Abacus Pharma (A) Ltd
Pharmaceutical Importer · Burundi · Advanced Antibiotics Focus · $497.5K Total Trade · DGFT Verified
Abacus Pharma (A) Ltd is a pharmaceutical importer based in Burundi with a total trade value of $497.5K across 4 products in 3 therapeutic categories. Based on 25 verified import shipments from Indian Customs (DGFT) records, Abacus Pharma (A) Ltd is the #1 buyer in 1 product including Volini. Abacus Pharma (A) Ltd sources from 23 verified Indian suppliers, with Ajanta Pharma Limited accounting for 20.2% of imports.
Abacus Pharma (A) Ltd — Import Portfolio & Supplier Network
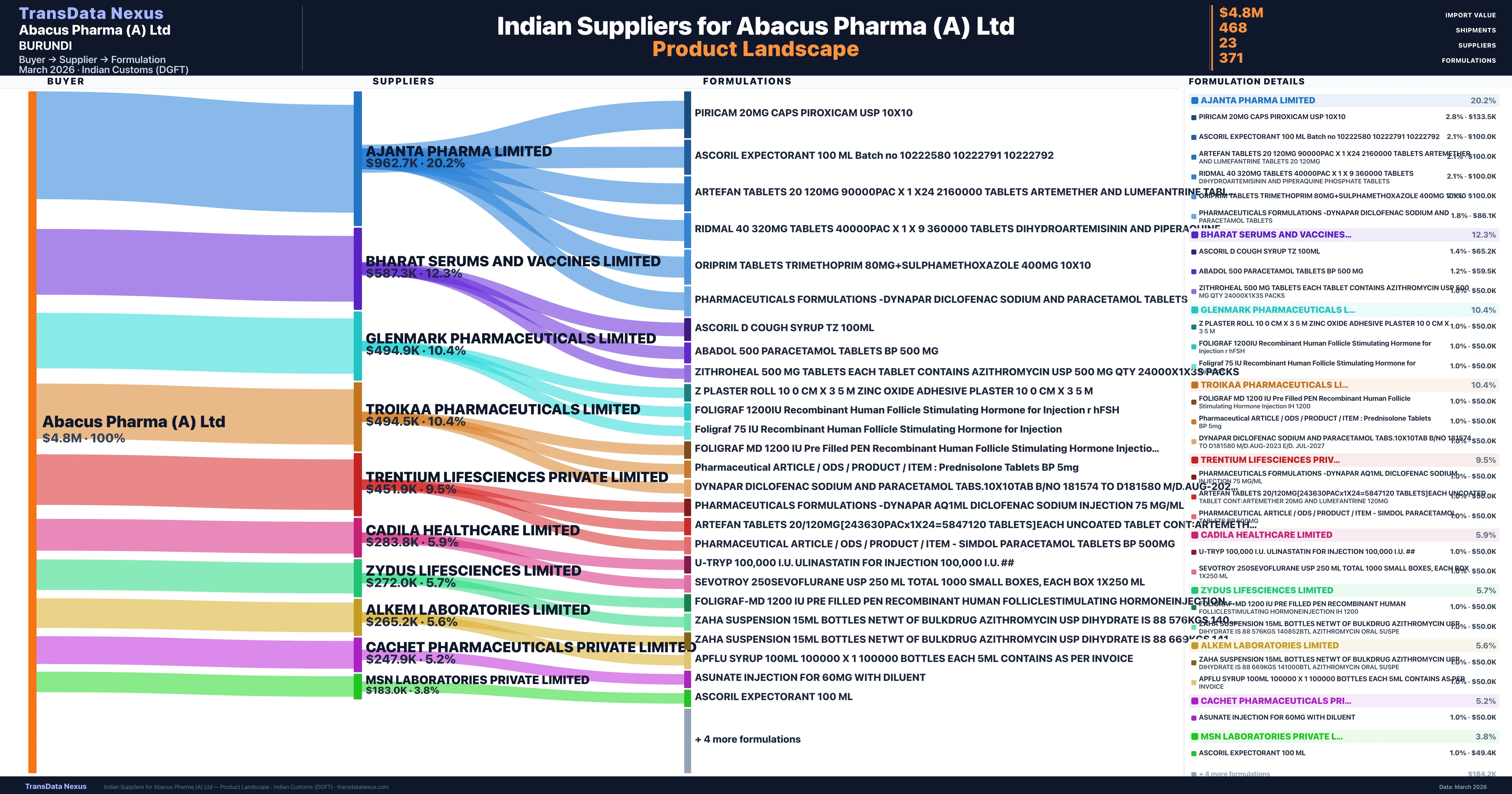
Who Are the Verified Indian Suppliers to Abacus Pharma (A) Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Ajanta Pharma Limited | $962.7K | 148 | 20.2% |
| Bharat Serums And Vaccines Limited | $587.3K | 26 | 12.3% |
| Glenmark Pharmaceuticals Limited | $494.9K | 50 | 10.4% |
| Troikaa Pharmaceuticals Limited | $494.5K | 21 | 10.4% |
| Trentium Lifesciences Private Limited | $451.9K | 35 | 9.5% |
| Cadila Healthcare Limited | $283.8K | 13 | 5.9% |
| Zydus Lifesciences Limited | $272.0K | 20 | 5.7% |
| Alkem Laboratories Limited | $265.2K | 21 | 5.6% |
| Cachet Pharmaceuticals Private Limited | $247.9K | 29 | 5.2% |
| Msn Laboratories Private Limited | $183.0K | 44 | 3.8% |
| Africure Pharmaceuticals India Private Limited | $123.7K | 3 | 2.6% |
| Wockhardt Limited | $106.0K | 12 | 2.2% |
| Psychotropics India Limited | $76.0K | 11 | 1.6% |
| Pinnacle Life Science Private Limited | $70.9K | 4 | 1.5% |
| Indasi Lifescience Private Limited | $50.0K | 1 | 1.0% |
| Meridian Enterprises Private Limited | $25.7K | 3 | 0.5% |
| Emami Limited | $16.5K | 3 | 0.3% |
| Meril Endo Surgery Private Limited | $14.9K | 8 | 0.3% |
| Dexin Medi Solutions Private Limited | $13.1K | 3 | 0.3% |
| Sanzyme Private Limited | $12.7K | 2 | 0.3% |
