Melon Globalcare
Indian Pharmaceutical Exporter · Advanced Oncology Specialist · $43.7K Total Trade · DGFT Verified
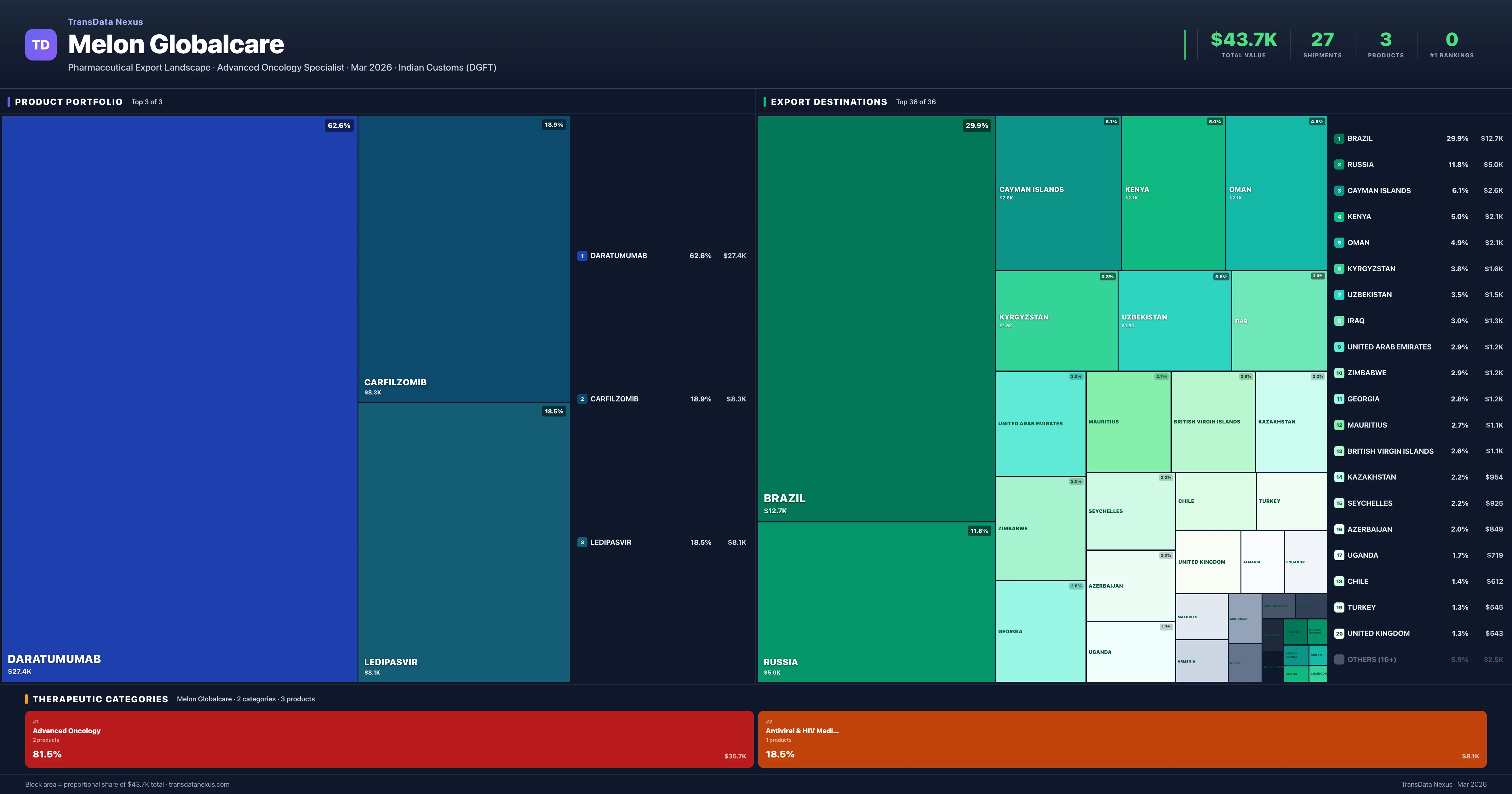
Melon Globalcare is an Indian pharmaceutical exporter with a total trade value of $43.7K across 3 products in 2 therapeutic categories. Based on 27 verified export shipments from Indian Customs (DGFT) records, the company actively competes across multiple product segments. Top exports include Daratumumab ($27.4K), Carfilzomib ($8.3K), Ledipasvir ($8.1K).
