializes in the importation and distribution of a comprehensive range of health products, including human medicines, surgical and orthopedic materials, and hospital equipment. Their clientele encompasses public and private hospitals, clinics, pharmacies, non-governmental organizations, and international aid programs.
The company's mission is to address the urgent need for quality medical supplies in Mozambique by ensuring the timely and efficient delivery of essential health products across the country. ACE Health Care LDA's commitment to quality is evident in its adherence to international standards, with all products complying with safety and efficacy regulations. The company operates certified warehouses and employs automated systems for temperature and humidity control, ensuring optimal storage conditions for its products.
2Distribution Network
ACE Health Care LDA's distribution network is strategically designed to cover key regions of Mozambique. The central warehouse, located in Matola, is equipped to handle large volumes of goods, facilitating efficient processing and distribution. Regional branches in Beira and Nampula further extend the company's reach, ensuring rapid and reliable delivery of products to the southern, central, and northern parts of the country. This extensive network enables ACE Health Care LDA to meet the diverse needs of its clients, regardless of geographic location.
3Industry Role
In Mozambique's pharmaceutical supply chain, ACE Health Care LDA functions as a primary wholesaler and distributor. By importing a wide array of pharmaceutical products and medical equipment, the company plays a crucial role in bridging the gap between international manufacturers and local healthcare providers. Its operations ensure that essential medicines and medical supplies are accessible to hospitals, clinics, pharmacies, and other healthcare institutions throughout the country. ACE Health Care LDA's extensive product portfolio and distribution capabilities position it as a key player in enhancing the availability and quality of healthcare services in Mozambique.
Supplier Relationship Intelligence — ACE Health Care LDA
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
ACE Health Care LDA's sourcing strategy exhibits a high degree of concentration, with a significant majority of its pharmaceutical imports originating from India. The company's total import value from India stands at $1.4 million USD, encompassing 51 shipments across three therapeutic categories. The top three products imported are Erythromycin ($1.2 million, 1.6% share), Ranitidine ($125,000, 1.0% share), and Ibuprofen Paracetamol ($48,000, 2.5% share). This concentration indicates a strategic choice to leverage India's robust pharmaceutical manufacturing capabilities and competitive pricing.
The relationship with Indian suppliers appears stable, as evidenced by the consistent import volumes and the presence of two verified suppliers: BIOMATRIX HEALTHCARE PRIVATE LIMITED, accounting for 99.4% of shipments, and INTAS PHARMACEUTICALS LIMITED, contributing 0.6%. This dual-supplier model mitigates risks associated with single-source dependency while maintaining a streamlined supply chain. However, the heavy reliance on a single supplier (BIOMATRIX HEALTHCARE PRIVATE LIMITED) could pose challenges in the event of supply disruptions or changes in supplier dynamics.
2Supply Chain Resilience
ACE Health Care LDA's supply chain resilience is primarily anchored in its partnership with BIOMATRIX HEALTHCARE PRIVATE LIMITED, which supplies the vast majority of the company's imported pharmaceutical products. This reliance underscores the importance of maintaining a strong and dependable relationship with this supplier to ensure uninterrupted product availability.
The company's import portfolio comprises 651 unique formulations, indicating a diverse range of products sourced from India. This diversity can help mitigate risks associated with over-reliance on a single product line or therapeutic category. However, the concentration of imports from a single supplier may expose ACE Health Care LDA to potential supply chain disruptions. To enhance resilience, the company could consider diversifying its supplier base and exploring alternative sourcing options.
3Strategic Implications
ACE Health Care LDA's sourcing pattern, characterized by a heavy reliance on a single supplier from India, has several strategic implications. While this approach allows the company to benefit from established relationships and potentially favorable terms, it also exposes ACE Health Care LDA to risks associated with supply chain disruptions, such as delays, quality issues, or geopolitical factors affecting trade.
For Indian exporters, the concentration of ACE Health Care LDA's imports with BIOMATRIX HEALTHCARE PRIVATE LIMITED presents an opportunity to strengthen their position as a preferred supplier. However, to mitigate risks, ACE Health Care LDA may seek to diversify its supplier base, creating opportunities for other Indian pharmaceutical companies to enter the market. Establishing relationships with ACE Health Care LDA could lead to increased market share and a more balanced distribution of supply chain risks.