| Charak Pharma Private Limited | $10.3K | 8 | 0.2% |
| Synokem Pharmaceuticals Limited | $4.3K | 1 | 0.1% |
| Biological E Limited | $3.5K | 2 | 0.1% |
Abacus Pharma (A) Ltd sources from 23 verified Indian suppliers across 371 distinct formulations. The supply base is diversified across 23 suppliers, reducing single-source dependency risk.
What Formulations Does Abacus Pharma (A) Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Piricam 20MG CAPS piroxicam USP | $133.5K | 3 |
| Ascoril expectorant 100 ML batch no 10222580 10222791 | $100.0K | 2 |
| Artefan tablets 20 120MG 90000pac x 1 x24 2160000 tablets artemether and lumefantrine tablets 20 120MG | $100.0K | 2 |
| Ridmal 40 320MG tablets 40000pac x 1 x 9 360000 tablets dihydroartemisinin and piperaquine phosphate | $100.0K | 2 |
| Oriprim tablets trimethoprim 80MG+sulphamethoxazole 400MG | $100.0K | 2 |
| Pharmaceuticals formulations -dynapar diclofenac sodium and paracetamol | $86.1K | 2 |
| Ascoril d cough syrup tz 100ML | $65.2K | 2 |
| Abadol 500 paracetamol tablets BP 500 MG | $59.5K | 2 |
| Zithroheal 500 MG tablets Each tablet contains azithromycin USP 500 MG qty 24000x1x3s | $50.0K | 1 |
| Z plaster roll 10 0 cm x 3 5 m zinc oxide adhesive plaster 10 0 cm x 3 5 | $50.0K | 1 |
| Foligraf 1200iu recombinant human follicle stimulating hormone for injection r | $50.0K | 1 |
| Foligraf 75 IU recombinant human follicle stimulating hormone for | $50.0K | 1 |
| Foligraf md 1200 IU pre filled pen recombinant human follicle stimulating hormone injection ih | $50.0K | 1 |
| Pharmaceutical article / ods / product / item : prednisolone tablets BP 5MG | $50.0K | 1 |
| Dynapar diclofenac sodium and paracetamol tabs.10x10tab b/no 181574 to d181580 m/d.aug-2023 e/d. | $50.0K | 1 |
Abacus Pharma (A) Ltd imports 371 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Abacus Pharma (A) Ltd Import?
