Fresenius Kabi Oncology Limited
Indian Pharmaceutical Exporter · Oncology Specialist · $117.2M Total Trade · DGFT Verified
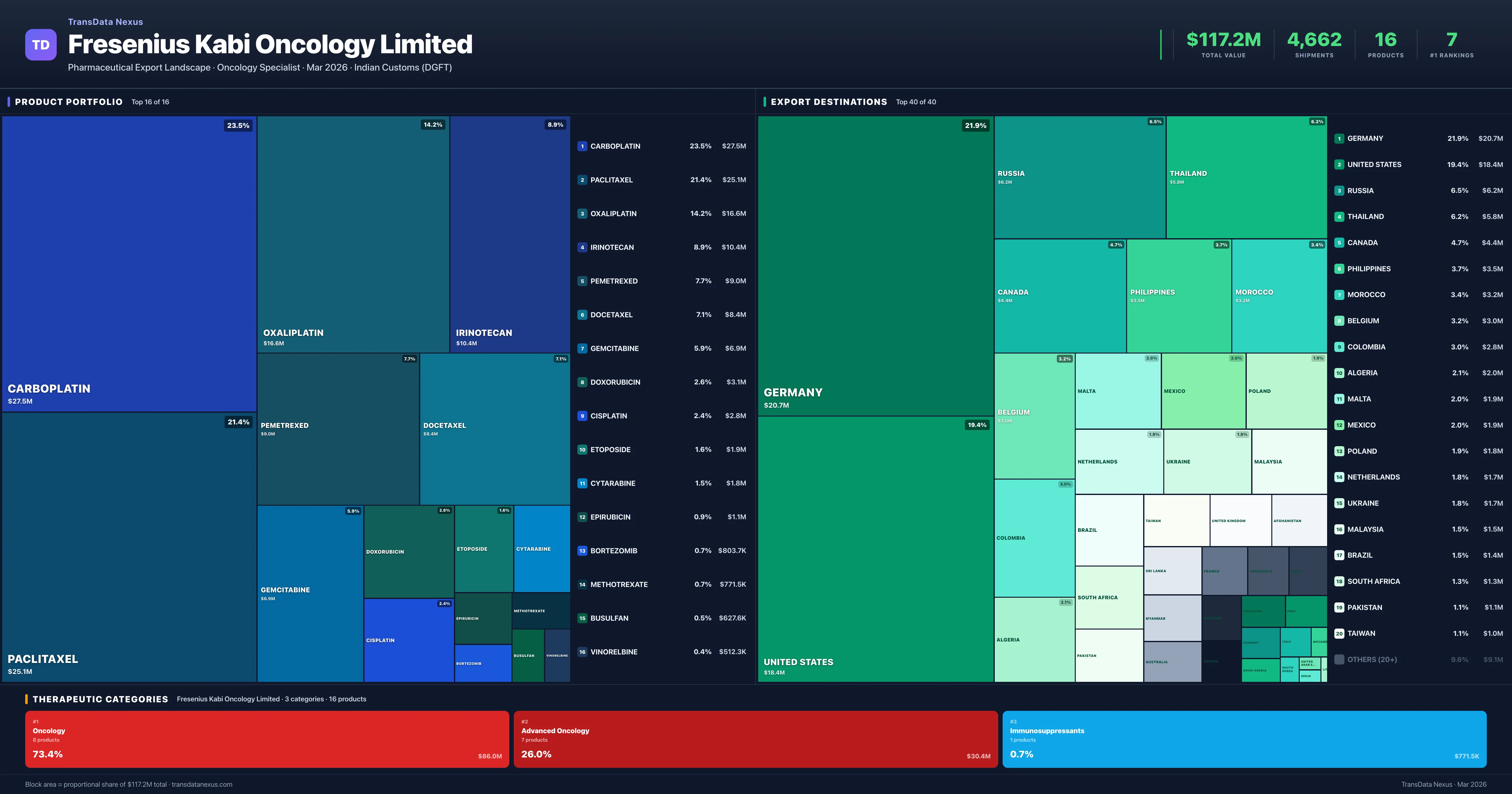
Fresenius Kabi Oncology Limited is an Indian pharmaceutical exporter with a total trade value of $117.2M across 16 products in 3 therapeutic categories. Based on 4,662 verified export shipments from Indian Customs (DGFT) records, Fresenius Kabi Oncology Limited is the #1 Indian exporter in 7 products including Carboplatin, Paclitaxel, Oxaliplatin. Top exports include Carboplatin ($27.5M), Paclitaxel ($25.1M), Oxaliplatin ($16.6M).
