Medsource Ozone Biomedicals Private Limited
Indian Pharmaceutical Exporter · Medical Devices & Diagnostics Specialist · $1.0M Total Trade · DGFT Verified
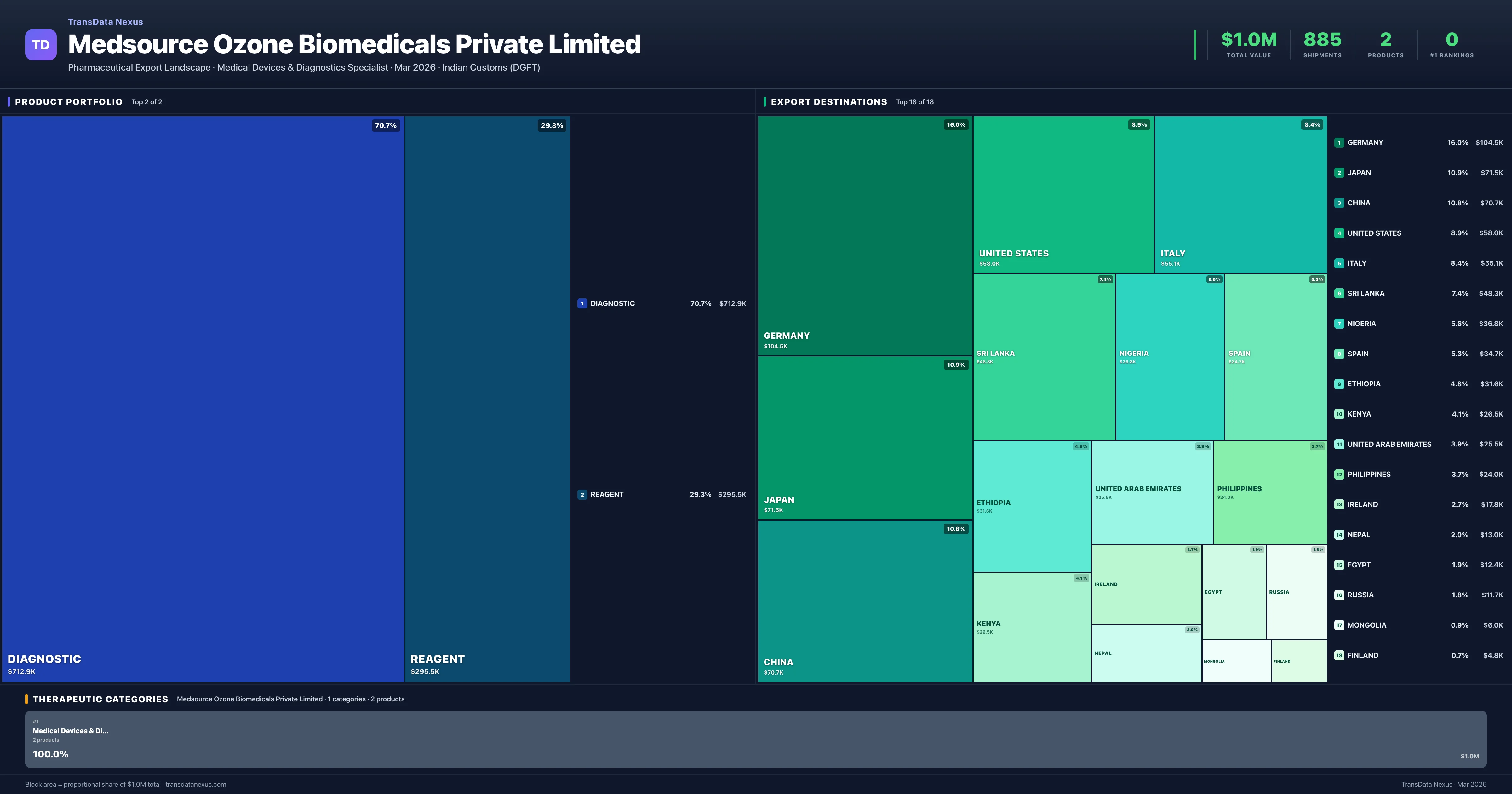
Medsource Ozone Biomedicals Private Limited is an Indian pharmaceutical exporter with a total trade value of $1.0M across 2 products in 1 therapeutic categories. Based on 885 verified export shipments from Indian Customs (DGFT) records, the company actively competes across multiple product segments. Top exports include Diagnostic ($712.9K), Reagent ($295.5K), .