Melon Globalcare — Export Portfolio & Destination Treemap
Who is Melon Globalcare? — Company Overview & Market Position
Melon Globalcare, established in 2018, is a Mumbai-based pharmaceutical exporter specializing in the global distribution of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The company operates as a partnership, with Mr. Ronit Kothari serving as the Managing Partner. While specific details regarding the company's Corporate Identification Number (CIN) and revenue are not publicly disclosed, Melon Globalcare employs a team of approximately 10 individuals. The company's official website is www.melonglobalcare.com.
The company has established a strong presence in the pharmaceutical industry, exporting a diverse range of products to various international markets. Their product portfolio includes anti-cancer medications, HIV treatments, and other therapeutic drugs, reflecting a commitment to addressing critical health needs globally.
What Does Melon Globalcare Export? — Product Portfolio Analysis
Top Products by Export Value
Melon Globalcare Therapeutic Categories — 2 Specializations
Melon Globalcare operates across 2 therapeutic categories, with Advanced Oncology (81.5%), Antiviral & HIV Medications (18.5%), representing the largest segments by export value. The portfolio is concentrated — the top 5 products account for 100% of total exports.
Advanced Oncology
2 products · 81.5% · $35.7K
Antiviral & HIV Medications
1 products · 18.5% · $8.1K
Product Portfolio — Top 3 by Export Value
Melon Globalcare exports 3 pharmaceutical products across 2 therapeutic categories.
| # | Product | Category | Export Value | Shipments | Market Share | Rank |
|---|---|---|---|---|---|---|
| 1 | Daratumumab | Advanced Oncology | $27.4K | 11 | 2.0% | 13 |
| 2 | Carfilzomib | Advanced Oncology | $8.3K | 5 | 0.7% | 8 |
| 3 | Ledipasvir | Antiviral & HIV Medications | $8.1K | 11 | 0.7% | 13 |
Melon Globalcare exports 3 pharmaceutical products across 2 therapeutic categories with a total export value of $43.7K. The top category is Advanced Oncology (81.5% of portfolio), followed by Antiviral & HIV Medications (18.5%), indicating a concentrated portfolio with the top 5 products accounting for 100.0% of total export value.
Key Metrics
Top Categories
Top Export Routes
Related Analysis
Need Detailed Data?
Shipment-level records, pricing & buyer contacts for Melon Globalcare.
Request DemoMelon Globalcare — Corporate Profile & Information
Headquarters, ownership, manufacturing facilities, and key leadership
1Corporate Overview
Melon Globalcare, established in 2018, is a Mumbai-based pharmaceutical exporter specializing in the global distribution of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The company operates as a partnership, with Mr. Ronit Kothari serving as the Managing Partner. While specific details regarding the company's Corporate Identification Number (CIN) and revenue are not publicly disclosed, Melon Globalcare employs a team of approximately 10 individuals. The company's official website is www.melonglobalcare.com.
The company has established a strong presence in the pharmaceutical industry, exporting a diverse range of products to various international markets. Their product portfolio includes anti-cancer medications, HIV treatments, and other therapeutic drugs, reflecting a commitment to addressing critical health needs globally.
2Manufacturing Facilities
Melon Globalcare operates from its headquarters located at Mazgaon Koliwada Road, Mumbai, Maharashtra, India. While the company serves as a third-party manufacturer, trader, and bulk exporter, specific details regarding the locations, capacities, and specializations of its manufacturing facilities are not publicly disclosed. The company emphasizes quality assurance and timely delivery, ensuring that all pharmaceutical products meet stringent standards before reaching international markets.