Top Products by Import Value
Abacus Pharma (A) Ltd Therapeutic Categories — 3 Specializations
Abacus Pharma (A) Ltd imports across 3 therapeutic categories, with Advanced Antibiotics (53.7%), Brand Names & OTC Products (40.2%), Advanced Oncology (6.1%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
2 products · 53.7% · $267.0K
Brand Names & OTC Products
1 products · 40.2% · $200.0K
Advanced Oncology
1 products · 6.1% · $30.5K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Volini | Brand Names & OTC Products | $200.0K | 4 | 6.8% | 1 |
| 2 | Secnidazole | Advanced Antibiotics | $167.0K | 12 | 0.6% | 14 |
| 3 | Trimethoprim | Advanced Antibiotics | $100.0K | 2 | 0.4% | 18 |
| 4 | Tofacitinib | Advanced Oncology | $30.5K | 7 | 0.8% | 9 |
Abacus Pharma (A) Ltd imports 4 pharmaceutical products across 3 categories into Burundi totaling $497.5K. The company is the #1 buyer for 1 product: Volini.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Abacus Pharma (A) Ltd.
Request DemoAbacus Pharma (A) Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Abacus Pharma (A) Ltd is a leading pharmaceutical distributor and manufacturer operating across East Africa, including Burundi. Established in 1995, the company has grown to become a significant player in the region's healthcare sector. Headquartered in Kampala, Uganda, Abacus Pharma (A) Ltd has expanded its operations to Kenya, Tanzania, Rwanda, and Burundi, with a presence in multiple countries across the continent.
In Burundi, Abacus Pharma (A) Ltd has been operational since 2006, positioning itself as a key distributor in the country's pharmaceutical market. The company offers a comprehensive range of pharmaceutical products, including both locally manufactured and imported medicines, catering to various therapeutic needs. Abacus Pharma (A) Ltd's commitment to quality and accessibility has contributed to its growing influence in Burundi's healthcare landscape.
2Distribution Network
Abacus Pharma (A) Ltd has established an extensive distribution network across East Africa, including Burundi. In Burundi, the company operates a warehouse located in Bujumbura, the country's economic capital. This strategic location enables efficient distribution of pharmaceutical products throughout the region. The company's logistics capabilities are supported by a fleet of vehicles and partnerships with local distributors, ensuring timely delivery to various healthcare facilities and pharmacies. Abacus Pharma (A) Ltd's geographic coverage extends beyond Burundi, with operations in Uganda, Kenya, Tanzania, and Rwanda, facilitating a robust supply chain across East Africa.
3Industry Role
In Burundi's pharmaceutical supply chain, Abacus Pharma (A) Ltd functions primarily as a wholesaler and distributor. The company imports a diverse range of pharmaceutical products, including both generic and branded medicines, and supplies them to hospitals, pharmacies, and other healthcare providers across the country. Abacus Pharma (A) Ltd's role is crucial in ensuring the availability of essential medicines, thereby supporting the healthcare infrastructure in Burundi. While the company also engages in local manufacturing, its primary focus in Burundi remains on distribution and wholesale activities.