Importing Pharmaceuticals into Mozambique — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Mozambique
1Regulatory Authority & Framework
The regulation of pharmaceutical products in Mozambique is overseen by the Autoridade Nacional Reguladora de Medicamentos de Moçambique (ANARME), established under the Medicines Act of 2017. ANARME is responsible for ensuring the quality, safety, efficacy, and traceability of all pharmaceutical products circulating in the country. This includes the regulation of medicines, vaccines, and biological products.
The legal framework governing pharmaceutical imports and marketing authorization in Mozambique is primarily based on Law 4/98 and Decree 22/99. These regulations outline the requirements for the registration of pharmaceutical products, including the submission of necessary documentation, compliance with quality standards, and adherence to safety and efficacy criteria. The establishment of ANARME has further strengthened the regulatory environment, aligning it with international best practices and enhancing the efficiency and transparency of the registration process.
2Import Licensing & GMP
Import licensing in Mozambique requires that all pharmaceutical products obtain marketing authorization from ANARME prior to importation, distribution, or sale. This process involves the submission of comprehensive documentation, including product dossiers, certificates of analysis, and evidence of compliance with Good Manufacturing Practices (GMP). ANARME evaluates these submissions to ensure that imported products meet the necessary quality and safety standards.
Mozambique recognizes GMP certifications from reputable international bodies, such as the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Pharmaceutical companies seeking to export to Mozambique must ensure that their manufacturing facilities hold valid GMP certifications from these recognized organizations. Additionally, wholesale distribution authorization is required for entities involved in the distribution of pharmaceutical products within the country. This authorization is granted by ANARME upon verification of compliance with regulatory requirements and standards.
3Quality & Labeling
Imported pharmaceutical products in Mozambique are subject to batch testing and stability requirements to ensure their quality and efficacy. ANARME may conduct random sampling and testing of imported products to verify compliance with established standards. Manufacturers and importers are required to provide certificates of analysis and other relevant documentation to demonstrate product quality.
Labeling requirements stipulate that product labels must be in Portuguese, the official language of Mozambique. Labels must include essential information such as product name, active ingredients, dosage form, strength, batch number, manufacturing and expiry dates, storage conditions, and the name and address of the manufacturer or distributor. Serialization mandates may be implemented to enhance traceability and prevent counterfeit products; however, specific requirements should be verified with ANARME.
4Recent Regulatory Changes
Between 2024 and 2026, Mozambique has implemented several policy changes affecting pharmaceutical imports. The establishment of ANARME in 2017 has led to more stringent regulatory oversight, including the requirement for all pharmaceutical products to obtain marketing authorization prior to importation. This has streamlined the registration process and improved the quality control of imported medicines. Additionally, Mozambique has aligned its regulatory framework with international standards, enhancing the efficiency and transparency of the pharmaceutical import process.
ACE Health Care LDA — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
ACE Health Care LDA's product strategy focuses on importing a diverse range of pharmaceutical products across three therapeutic categories: antibiotics (87.8%), gastrointestinal (8.8%), and combination drugs (3.4%). The top three products imported are Erythromycin, Ranitidine, and Ibuprofen Paracetamol, indicating a strategic emphasis on essential medicines commonly used in the treatment of infections, gastrointestinal disorders, and pain management. This product mix aligns with the prevalent health needs in Mozambique, where infectious diseases, gastrointestinal issues, and pain-related conditions are common. By focusing on these therapeutic areas, ACE Health Care LDA aims to address critical healthcare challenges and improve patient outcomes.
The
Frequently Asked Questions — ACE Health Care LDA
What products does ACE Health Care LDA import from India?
ACE Health Care LDA imports 3 pharmaceutical products across 3 categories. Top imports: Erythromycin ($1.2M), Ranitidine ($125.2K), Ibuprofen Paracetamol ($47.8K).
Who supplies pharmaceuticals to ACE Health Care LDA from India?
ACE Health Care LDA sources from 2 verified Indian suppliers. The primary supplier is Biomatrix Healthcare Private Limited (99.4% of imports, $14.4M).
What is ACE Health Care LDA's total pharmaceutical import value?
ACE Health Care LDA's total pharmaceutical import value from India is $1.4M, based on 51 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does ACE Health Care LDA focus on?
ACE Health Care LDA imports across 3 categories. The largest: Antibiotics (87.8%), Gastrointestinal (8.8%), Combination Drugs (3.4%).
Get Full ACE Health Care LDA Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: ACE Health Care LDA identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as ACE Health Care LDA's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 51 individual customs records matching ACE Health Care LDA.
- 5.Supplier Verification: ACE Health Care LDA sources from 2 verified Indian suppliers across 651 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.