ZIM Laboratories Limited.
Indian Pharmaceutical Exporter · Antimalarial & Antiparasitic Specialist · $2.7M Total Trade · DGFT Verified
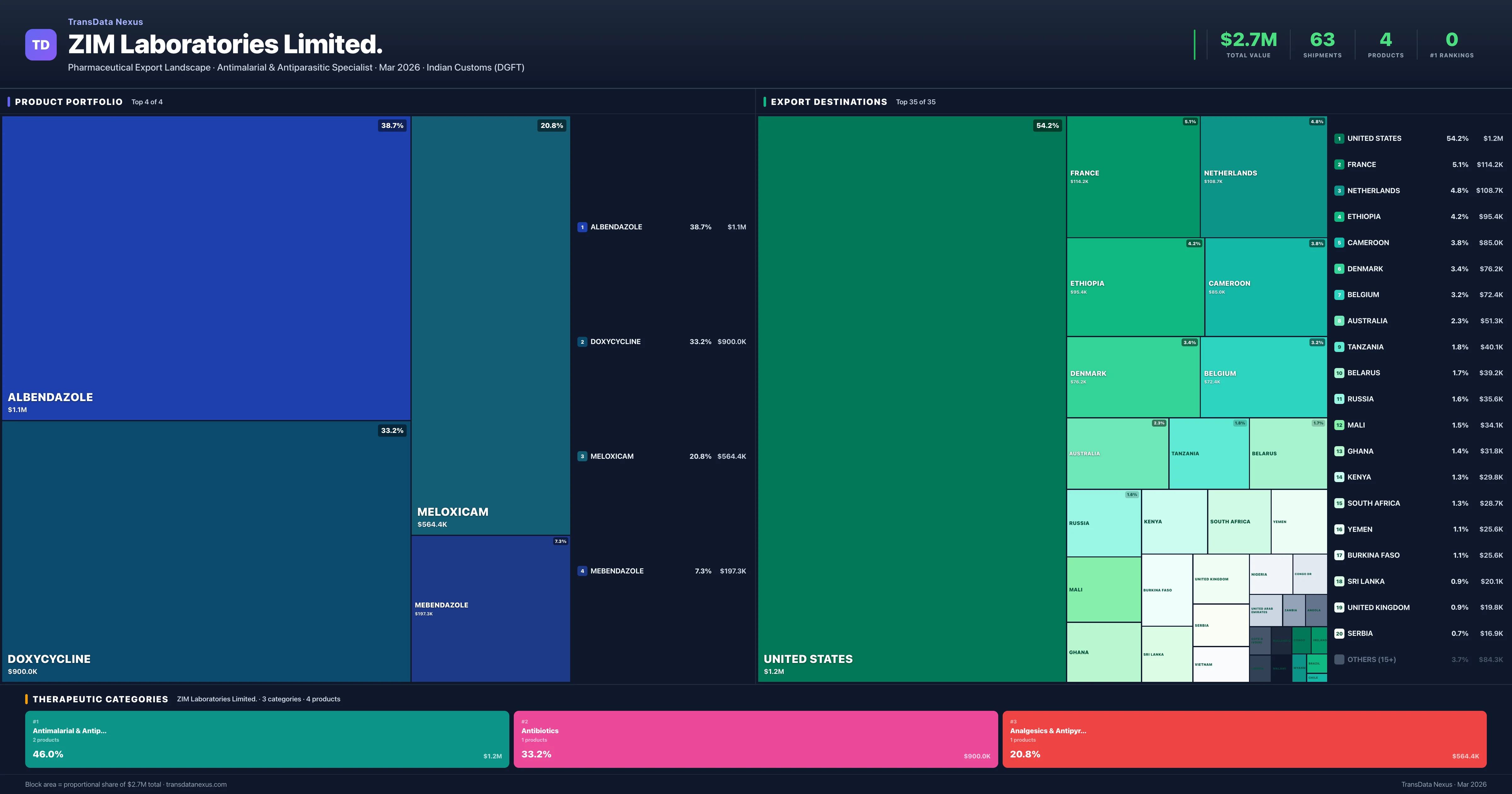
ZIM Laboratories Limited. is an Indian pharmaceutical exporter with a total trade value of $2.7M across 4 products in 3 therapeutic categories. Based on 63 verified export shipments from Indian Customs (DGFT) records, the company actively competes across multiple product segments. Top exports include Albendazole ($1.1M), Doxycycline ($900.0K), Meloxicam ($564.4K).