Fresenius Kabi Oncology Limited — Export Portfolio & Destination Treemap
Who is Fresenius Kabi Oncology Limited? — Company Overview & Market Position
Fresenius Kabi Oncology Limited, established in 2003, is a wholly owned subsidiary of Fresenius Kabi, a global healthcare company specializing in lifesaving medicines and technologies for infusion, transfusion, and clinical nutrition. The company is headquartered in Gurugram, Haryana, India, with its registered office located at B-310, Som Datt Chambers-1, Bhikaji Cama Place, New Delhi-110066, India. The Corporate Identification Number (CIN) is U24231DL2003PLC119441. The company employs approximately 1,152 individuals. For more information, visit their official website at www.fresenius-kabi-oncology.com.
What Does Fresenius Kabi Oncology Limited Export? — Product Portfolio Analysis
Fresenius Kabi Oncology Limited Therapeutic Categories — 3 Specializations
Fresenius Kabi Oncology Limited operates across 3 therapeutic categories, with Oncology (73.4%), Advanced Oncology (26.0%), Immunosuppressants (0.7%) representing the largest segments by export value. The portfolio is concentrated — the top 5 products account for 76% of total exports.
Oncology
8 products · 73.4% · $86.0M
Advanced Oncology
7 products · 26.0% · $30.4M
Immunosuppressants
1 products · 0.7% · $771.5K
Product Portfolio — Top 16 by Export Value
Fresenius Kabi Oncology Limited exports 16 pharmaceutical products across 3 therapeutic categories. Market leader (#1 exporter) in 7 products.
| # | Product | Category | Export Value | Shipments | Market Share | Rank |
|---|---|---|---|---|---|---|
| 1 | Carboplatin | Oncology | $27.5M | 735 | 38.8% | 1 |
| 2 | Paclitaxel | Oncology | $25.1M | 854 | 30.2% | 1 |
| 3 | Oxaliplatin | Advanced Oncology | $16.6M | 602 | 35.7% | 1 |
| 4 | Irinotecan | Oncology | $10.4M | 670 | 25.4% | 2 |
| 5 | Pemetrexed | Advanced Oncology | $9.0M | 333 | 31.1% | 1 |
| 6 | Docetaxel | Oncology | $8.4M | 356 | 21.1% | 1 |
| 7 | Gemcitabine | Oncology | $6.9M | 339 | 14.8% | 1 |
| 8 | Doxorubicin | Oncology | $3.1M | 143 | 4.6% | 3 |
| 9 | Cisplatin | Oncology | $2.8M | 60 | 21.5% | 1 |
| 10 | Etoposide | Oncology | $1.9M | 126 | 17.3% | 2 |
| 11 | Cytarabine | Advanced Oncology | $1.8M | 98 | 16.9% | 2 |
| 12 | Epirubicin | Advanced Oncology | $1.1M | 42 | 16.9% | 3 |
| 13 | Bortezomib | Advanced Oncology | $803.7K | 99 | 3.7% | 5 |
| 14 | Methotrexate | Immunosuppressants | $771.5K | 101 | 2.2% | 8 |
| 15 | Busulfan | Advanced Oncology | $627.6K | 77 | 12.7% | 3 |
| 16 | Vinorelbine | Advanced Oncology | $512.3K | 27 | 15.1% | 3 |
Fresenius Kabi Oncology Limited exports 16 pharmaceutical products across 3 therapeutic categories with a total export value of $117.2M. The company is the #1 Indian exporter in 7 products: Carboplatin, Paclitaxel, Oxaliplatin, Pemetrexed, Docetaxel and 2 more. The top category is Oncology (73.4% of portfolio), followed by Advanced Oncology (26.0%), indicating a concentrated portfolio with the top 5 products accounting for 75.6% of total export value.
Key Metrics
Top Categories
Top Export Routes
Related Analysis
Need Detailed Data?
Shipment-level records, pricing & buyer contacts for Fresenius Kabi Oncology Limited.
Request DemoFresenius Kabi Oncology Limited — Corporate Profile & Information
Headquarters, ownership, manufacturing facilities, and key leadership
1Corporate Overview
Fresenius Kabi Oncology Limited, established in 2003, is a wholly owned subsidiary of Fresenius Kabi, a global healthcare company specializing in lifesaving medicines and technologies for infusion, transfusion, and clinical nutrition. The company is headquartered in Gurugram, Haryana, India, with its registered office located at B-310, Som Datt Chambers-1, Bhikaji Cama Place, New Delhi-110066, India. The Corporate Identification Number (CIN) is U24231DL2003PLC119441. The company employs approximately 1,152 individuals. For more information, visit their official website at www.fresenius-kabi-oncology.com.
2Manufacturing Facilities
Fresenius Kabi Oncology Limited operates two primary manufacturing facilities in India. The Baddi plant in Himachal Pradesh specializes in the production of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The Kalyani facility in West Bengal focuses on the manufacturing of Active Pharmaceutical Ingredients (APIs) and intermediates. Both facilities adhere to stringent quality management systems and comply with current Good Manufacturing Practices (cGMP) to ensure the safety and efficacy of their products.
