Truestar Enterprise Private Limited
Indian Pharmaceutical Exporter · Antihistamines & Allergy Specialist · $1.7M Total Trade · DGFT Verified
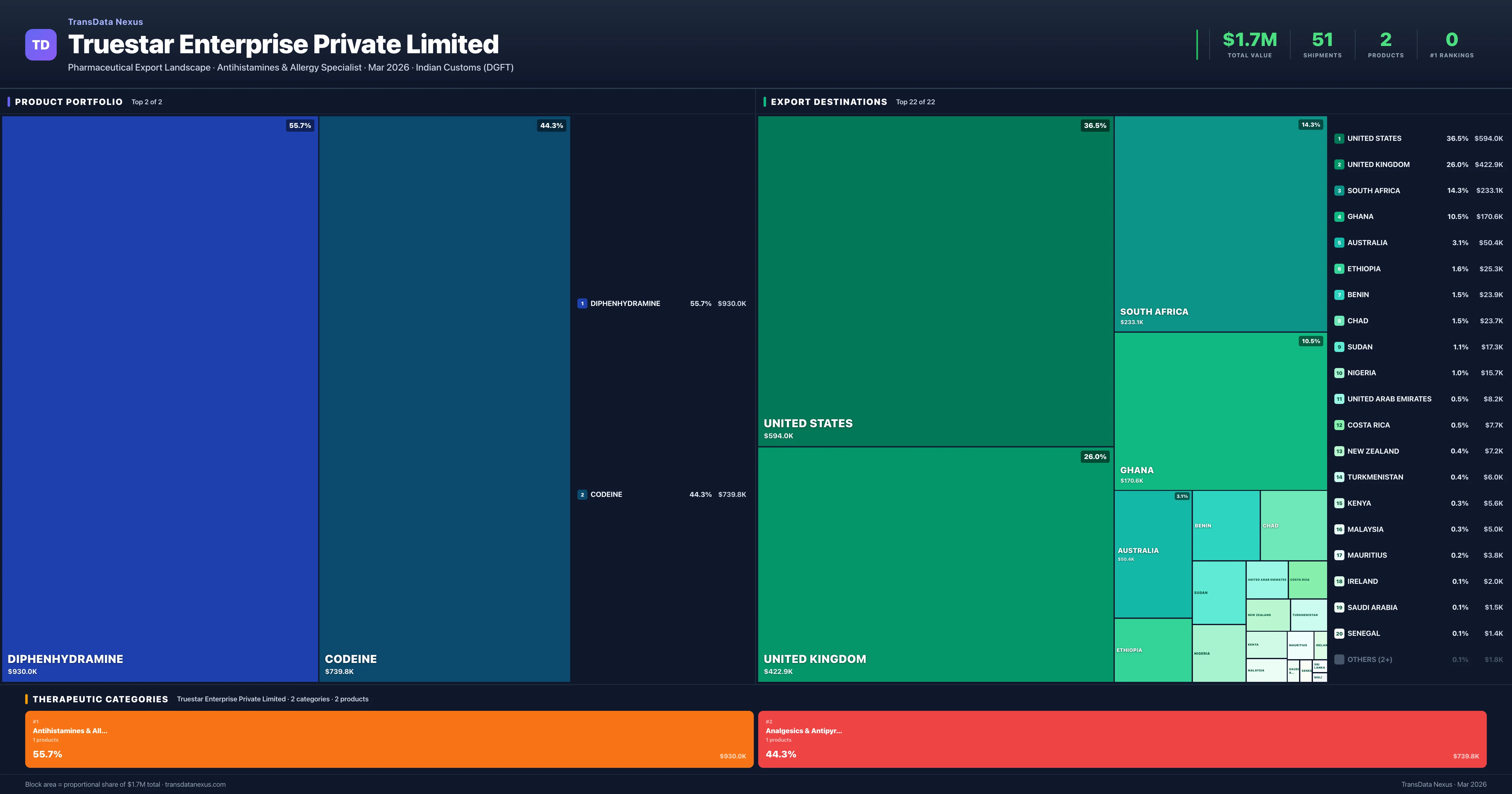
Truestar Enterprise Private Limited is an Indian pharmaceutical exporter with a total trade value of $1.7M across 2 products in 2 therapeutic categories. Based on 51 verified export shipments from Indian Customs (DGFT) records, the company actively competes across multiple product segments. Top exports include Diphenhydramine ($930.0K), Codeine ($739.8K), .
Truestar Enterprise Private Limited — Export Portfolio & Destination Treemap
Who is Truestar Enterprise Private Limited? — Company Overview & Market Position
Truestar Enterprise Private Limited is a privately held pharmaceutical company established on September 27, 2019, in Malad West, Maharashtra, India. The company is registered under the Corporate Identification Number (CIN) U74999MH2019PTC331077 and is classified as a non-government company. Its authorized and paid-up capital is ₹200,000.