ZIM Laboratories Limited. — Export Portfolio & Destination Treemap
Who is ZIM Laboratories Limited.? — Company Overview & Market Position
ZIM Laboratories Limited, established in 1984, is a research-driven pharmaceutical company headquartered in Nagpur, Maharashtra, India. The company specializes in the development, manufacture, and supply of generic pharmaceutical and nutraceutical products, focusing on oral solid dosage forms such as tablets, capsules, and oral thin films. ZIM Laboratories offers therapeutics across various therapeutic areas, including urology, gastroenterology, antibiotics, anti-infectives, cardiovascular, central nervous system, NSAIDs/pain analgesics, and vitamins and supplements.
The company is publicly listed on the Bombay Stock Exchange (BSE) under the ticker symbol 541400 and on the National Stock Exchange of India (NSE) under the symbol ZIMLAB. As of March 13, 2026, ZIM Laboratories' stock price was ₹66.69, reflecting a 2.84% increase from the previous close. The company has a paid-up capital of ₹487,258,140 and an authorized capital of ₹750,000,000. ZIM Laboratories employs approximately 577 individuals, indicating a robust workforce dedicated to its operations.
What Does ZIM Laboratories Limited. Export? — Product Portfolio Analysis
Top Products by Export Value
ZIM Laboratories Limited. Therapeutic Categories — 3 Specializations
ZIM Laboratories Limited. operates across 3 therapeutic categories, with Antimalarial & Antiparasitic (46.0%), Antibiotics (33.2%), Analgesics & Antipyretics (20.8%) representing the largest segments by export value. The portfolio is concentrated — the top 5 products account for 100% of total exports.
Antimalarial & Antiparasitic
2 products · 46.0% · $1.2M
Antibiotics
1 products · 33.2% · $900.0K
Analgesics & Antipyretics
1 products · 20.8% · $564.4K
Product Portfolio — Top 4 by Export Value
ZIM Laboratories Limited. exports 4 pharmaceutical products across 3 therapeutic categories.
| # | Product | Category | Export Value | Shipments | Market Share | Rank |
|---|---|---|---|---|---|---|
| 1 | Albendazole | Antimalarial & Antiparasitic | $1.1M | 21 | 1.4% | 13 |
| 2 | Doxycycline | Antibiotics | $900.0K | 18 | 0.8% | 11 |
| 3 | Meloxicam | Analgesics & Antipyretics | $564.4K | 20 | 0.3% | 13 |
| 4 | Mebendazole | Antimalarial & Antiparasitic | $197.3K | 4 | 1.4% | 11 |
ZIM Laboratories Limited. exports 4 pharmaceutical products across 3 therapeutic categories with a total export value of $2.7M. The top category is Antimalarial & Antiparasitic (46.0% of portfolio), followed by Antibiotics (33.2%), indicating a concentrated portfolio with the top 5 products accounting for 100.0% of total export value.
Key Metrics
Top Categories
Top Export Routes
Related Analysis
Need Detailed Data?
Shipment-level records, pricing & buyer contacts for ZIM Laboratories Limited..
Request DemoZIM Laboratories Limited. — Corporate Profile & Information
Headquarters, ownership, manufacturing facilities, and key leadership
1Corporate Overview
ZIM Laboratories Limited, established in 1984, is a research-driven pharmaceutical company headquartered in Nagpur, Maharashtra, India. The company specializes in the development, manufacture, and supply of generic pharmaceutical and nutraceutical products, focusing on oral solid dosage forms such as tablets, capsules, and oral thin films. ZIM Laboratories offers therapeutics across various therapeutic areas, including urology, gastroenterology, antibiotics, anti-infectives, cardiovascular, central nervous system, NSAIDs/pain analgesics, and vitamins and supplements.
The company is publicly listed on the Bombay Stock Exchange (BSE) under the ticker symbol 541400 and on the National Stock Exchange of India (NSE) under the symbol ZIMLAB. As of March 13, 2026, ZIM Laboratories' stock price was ₹66.69, reflecting a 2.84% increase from the previous close. The company has a paid-up capital of ₹487,258,140 and an authorized capital of ₹750,000,000. ZIM Laboratories employs approximately 577 individuals, indicating a robust workforce dedicated to its operations.