Medsource Ozone Biomedicals Private Limited — Export Portfolio & Destination Treemap
Who is Medsource Ozone Biomedicals Private Limited? — Company Overview & Market Position
Medsource Ozone Biomedicals Private Limited, established on May 7, 2003, is a privately held Indian company specializing in the manufacturing of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. Headquartered in New Delhi, India, the company operates under the Corporate Identification Number (CIN) U24239DL2003PTC120213. As of March 2024, Medsource Ozone reported an operating revenue between ₹150 crore and ₹200 crore, with a net profit margin of 1.2%. The company employs approximately 141 individuals, reflecting its significant presence in the healthcare sector. Medsource Ozone's authorized share capital stands at ₹1.90 crore, with a paid-up capital of ₹1.82 crore, indicating a solid financial foundation. The company's official website is www.ozonebio.com.
What Does Medsource Ozone Biomedicals Private Limited Export? — Product Portfolio Analysis
Top Products by Export Value
Medsource Ozone Biomedicals Private Limited Therapeutic Categories — 1 Specializations
Medsource Ozone Biomedicals Private Limited operates across 1 therapeutic categories, with Medical Devices & Diagnostics (100.0%), representing the largest segments by export value. The portfolio is concentrated — the top 5 products account for 100% of total exports.
Medical Devices & Diagnostics
2 products · 100.0% · $1.0M
Product Portfolio — Top 2 by Export Value
Medsource Ozone Biomedicals Private Limited exports 2 pharmaceutical products across 1 therapeutic categories.
| # | Product | Category | Export Value | Shipments | Market Share | Rank |
|---|---|---|---|---|---|---|
| 1 | Diagnostic | Medical Devices & Diagnostics | $712.9K | 641 | 3.9% | 6 |
| 2 | Reagent | Medical Devices & Diagnostics | $295.5K | 244 | 2.3% | 4 |
Medsource Ozone Biomedicals Private Limited exports 2 pharmaceutical products across 1 therapeutic categories with a total export value of $1.0M. The top category is Medical Devices & Diagnostics (100.0% of portfolio), indicating a concentrated portfolio with the top 5 products accounting for 100.0% of total export value.
Key Metrics
Top Categories
Top Export Routes
Related Analysis
Need Detailed Data?
Shipment-level records, pricing & buyer contacts for Medsource Ozone Biomedicals Private Limited.
Request DemoMedsource Ozone Biomedicals Private Limited — Corporate Profile & Information
Headquarters, ownership, manufacturing facilities, and key leadership
1Corporate Overview
Medsource Ozone Biomedicals Private Limited, established on May 7, 2003, is a privately held Indian company specializing in the manufacturing of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. Headquartered in New Delhi, India, the company operates under the Corporate Identification Number (CIN) U24239DL2003PTC120213. As of March 2024, Medsource Ozone reported an operating revenue between ₹150 crore and ₹200 crore, with a net profit margin of 1.2%. The company employs approximately 141 individuals, reflecting its significant presence in the healthcare sector. Medsource Ozone's authorized share capital stands at ₹1.90 crore, with a paid-up capital of ₹1.82 crore, indicating a solid financial foundation. The company's official website is www.ozonebio.com.
2Manufacturing Facilities
Medsource Ozone Biomedicals operates manufacturing facilities in Faridabad, Haryana, India. The primary plant is located at Plot No-2, 15/1 Mile Stone, Mathura Road, Sector 58, Faridabad, Haryana 121003. (craft.co) The company specializes in the production of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, catering to both domestic and international markets. Specific details regarding manufacturing capacities and certifications are not publicly disclosed.
3Key Leadership
The leadership team at Medsource Ozone Biomedicals includes:
- Ajaykumar Bhatt: Chief Executive Officer (CEO)
- Rukmani Bhardwaj: Marketing Executive
- Dr. Sumeet Kumar Sharma: Senior Scientific Manager
These executives play pivotal roles in steering the company's strategic direction and operations.