3Key Leadership
Mr. Ronit Kothari serves as the Managing Partner of Melon Globalcare. Under his leadership, the company has expanded its global footprint, exporting pharmaceutical products to countries such as the United States, Germany, Saudi Arabia, and Sri Lanka. The company's team comprises skilled professionals dedicated to maintaining high-quality standards and ensuring timely delivery of products to international clients.
Where Does Melon Globalcare Export? — Destination Intelligence
Regulated market presence, emerging market penetration, and geographic strategy
1Regulated Markets
Melon Globalcare has established a presence in several regulated markets, including the United States, Germany, Saudi Arabia, and Sri Lanka. The company exports a range of pharmaceutical products, including anti-cancer medications and HIV treatments, to these countries. While specific details regarding regulatory filings, approvals, and market access status in these regions are not publicly disclosed, the company's adherence to international quality standards and its partnerships with reputable manufacturers suggest compliance with regulatory requirements.
In the United States, Melon Globalcare's products are likely subject to the Food and Drug Administration (FDA) regulations. The company's commitment to quality assurance and ethical business practices indicates a focus on meeting FDA standards for pharmaceutical exports. Similarly, in the European Union, the Medicines and Healthcare products Regulatory Agency (MHRA) oversees pharmaceutical products, and Melon Globalcare's adherence to international quality norms suggests compliance with EU regulations. In Australia, the Therapeutic Goods Administration (TGA) regulates pharmaceutical products, and the company's global distribution network indicates potential compliance with TGA standards. In Japan, the Pharmaceuticals and Medical Devices Agency (PMDA) oversees pharmaceutical products, and Melon Globalcare's international presence suggests adherence to PMDA regulations.
2Emerging Markets
Melon Globalcare has expanded its reach to emerging markets in Africa, Latin America, and Southeast Asia. The company's export activities in these regions include supplying anti-cancer and HIV medications, addressing critical health needs in these areas. While specific details regarding WHO prequalification and other regulatory approvals in these markets are not publicly disclosed, the company's global distribution network and partnerships with reputable manufacturers suggest a commitment to meeting international quality standards.
3Geographic Strategy
Melon Globalcare's geographic strategy demonstrates a diversified approach, targeting both developed and emerging markets. The company's export activities span regions such as North America, Europe, the Middle East, and parts of Asia, reflecting a broad market reach. This diversification helps mitigate concentration risk and positions the company to leverage growth opportunities across different regions. The strategic direction appears to focus on expanding the company's global footprint while maintaining a commitment to quality and timely delivery.
Melon Globalcare — Manufacturing & Regulatory Profile
FDA status, WHO-GMP certification, EU GMP compliance, and regulatory track record
1FDA Status
Specific details regarding Melon Globalcare's FDA facility registrations, approved Abbreviated New Drug Applications (ANDAs), Drug Master File (DMF) filings, and inspection history are not publicly disclosed. However, the company's export activities to the United States suggest a focus on compliance with FDA regulations. The company's commitment to quality assurance and ethical business practices indicates an emphasis on meeting FDA standards for pharmaceutical exports.
2WHO & EU GMP
While specific details regarding Melon Globalcare's World Health Organization (WHO) prequalification and European Union Good Manufacturing Practice (EU GMP) certifications are not publicly disclosed, the company's adherence to international quality standards and its partnerships with reputable manufacturers suggest compliance with these certifications. The company's global distribution network and export activities to various countries indicate a commitment to meeting WHO and EU GMP standards.
3CDSCO & Indian Regulatory
Melon Globalcare operates under the regulations of the Central Drugs Standard Control Organisation (CDSCO) in India. The company holds the necessary manufacturing licenses and approvals from state drug controllers to export pharmaceutical products. Specific details regarding export No Objection Certificates (NOCs) and other regulatory approvals are not publicly disclosed. However, the company's adherence to international quality standards and ethical business practices indicates compliance with Indian regulatory requirements.
4Recent Regulatory Actions
As of March 28, 2026, there are no publicly available records of Form 483 observations, warning letters, or import alerts issued to Melon Globalcare. The company's commitment to quality assurance and ethical business practices suggests a proactive approach to regulatory compliance. However, specific details regarding any past regulatory actions are not publicly disclosed.