The registered office is located at Office No. 807, 8th Floor, Jaswanti Allied Business Premises CSL, Kachpada, Ramchandra Lane Extension, Malad West, Maharashtra, 400064, India. As of March 31, 2024, the company reported a significant 47.93% increase in total revenue compared to the previous fiscal year, indicating robust growth.
What Does Truestar Enterprise Private Limited Export? — Product Portfolio Analysis
Top Products by Export Value
Truestar Enterprise Private Limited Therapeutic Categories — 2 Specializations
Truestar Enterprise Private Limited operates across 2 therapeutic categories, with Antihistamines & Allergy (55.7%), Analgesics & Antipyretics (44.3%), representing the largest segments by export value. The portfolio is concentrated — the top 5 products account for 100% of total exports.
Antihistamines & Allergy
1 products · 55.7% · $930.0K
Analgesics & Antipyretics
1 products · 44.3% · $739.8K
Product Portfolio — Top 2 by Export Value
Truestar Enterprise Private Limited exports 2 pharmaceutical products across 2 therapeutic categories.
| # | Product | Category | Export Value | Shipments | Market Share | Rank |
|---|---|---|---|---|---|---|
| 1 | Diphenhydramine | Antihistamines & Allergy | $930.0K | 27 | 2.9% | 7 |
| 2 | Codeine | Analgesics & Antipyretics | $739.8K | 24 | 0.7% | 8 |
Truestar Enterprise Private Limited exports 2 pharmaceutical products across 2 therapeutic categories with a total export value of $1.7M. The top category is Antihistamines & Allergy (55.7% of portfolio), followed by Analgesics & Antipyretics (44.3%), indicating a concentrated portfolio with the top 5 products accounting for 100.0% of total export value.
Key Metrics
Top Categories
Top Export Routes
Related Analysis
Need Detailed Data?
Shipment-level records, pricing & buyer contacts for Truestar Enterprise Private Limited.
Request DemoTruestar Enterprise Private Limited — Corporate Profile & Information
Headquarters, ownership, manufacturing facilities, and key leadership
1Corporate Overview
Truestar Enterprise Private Limited is a privately held pharmaceutical company established on September 27, 2019, in Malad West, Maharashtra, India. The company is registered under the Corporate Identification Number (CIN) U74999MH2019PTC331077 and is classified as a non-government company. Its authorized and paid-up capital is ₹200,000.
The registered office is located at Office No. 807, 8th Floor, Jaswanti Allied Business Premises CSL, Kachpada, Ramchandra Lane Extension, Malad West, Maharashtra, 400064, India. As of March 31, 2024, the company reported a significant 47.93% increase in total revenue compared to the previous fiscal year, indicating robust growth.
2Manufacturing Facilities
Specific details regarding Truestar Enterprise Private Limited's manufacturing facilities, including locations, capacities, and specializations, are not publicly disclosed. The company's export data indicates a focus on finished pharmaceutical formulations, such as tablets, capsules, syrups, and injections, rather than raw Active Pharmaceutical Ingredients (APIs) or bulk drugs. This suggests that the manufacturing operations are likely tailored to produce these dosage forms.
3Key Leadership
The leadership team of Truestar Enterprise Private Limited comprises several directors:
- Dharmesh Gunvantrai Vora: Director, appointed on April 1, 2020.
- Chirag Deepak Dedhia: Additional Director, appointed on October 17, 2024.
- Manisha Arvind Sharma: Additional Director, appointed on October 17, 2024.
- Pooja Mehul Vora: Additional Director, appointed on March 19, 2025.
- Devi Charan Yadav: Additional Director, appointed on March 19, 2025.
These appointments reflect the company's commitment to strengthening its leadership team to support its growth and strategic objectives.
Where Does Truestar Enterprise Private Limited Export? — Destination Intelligence
Regulated market presence, emerging market penetration, and geographic strategy
1Regulated Markets
Truestar Enterprise Private Limited's export data indicates shipments to various countries, including Benin, Ghana, and Chad. However, specific information regarding regulatory filings, approvals, and market access status in the United States, European Union, United Kingdom, Australia, and Japan is not publicly available. The company's focus appears to be on markets in Africa and the Middle East, as evidenced by its export destinations.
2Emerging Markets
The company's export activities in Africa, particularly in countries like Benin, Ghana, and Chad, suggest a strategic focus on emerging markets. While specific details about WHO prequalification are not available, the company's engagement in these regions indicates an interest in expanding its footprint in developing markets.
3Geographic Strategy
Truestar Enterprise Private Limited's export data reveals a concentration in specific markets, notably in Africa. This focus suggests a strategic approach to penetrate emerging markets, potentially reducing exposure to risks associated with over-diversification. The company's future direction may involve expanding its geographic reach to mitigate concentration risks.
Truestar Enterprise Private Limited — Manufacturing & Regulatory Profile
FDA status, WHO-GMP certification, EU GMP compliance, and regulatory track record
1FDA Status
Specific information regarding Truestar Enterprise Private Limited's FDA facility registrations, approved Abbreviated New Drug Applications (ANDAs), Drug Master File (DMF) filings, and inspection history is not publicly available. The company's export data does not indicate shipments to the United States, suggesting limited or no current market presence in the U.S.