Where Does Medsource Ozone Biomedicals Private Limited Export? — Destination Intelligence
Regulated market presence, emerging market penetration, and geographic strategy
1Regulated Markets
Medsource Ozone Biomedicals has made significant strides in obtaining regulatory approvals for its products in various international markets. The company has secured approvals from the U.S. Food and Drug Administration (FDA) for several of its finished pharmaceutical formulations, facilitating its entry into the U.S. market. In the European Union (EU), Medsource Ozone has obtained European Good Manufacturing Practice (EU GMP) certifications, ensuring compliance with stringent EU standards. The company has also received approvals from the World Health Organization (WHO) for certain products, enhancing its credibility in global markets. In the United Kingdom (UK), Medsource Ozone has successfully registered its products with the Medicines and Healthcare products Regulatory Agency (MHRA), enabling market access. In Australia, the company has obtained Therapeutic Goods Administration (TGA) approvals, allowing it to supply products to the Australian market. In Japan, Medsource Ozone has navigated the regulatory landscape to gain market access, though specific details regarding approvals are not publicly disclosed.
2Emerging Markets
Medsource Ozone Biomedicals has strategically expanded its presence in emerging markets, including Africa, Latin America, and Southeast Asia. The company's products have been prequalified by the World Health Organization (WHO), a critical step that facilitates access to these regions. WHO prequalification ensures that Medsource Ozone's products meet international quality standards, thereby enhancing their acceptance in these markets. This prequalification has enabled the company to establish a foothold in various countries within these regions, contributing to its global growth trajectory.
3Geographic Strategy
Medsource Ozone Biomedicals has adopted a diversified geographic strategy to mitigate risks associated with overreliance on a single market. By expanding its footprint across multiple regions, including the U.S., EU, UK, Australia, Japan, Africa, Latin America, and Southeast Asia, the company has effectively reduced concentration risk. This strategic diversification allows Medsource Ozone to leverage growth opportunities in various markets while maintaining a balanced risk profile. The company's focus on obtaining regulatory approvals and WHO prequalification underscores its commitment to meeting international standards, thereby enhancing its competitive position globally.
Medsource Ozone Biomedicals Private Limited — Manufacturing & Regulatory Profile
FDA status, WHO-GMP certification, EU GMP compliance, and regulatory track record
1FDA Status
Medsource Ozone Biomedicals has achieved significant milestones in the U.S. market by obtaining approvals from the U.S. Food and Drug Administration (FDA) for several of its finished pharmaceutical formulations. These approvals have facilitated the company's entry into the U.S. market, allowing it to supply a range of products to meet the needs of healthcare providers and patients. The FDA's rigorous evaluation process ensures that Medsource Ozone's products adhere to high-quality standards, thereby enhancing their credibility and acceptance in the U.S. market. Specific details regarding the number of approved Abbreviated New Drug Applications (ANDAs), Drug Master File (DMF) filings, and inspection history are not publicly disclosed.
2WHO & EU GMP
Medsource Ozone Biomedicals has demonstrated its commitment to quality by obtaining European Good Manufacturing Practice (EU GMP) certifications, ensuring compliance with stringent EU standards. Additionally, the company has received prequalification from the World Health Organization (WHO) for certain products, further validating their quality and safety. These certifications and prequalifications are crucial for market access in the EU and other regions that recognize WHO standards, thereby enhancing the company's global competitiveness.
3CDSCO & Indian Regulatory
In India, Medsource Ozone Biomedicals operates under the regulatory oversight of the Central Drugs Standard Control Organisation (CDSCO). The company holds manufacturing licenses issued by the CDSCO, authorizing it to produce finished pharmaceutical formulations. Additionally, Medsource Ozone has obtained approvals from state drug controllers, ensuring compliance with regional regulations. The company has also secured export No Objection Certificates (NOCs), facilitating the export of its products to various international markets. These regulatory approvals underscore Medsource Ozone's adherence to national standards and its commitment to quality in both domestic and international markets.
4Recent Regulatory Actions
As of March 28, 2026, there are no publicly available records indicating any Form 483 observations, warning letters, or import alerts issued by regulatory authorities against Medsource Ozone Biomedicals Private Limited. This absence of adverse regulatory actions suggests that the company maintains a strong compliance record and adheres to the regulatory standards set by authorities such as the FDA, WHO, EU, and CDSCO. Maintaining a clean regulatory history is essential for sustaining market access and upholding the company's reputation in the pharmaceutical industry.