2Manufacturing Facilities
ZIM Laboratories operates manufacturing facilities in Nagpur, Maharashtra, India. The company specializes in the production of oral solid dosage forms, including tablets, capsules, dry syrups, and oral thin films. These facilities are equipped with advanced technology platforms such as multi-particulates (pellets), taste masking, innovative granulation (direct compression granules), and orally disintegrating strips (thin films). The manufacturing plants adhere to stringent quality standards, ensuring the production of high-quality pharmaceutical products.
3Key Leadership
The leadership team at ZIM Laboratories is headed by Executive Chairman and Managing Director, Anwar Siraj Daud, who has been with the company since 2002. The Chief Financial Officer is Shyam Mohan Patro, who joined the company in April 2019. Other key executives include Niraj Pukhraj Dhadiwal, Director of Business Development and Executive Director since 2013; Zulfiquar Murtaza Kamal, Director of Finance and Executive Director since 2012; and Prakash Pralhadrao Sapkal, Head of ODS Business, Formulations of Domestic, ROW, and Emerging Markets & Director since 2013. This experienced leadership team drives the company's strategic initiatives and operational excellence.
Where Does ZIM Laboratories Limited. Export? — Destination Intelligence
Regulated market presence, emerging market penetration, and geographic strategy
1Regulated Markets
ZIM Laboratories has made significant strides in expanding its presence in regulated markets, including the United States, European Union, United Kingdom, Australia, and Japan. The company has received approval from the Central Drugs Standard Control Organization (CDSCO) in India for its novel Fixed Dose Combination formulation: Naproxen Delayed Release and Esomeprazole Capsules (375 mg/500 mg + 20 mg/20 mg). This product, developed using ZIM's proprietary drug delivery systems, is indicated for the symptomatic treatment of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis, as well as reducing the risk of NSAID-associated gastric and duodenal ulcers. The approval, granted on March 12, 2025, strengthens ZIM's presence in the pain management and gastro-protective therapeutic areas.
In the United States, ZIM Laboratories has registered its manufacturing facilities with the Food and Drug Administration (FDA), enabling the export of its products to the U.S. market. The company has also filed Abbreviated New Drug Applications (ANDAs) for several products, seeking approval for marketing in the U.S. In the European Union, ZIM Laboratories has obtained European Union Good Manufacturing Practice (EU GMP) certification, ensuring compliance with EU standards for pharmaceutical manufacturing. The company has also filed for marketing authorizations in various EU member states, facilitating access to these markets. In Australia, ZIM Laboratories has established a subsidiary, ZIMTAS Pty Ltd, incorporated in December 2022, to handle product registrations and marketing and distribution initiatives in Australia and New Zealand.
2Emerging Markets
ZIM Laboratories has strategically expanded its footprint in emerging markets, including Africa, Latin America, and Southeast Asia. The company has established subsidiaries such as ZIM Laboratories FZE in Sharjah, UAE, and ZIMTAS Pty Ltd in Australia, to enhance its presence in these regions. These subsidiaries focus on business development, marketing, product registrations, and distribution initiatives, enabling ZIM Laboratories to tap into new markets and strengthen its global presence. Additionally, the company's adherence to World Health Organization (WHO) prequalification standards facilitates access to various international markets, ensuring the quality and safety of its products.
3Geographic Strategy
ZIM Laboratories has adopted a diversified geographic strategy to mitigate concentration risk and capitalize on growth opportunities across different regions. The establishment of subsidiaries in strategic locations, including the UAE, Australia, and the EU, reflects the company's commitment to expanding its global footprint. This approach allows ZIM Laboratories to better serve international clients, comply with local regulations, and foster closer relationships with customers and partners worldwide. The company's focus on both developed and emerging markets demonstrates a balanced approach to growth and risk management.
ZIM Laboratories Limited. — Manufacturing & Regulatory Profile
FDA status, WHO-GMP certification, EU GMP compliance, and regulatory track record
1FDA Status
ZIM Laboratories has registered its manufacturing facilities with the FDA, enabling the export of its products to the U.S. market. The company has filed ANDAs for several products, seeking approval for marketing in the U.S. These filings indicate ZIM Laboratories' commitment to expanding its presence in the U.S. pharmaceutical market. The company's adherence to FDA regulations and standards underscores its dedication to quality and compliance in its manufacturing processes.