Melon Globalcare — Competitive Positioning Among Indian Pharma Exporters
Market share comparison, key differentiators, and strategic analysis
1Competitive Landscape
Melon Globalcare operates in a competitive landscape with several key players in the pharmaceutical export sector. Top competitors in overlapping categories include Cipla Ltd, Intas Pharmaceuticals Ltd, and Aurobindo Pharma. These companies have established global distribution networks and a diverse product portfolio. In comparison, Melon Globalcare's focus on exporting anti-cancer and HIV medications positions it in a niche segment, potentially allowing for specialized expertise and targeted market strategies. While specific market share comparisons are not publicly available, Melon Globalcare's strategic focus on quality and timely delivery contributes to its competitive positioning.
2Key Differentiators
Melon Globalcare's key differentiators include its specialized focus on exporting anti-cancer and HIV medications, addressing critical health needs in various regions. The company's commitment to quality assurance and ethical business practices ensures that all pharmaceutical products meet stringent standards before reaching international markets. Additionally, Melon Globalcare's global distribution network and partnerships with reputable manufacturers enable it to offer a reliable marketplace for pharmaceutical supplies worldwide.
3Strategic Position
Melon Globalcare's current strategic direction focuses on exporting generic pharmaceutical drugs, particularly in the anti-cancer and HIV segments. The company's future outlook includes expanding its global footprint by entering new markets and enhancing its product portfolio to address a broader range of therapeutic areas. By maintaining a commitment to quality and timely delivery, Melon Globalcare aims to strengthen its position in the global pharmaceutical export sector.
Buyer Due Diligence Brief — Evaluating Melon Globalcare as a Supplier
Track record assessment, certifications to verify, and pre-order checks
1Supplier Assessment
Melon Globalcare has a track record of exporting pharmaceutical products to various international markets, including the United States, Germany, Saudi Arabia, and Sri Lanka. The company's export volume and consistency indicate a reliable supply chain. While specific reliability indicators are not publicly disclosed, the company's adherence to international quality standards and ethical business practices suggests a strong commitment to maintaining product integrity and customer satisfaction.
2Certifications to Verify
Importers should verify the following certifications when considering Melon Globalcare as a supplier:
- FDA Approval: Confirm that the company's products are approved by the U.S. Food and Drug Administration (FDA). Verification can be done through the FDA's official website or by contacting the company directly.
- WHO-GMP Certification: Ensure that the company holds World Health Organization Good Manufacturing Practice (WHO-GMP) certification. This can be verified by checking the WHO's official website or by requesting certification details from the company.
- EU GMP Certification: Verify that the company complies with European Union Good Manufacturing Practice (EU GMP) standards. This information can be obtained from the European Medicines Agency (EMA) or by contacting the company directly.
- ISO Certification: Check for ISO certifications related to quality management systems. Verification can be done through the International Organization for Standardization (ISO) website or by requesting certification details from the company.
3Due Diligence Checklist
When conducting due diligence
Frequently Asked Questions — Melon Globalcare
How many pharmaceutical products does Melon Globalcare export from India?
Melon Globalcare exports 3 pharmaceutical products across 2 therapeutic categories. The top exports are Daratumumab ($27.4K), Carfilzomib ($8.3K), Ledipasvir ($8.1K). Total export value is $43.7K.
What is Melon Globalcare's total pharmaceutical export value?
Melon Globalcare's total pharmaceutical export value is $43.7K, based on 27 verified shipments recorded in Indian Customs (DGFT) data.
What therapeutic categories does Melon Globalcare cover?
Melon Globalcare exports across 2 therapeutic categories. The largest are Advanced Oncology (81.5%, 2 products), Antiviral & HIV Medications (18.5%, 1 products).
Get Full Melon Globalcare Export Intelligence
Access shipment-level details, pricing data, buyer connections, product breakdown, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify manufacturer licensing, GMP certification, and export registration with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Matching: Melon Globalcare identified across shipments using name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 3.Market Share: Calculated per product as Melon Globalcare's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 27 individual customs records matching Melon Globalcare.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports per product category. For current shipment-level data, contact TransData Nexus.