2WHO & EU GMP
There is no publicly available information confirming whether Truestar Enterprise Private Limited holds WHO prequalification or EU Good Manufacturing Practice (GMP) certificates. The company's export data does not provide insights into its compliance with these international standards.
3CDSCO & Indian Regulatory
Specific details regarding Truestar Enterprise Private Limited's manufacturing licenses from the Central Drugs Standard Control Organisation (CDSCO), approvals from state drug controllers, and export No Objection Certificates (NOCs) are not publicly disclosed. The company's export data does not offer insights into its regulatory standing within India.
4Recent Regulatory Actions
There is no publicly available information regarding any Form 483 observations, warning letters, or import alerts associated with Truestar Enterprise Private Limited. The company's export data does not indicate any regulatory challenges or compliance issues.
Truestar Enterprise Private Limited — Competitive Positioning Among Indian Pharma Exporters
Market share comparison, key differentiators, and strategic analysis
1Competitive Landscape
Truestar Enterprise Private Limited operates in the pharmaceutical export sector, focusing on finished pharmaceutical formulations. While specific competitors are not identified, the company's export data suggests that it faces competition from other pharmaceutical exporters targeting similar markets, particularly in Africa. The company's market share in its top products indicates a competitive presence in its chosen export destinations.
2Key Differentiators
Truestar Enterprise Private Limited's focus on finished pharmaceutical formulations, such as tablets, capsules, syrups, and injections, differentiates it from competitors that may focus on raw APIs or bulk drugs. Its strategic focus on specific export markets, particularly in Africa, allows for tailored marketing and distribution strategies.
3Strategic Position
The company's current strategic direction appears to be centered on exporting finished pharmaceutical formulations to select international markets. While there is no publicly available information regarding its future outlook, the company's growth in revenue and profitability suggests a positive trajectory. Future strategies may involve expanding its product portfolio and exploring new markets to enhance its global presence.
Buyer Due Diligence Brief — Evaluating Truestar Enterprise Private Limited as a Supplier
Track record assessment, certifications to verify, and pre-order checks
1Supplier Assessment
Truestar Enterprise Private Limited's export data indicates a consistent volume of shipments, primarily focusing on finished pharmaceutical formulations. The company's engagement with multiple buyers across different countries suggests reliability and a stable supply chain. However, specific indicators of supplier reliability, such as on-time delivery rates or quality compliance records, are not publicly available.
2Certifications to Verify
Importers should verify the following certifications when considering Truestar Enterprise Private Limited as a supplier:
- FDA Registration: To confirm compliance with U.S. regulatory standards.
- WHO-GMP Certification: To ensure adherence to World Health Organization Good Manufacturing Practices.
- EU GMP Certification: To verify compliance with European Union Good Manufacturing Practices.
- ISO Certifications: To assess quality management and other relevant standards.
These certifications can typically be verified through official regulatory bodies or the company's official communications.
3Due Diligence Checklist
When conducting due diligence on Truestar Enterprise Private Limited, consider the following steps:
- Verify Regulatory Compliance: Confirm the company's certifications and approvals from relevant regulatory bodies.
- Assess Financial Health: Review financial statements to evaluate profitability and revenue growth.
- Evaluate Supply Chain Reliability: Assess the company's track record in fulfilling export orders and maintaining product quality.
- Check for Regulatory Actions: Investigate any past or ongoing regulatory issues or compliance violations.
Be cautious of red flags such as inconsistent financial records, lack of transparency in operations, or unresolved regulatory issues. Recommended pre-order checks include requesting recent financial statements, verifying certifications, and seeking references from previous clients or partners.
Frequently Asked Questions — Truestar Enterprise Private Limited
How many pharmaceutical products does Truestar Enterprise Private Limited export from India?
Truestar Enterprise Private Limited exports 2 pharmaceutical products across 2 therapeutic categories. The top exports are Diphenhydramine ($930.0K), Codeine ($739.8K). Total export value is $1.7M.
What is Truestar Enterprise Private Limited's total pharmaceutical export value?
Truestar Enterprise Private Limited's total pharmaceutical export value is $1.7M, based on 51 verified shipments recorded in Indian Customs (DGFT) data.
What therapeutic categories does Truestar Enterprise Private Limited cover?
Truestar Enterprise Private Limited exports across 2 therapeutic categories. The largest are Antihistamines & Allergy (55.7%, 1 products), Analgesics & Antipyretics (44.3%, 1 products).
Get Full Truestar Enterprise Private Limited Export Intelligence
Access shipment-level details, pricing data, buyer connections, product breakdown, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify manufacturer licensing, GMP certification, and export registration with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Matching: Truestar Enterprise Private Limited identified across shipments using name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 3.Market Share: Calculated per product as Truestar Enterprise Private Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 51 individual customs records matching Truestar Enterprise Private Limited.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports per product category. For current shipment-level data, contact TransData Nexus.