Supplier Relationship Intelligence — Abacus Pharma (A) Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Abacus Pharma (A) Ltd's sourcing strategy reveals a significant concentration on a select group of Indian pharmaceutical suppliers. The company's total import value from India amounts to $498,000 USD, distributed across 25 shipments and 4 products. The top five products imported from India include Volini ($200,000), Secnidazole ($167,000), Trimethoprim ($100,000), and Tofacitinib ($31,000). Notably, Volini accounts for 6.8% of the total import value, indicating a strong preference for this product.
This sourcing pattern suggests a strategic choice to maintain a focused product portfolio, potentially simplifying inventory management and strengthening supplier relationships. However, the heavy reliance on a limited number of suppliers may pose risks related to supply chain disruptions or price fluctuations. The shipment data indicates a stable relationship with these suppliers, as evidenced by the consistent import volumes over the period. Nonetheless, diversifying the supplier base could enhance resilience against potential supply chain challenges.
2Supply Chain Resilience
Abacus Pharma (A) Ltd's supply chain resilience is influenced by its sourcing strategy from Indian pharmaceutical manufacturers. The company's portfolio includes a diverse range of formulations, encompassing 371 unique products, which indicates a broad product offering. However, the concentration on a few key suppliers may expose the company to risks associated with supply chain disruptions, such as production delays or regulatory changes affecting these suppliers.
To mitigate these risks, Abacus Pharma (A) Ltd could consider establishing relationships with additional suppliers to diversify its sourcing base. Additionally, evaluating alternative shipping routes and maintaining strategic stock levels could enhance supply chain resilience. Ensuring that all suppliers adhere to recognized Good Manufacturing Practices (GMP) standards is essential to maintain product quality and regulatory compliance.
3Strategic Implications
Abacus Pharma (A) Ltd's focused sourcing strategy positions the company to leverage strong relationships with key Indian pharmaceutical suppliers, potentially securing favorable terms and consistent product quality. This approach may also streamline operations and reduce complexities associated with managing a diverse supplier base.
For Indian exporters, this concentrated sourcing pattern presents an opportunity to strengthen partnerships with Abacus Pharma (A) Ltd by offering high-quality products that align with the company's portfolio. However, to become alternative suppliers, Indian exporters would need to demonstrate reliability, competitive pricing, and compliance with international quality standards to meet Abacus Pharma (A) Ltd's requirements.
Importing Pharmaceuticals into Burundi — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Burundi
1Regulatory Authority & Framework
In Burundi, the primary regulatory authority overseeing pharmaceutical imports and distribution is the Ministry of Public Health and the Fight Against AIDS. This ministry is responsible for establishing and enforcing regulations related to the importation, registration, and distribution of pharmaceutical products. Key legislation governing pharmaceutical imports includes the Law No. 1/10 of 2009, which outlines the conditions for the importation and commercialization of medicines.
The marketing authorization pathway for Indian generics involves several steps:
1. Product Registration: Pharmaceutical products must be registered with the Ministry of Public Health, providing detailed information on the product's composition, manufacturing process, and clinical efficacy.
2. Quality Control: Imported medicines are subject to quality control tests to ensure they meet the required standards.
3. Import License: An import license is required for each shipment, ensuring that only authorized entities can bring pharmaceutical products into the country.
4. Distribution Authorization: Companies must obtain authorization to distribute pharmaceutical products within Burundi, ensuring compliance with local regulations and standards.
2Import Licensing & GMP
Import licensing requirements in Burundi stipulate that all pharmaceutical products must be accompanied by an import license issued by the Ministry of Public Health. This license ensures that the products meet the necessary quality and safety standards before entering the market.
Burundi recognizes Good Manufacturing Practice (GMP) certificates from reputable international bodies, including the World Health Organization (WHO) and the European Union (EU). Pharmaceutical companies exporting to Burundi should ensure that their manufacturing facilities hold valid GMP certifications from these recognized authorities.