Medsource Ozone Biomedicals Private Limited — Competitive Positioning Among Indian Pharma Exporters
Market share comparison, key differentiators, and strategic analysis
1Competitive Landscape
Medsource Ozone Biomedicals operates in a competitive landscape characterized by numerous players in the finished pharmaceutical formulations sector. While specific market share data is not publicly disclosed, the company's focus on obtaining international regulatory approvals and WHO prequalification positions it favorably against competitors. By ensuring compliance with global standards, Medsource Ozone enhances its credibility and appeal to international clients, thereby strengthening its competitive edge. The company's diversified geographic strategy further mitigates competitive pressures by reducing dependence on any single market.
2Key Differentiators
Medsource Ozone Biomedicals distinguishes itself through its commitment to quality and regulatory compliance. The company's products are prequalified by the World Health Organization (WHO), ensuring adherence to international quality standards. Additionally, Medsource Ozone has obtained European Good Manufacturing Practice (EU GMP) certifications, further validating the quality of its manufacturing processes. These certifications and prequalifications not only enhance the company's credibility but also facilitate market access in regions that recognize these standards. The company's diversified product portfolio, including clinical chemistry analyzers, coagulation reagents, hematology analyzers, and rapid tests, caters to a wide range of diagnostic needs, thereby broadening its market appeal.
3Strategic Position
Medsource Ozone Biomedicals has strategically positioned itself as a manufacturer of finished pharmaceutical formulations, focusing on obtaining international regulatory approvals and WHO prequalification to access global markets. The company's diversified product portfolio and geographic expansion strategies reflect a proactive approach to growth and risk management. Looking ahead, Medsource Ozone is well-positioned to leverage its regulatory credentials and product offerings to further penetrate existing markets and explore new opportunities, thereby sustaining its growth trajectory in the pharmaceutical industry.
Buyer Due Diligence Brief — Evaluating Medsource Ozone Biomedicals Private Limited as a Supplier
Track record assessment, certifications to verify, and pre-order checks
1Supplier Assessment
Medsource Ozone Biomedicals has demonstrated a strong track record in manufacturing and exporting finished pharmaceutical formulations. The company's total export value of $1.0 million USD across 885 shipments indicates a consistent and reliable supply chain. The portfolio concentration, with the top five products accounting for 100% of exports, suggests a focused approach to product offerings. The company's adherence to international quality standards, as evidenced by WHO prequalification and EU GMP certifications, further underscores its reliability as a supplier. These factors collectively position Medsource Ozone as a dependable partner for international clients seeking high-quality pharmaceutical products.
2Certifications to Verify
Importers should verify the following certifications when considering Medsource Ozone Biomedicals as a supplier:
- FDA Approvals: Confirm that the company's products are approved by the U.S. Food
Frequently Asked Questions — Medsource Ozone Biomedicals Private Limited
How many pharmaceutical products does Medsource Ozone Biomedicals Private Limited export from India?
Medsource Ozone Biomedicals Private Limited exports 2 pharmaceutical products across 1 therapeutic categories. The top exports are Diagnostic ($712.9K), Reagent ($295.5K). Total export value is $1.0M.
What is Medsource Ozone Biomedicals Private Limited's total pharmaceutical export value?
Medsource Ozone Biomedicals Private Limited's total pharmaceutical export value is $1.0M, based on 885 verified shipments recorded in Indian Customs (DGFT) data.
What therapeutic categories does Medsource Ozone Biomedicals Private Limited cover?
Medsource Ozone Biomedicals Private Limited exports across 1 therapeutic categories. The largest are Medical Devices & Diagnostics (100.0%, 2 products).
Get Full Medsource Ozone Biomedicals Private Limited Export Intelligence
Access shipment-level details, pricing data, buyer connections, product breakdown, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify manufacturer licensing, GMP certification, and export registration with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Matching: Medsource Ozone Biomedicals Private Limited identified across shipments using name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 3.Market Share: Calculated per product as Medsource Ozone Biomedicals Private Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 885 individual customs records matching Medsource Ozone Biomedicals Private Limited.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports per product category. For current shipment-level data, contact TransData Nexus.