Troikaa Pharmaceuticals Limited
Indian Pharmaceutical Exporter · Analgesics & Antipyretics Specialist · $40.4M Total Trade · DGFT Verified
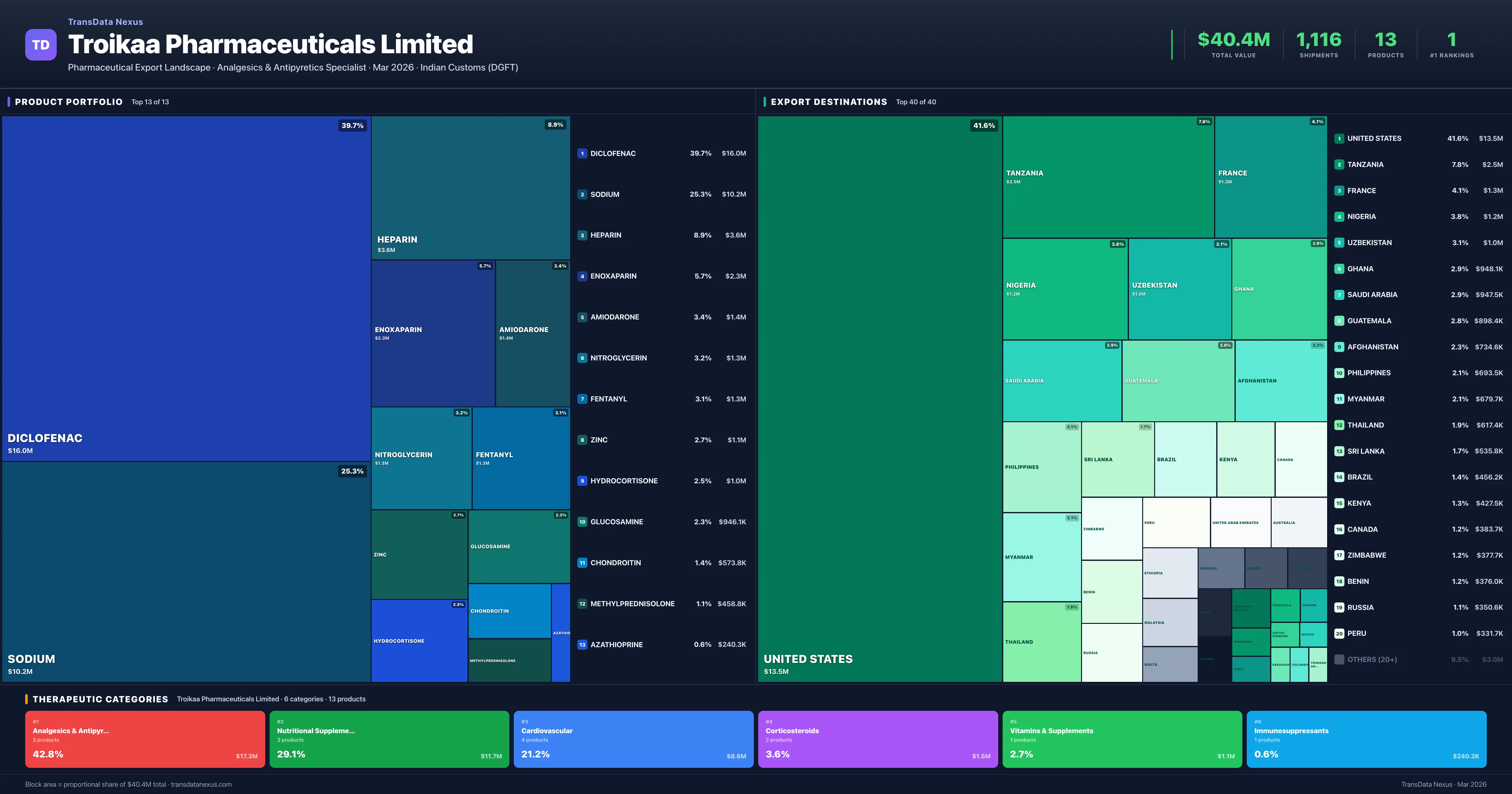
Troikaa Pharmaceuticals Limited is an Indian pharmaceutical exporter with a total trade value of $40.4M across 13 products in 6 therapeutic categories. Based on 1,116 verified export shipments from Indian Customs (DGFT) records, Troikaa Pharmaceuticals Limited is the #1 Indian exporter in 1 product including Fentanyl. Top exports include Diclofenac ($16.0M), Sodium ($10.2M), Heparin ($3.6M).
Troikaa Pharmaceuticals Limited — Export Portfolio & Destination Treemap
Who is Troikaa Pharmaceuticals Limited? — Company Overview & Market Position
Troikaa Pharmaceuticals Limited, established in 1983 by Mr. Rajni V. Patel, is a prominent Indian multinational pharmaceutical company headquartered in Ahmedabad, Gujarat. The company is registered under the Corporate Identification Number (CIN) U24100GJ1994PLC022162. As of March 2026, Troikaa Pharmaceuticals is a public unlisted company, classified as a company limited by shares.
Troikaa specializes in the development and manufacturing of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, with a strong emphasis on Novel Drug Delivery Systems (NDDS). The company's product portfolio spans various therapeutic categories, such as pain management, critical care, anesthesiology, and cardiology. Troikaa's innovative approach has led to the creation of path-breaking brands, with a lead time from concept to commercialization effectively maintained between 6 to 10 years.
With a workforce of over 2,800 employees, Troikaa Pharmaceuticals fosters a culture of creativity and continuous learning. The company operates three state-of-the-art manufacturing facilities in Gujarat and Uttarakhand, all certified by the World Health Organization's Good Manufacturing Practices (WHO GMP). These facilities are also approved by various regulatory agencies, including the Pharmaceutical Inspection Convention & Pharmaceutical Inspection Co-operation Scheme (PIC/S), ANVISA (Brazil), and INVIMA (Colombia).