Additionally, companies must obtain wholesale distribution authorization from the Ministry of Public Health to legally distribute pharmaceutical products within Burundi. This authorization confirms that the distributor complies with all regulatory requirements and maintains the necessary infrastructure for safe and effective distribution.
3Quality & Labeling
Imported pharmaceutical products are subject to batch testing and stability requirements to ensure their safety and efficacy. The Ministry of Public Health conducts these tests upon arrival to verify that the products meet the established standards.
Labeling requirements include providing information in French, the official language of Burundi. Labels must include details such as the product name, active ingredients, dosage form, batch number, manufacturing and expiry dates, storage conditions, and the manufacturer's contact information.
Serialization mandates may be implemented to enhance traceability and prevent counterfeit products. While specific serialization requirements are not detailed, it is advisable for exporters to stay informed about any regulatory changes in this area.
4Recent Regulatory Changes
Between 2024 and 2026, Burundi has implemented several policy changes affecting pharmaceutical imports:
1. Enhanced Quality Control Measures: The Ministry of Public Health has introduced stricter quality control protocols for imported medicines, including more rigorous batch testing and stability assessments.
2. Revised Import Licensing Procedures: The process for obtaining import licenses has been streamlined to reduce processing times, but now requires additional documentation to verify compliance with international standards.
3. Updated Labeling Requirements: New regulations mandate that all pharmaceutical product labels include QR codes linking to digital certificates of authenticity, enhancing transparency and consumer trust.
Abacus Pharma (A) Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Abacus Pharma (A) Ltd's product category focus is strategically aligned with the healthcare needs of the East African region. The company's imports from India are concentrated in three therapeutic categories:
1. Advanced Antibiotics (53.7%): This category addresses the growing demand for effective treatments against bacterial infections, a significant concern in the region.
2. Brand Names & OTC Products (40.2%): Offering well-known brands and over-the-counter medications caters to consumer preferences for trusted products.
3. Advanced Oncology (6.1%): Importing specialized oncology products reflects a commitment to providing treatments for cancer, a critical area of healthcare.
The market demand driving these imports includes the need for effective antibiotics to combat infectious diseases, the popularity of established brand-name products, and the rising incidence of cancer necessitating advanced treatments.
2Sourcing Profile
Abacus Pharma (A) Ltd's sourcing strategy emphasizes importing finished pharmaceutical formulations from India, focusing on generic drugs. This approach allows the company to offer affordable medicines while maintaining a diverse product portfolio.
Frequently Asked Questions — Abacus Pharma (A) Ltd
What products does Abacus Pharma (A) Ltd import from India?
Abacus Pharma (A) Ltd imports 4 pharmaceutical products across 3 categories. Top imports: Volini ($200.0K), Secnidazole ($167.0K), Trimethoprim ($100.0K), Tofacitinib ($30.5K).
Who supplies pharmaceuticals to Abacus Pharma (A) Ltd from India?
Abacus Pharma (A) Ltd sources from 23 verified Indian suppliers. The primary supplier is Ajanta Pharma Limited (20.2% of imports, $962.7K).
What is Abacus Pharma (A) Ltd's total pharmaceutical import value?
Abacus Pharma (A) Ltd's total pharmaceutical import value from India is $497.5K, based on 25 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Abacus Pharma (A) Ltd focus on?
Abacus Pharma (A) Ltd imports across 3 categories. The largest: Advanced Antibiotics (53.7%), Brand Names & OTC Products (40.2%), Advanced Oncology (6.1%).
Get Full Abacus Pharma (A) Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Abacus Pharma (A) Ltd identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Abacus Pharma (A) Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 25 individual customs records matching Abacus Pharma (A) Ltd.
- 5.Supplier Verification: Abacus Pharma (A) Ltd sources from 23 verified Indian suppliers across 371 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.