2WHO & EU GMP
ZIM Laboratories has obtained EU GMP certification, ensuring compliance with European standards for pharmaceutical manufacturing. This certification facilitates the company's access to the European market, allowing it to market and distribute its products within the EU. The company's adherence to WHO prequalification standards further demonstrates its commitment to maintaining high-quality manufacturing practices, which is essential for accessing various international markets.
3CDSCO & Indian Regulatory
In India, ZIM Laboratories holds manufacturing licenses issued by the CDSCO, authorizing it to produce pharmaceutical products for domestic and export markets. The company's compliance with Indian regulatory standards ensures the safety, efficacy, and quality of its products. ZIM Laboratories has also obtained export No Objection Certificates (NOCs) from the Directorate General of Foreign Trade (DGFT), facilitating the export of its products to various international markets. These regulatory approvals reflect the company's commitment to adhering to national and international standards in its manufacturing and export operations.
4Recent Regulatory Actions
On March 12, 2025, ZIM Laboratories received approval from the CDSCO for its novel Fixed Dose Combination formulation: Naproxen Delayed Release and Esomeprazole Capsules (375 mg/500 mg + 20 mg/20 mg). This approval strengthens the company's presence in the pain management and gastro-protective therapeutic areas. There are no publicly available records of Form 483 observations, warning letters, or import alerts issued to ZIM Laboratories by the FDA or other regulatory bodies. The company's adherence to regulatory standards and proactive approach to compliance contribute to its positive regulatory standing.
ZIM Laboratories Limited. — Competitive Positioning Among Indian Pharma Exporters
Market share comparison, key differentiators, and strategic analysis
1Competitive Landscape
ZIM Laboratories operates in a competitive pharmaceutical industry, with several key players in the generic pharmaceutical and nutraceutical sectors. While specific market share data is not publicly available, ZIM Laboratories' focus on differentiated drug delivery solutions, such as its proprietary oral thin film technology, positions it uniquely in the market. The company's diverse product portfolio across various therapeutic areas, including urology, gastroenterology, antibiotics, anti-infectives, cardiovascular, central nervous system, NSAIDs/pain analgesics, and vitamins and supplements, allows it to cater to a broad patient base. ZIM Laboratories' strategic expansion into emerging markets further enhances its competitive position.
2Key Differentiators
ZIM Laboratories' key differentiators include its focus on research-driven pharmaceutical development, proprietary drug delivery technologies, and a diverse product portfolio across multiple therapeutic areas. The company's in-house R&D capabilities enable it to offer non-infringing drug delivery solutions based on process technologies, enhancing patient compliance and reducing treatment costs. Additionally, ZIM Laboratories' commitment to quality is demonstrated through its adherence to international standards, including WHO prequalification and EU GMP certifications. The establishment of subsidiaries in strategic locations worldwide reflects the company's dedication to expanding its global presence and serving international clients effectively.
3Strategic Position
ZIM Laboratories' current strategic direction focuses on the development and supply of differentiated generic pharmaceutical and nutraceutical
Frequently Asked Questions — ZIM Laboratories Limited.
How many pharmaceutical products does ZIM Laboratories Limited. export from India?
ZIM Laboratories Limited. exports 4 pharmaceutical products across 3 therapeutic categories. The top exports are Albendazole ($1.1M), Doxycycline ($900.0K), Meloxicam ($564.4K), Mebendazole ($197.3K). Total export value is $2.7M.
What is ZIM Laboratories Limited.'s total pharmaceutical export value?
ZIM Laboratories Limited.'s total pharmaceutical export value is $2.7M, based on 63 verified shipments recorded in Indian Customs (DGFT) data.
What therapeutic categories does ZIM Laboratories Limited. cover?
ZIM Laboratories Limited. exports across 3 therapeutic categories. The largest are Antimalarial & Antiparasitic (46.0%, 2 products), Antibiotics (33.2%, 1 products), Analgesics & Antipyretics (20.8%, 1 products).
Get Full ZIM Laboratories Limited. Export Intelligence
Access shipment-level details, pricing data, buyer connections, product breakdown, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify manufacturer licensing, GMP certification, and export registration with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Matching: ZIM Laboratories Limited. identified across shipments using name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 3.Market Share: Calculated per product as ZIM Laboratories Limited.'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 63 individual customs records matching ZIM Laboratories Limited..
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports per product category. For current shipment-level data, contact TransData Nexus.