3Key Leadership
The leadership team at Fresenius Kabi Oncology Limited includes:
- Mr. Eduardo Ricardo Santos Montanha: Non-Executive Chairman
- Mr. Nikhil Kulshreshtha: Head of Legal, Compliance Officer, Company Secretary, and Whole-Time Director since 2007
- Mr. Michael Schonhofen: Non-Executive Director since 2008
- Mr. Rakesh Bhargava: Non-Executive Director since 2008
- Mr. Rajiv Lochan Jain: Non-Executive Independent Director since 2013
- Mr. Karsten Peter Lerch: Non-Executive Director since 2018
- Mr. Steffen Georg Roser: Non-Executive Director
- Ms. Heike Susanne Van Dawen: Non-Executive Director
- Mr. Maximilian Alexander Boehmer: Non-Executive Director
- Mr. Phaisal Sirisinsuk: Director of Dabur Pharma (Thailand) Company Ltd since 2003
These leaders bring a wealth of experience and expertise to the company's strategic direction and operations.
Where Does Fresenius Kabi Oncology Limited Export? — Destination Intelligence
Regulated market presence, emerging market penetration, and geographic strategy
1Regulated Markets
Fresenius Kabi Oncology Limited has established a significant presence in several regulated markets, including the United States, European Union, United Kingdom, Australia, and Japan. The company has obtained approvals for its finished pharmaceutical formulations from various regulatory bodies, ensuring compliance with international standards. In the United States, the company has faced regulatory scrutiny, with warning letters issued by the FDA in December 2017 for facilities in Kalyani and Baddi. These letters highlighted violations of current Good Manufacturing Practices (cGMP), leading to corrective actions and improvements in manufacturing processes. Despite these challenges, Fresenius Kabi Oncology Limited continues to supply products to the U.S. market, demonstrating resilience and commitment to quality.
2Emerging Markets
Fresenius Kabi Oncology Limited has expanded its reach into emerging markets across Africa, Latin America, and Southeast Asia. The company's commitment to quality is endorsed by regulatory authorities such as the Therapeutic Goods Administration (TGA) in Australia, the European Directorate for the Quality of Medicines & HealthCare (EDQM), and the U.S. Food and Drug Administration (FDA). These certifications facilitate access to various markets, enabling the company to provide affordable and effective oncology treatments to a broader patient population.
3Geographic Strategy
Fresenius Kabi Oncology Limited's geographic strategy involves a balanced approach to market diversification and risk management. While the company has a strong presence in regulated markets, it continues to expand into emerging markets, leveraging its quality certifications to gain market access. This strategic direction aims to mitigate concentration risks and enhance the company's global footprint, ensuring sustainable growth and resilience in the competitive pharmaceutical industry.
Fresenius Kabi Oncology Limited — Manufacturing & Regulatory Profile
FDA status, WHO-GMP certification, EU GMP compliance, and regulatory track record
1FDA Status
Fresenius Kabi Oncology Limited's facilities have been subject to inspections by the U.S. Food and Drug Administration (FDA). In May 2017, the FDA inspected the Kalyani facility in West Bengal, leading to a warning letter issued in December 2017. Similarly, the Baddi facility in Himachal Pradesh was inspected in April 2017, resulting in another warning letter in December 2017. These inspections identified violations of current Good Manufacturing Practices (cGMP), prompting the company to implement corrective actions to address the issues raised. Despite these challenges, Fresenius Kabi Oncology Limited continues to supply products to the U.S. market, demonstrating its commitment to compliance and quality.
2WHO & EU GMP
Fresenius Kabi Oncology Limited's manufacturing facilities adhere to stringent quality management systems and comply with current Good Manufacturing Practices (cGMP) as endorsed by regulatory authorities such as the Therapeutic Goods Administration (TGA) in Australia, the European Directorate for the Quality of Medicines & HealthCare (EDQM), and the U.S. Food and Drug Administration (FDA). These certifications facilitate access to various markets, enabling the company to provide affordable and effective oncology treatments to a broader patient population.
3CDSCO & Indian Regulatory
Fresenius Kabi Oncology Limited operates under the regulatory framework of the Central Drugs Standard Control Organisation (CDSCO) in India. The company's manufacturing facilities hold the necessary licenses and approvals from the CDSCO, ensuring compliance with national standards for pharmaceutical manufacturing. Additionally, the company obtains export No Objection Certificates (NOCs) from the Directorate General of Foreign Trade (DGFT) to facilitate international trade. These regulatory compliances underscore the company's commitment to maintaining high-quality manufacturing practices and adhering to both domestic and international standards.
4Recent Regulatory Actions
In December 2017, the U.S. Food and Drug Administration (FDA) issued warning letters to Fresenius Kabi Oncology Limited for its facilities in Kalyani and Baddi, India. The warning letters cited violations of current Good Manufacturing Practices (cGMP), including inadequate investigations into unexplained discrepancies and sterility failures. The company has since implemented corrective actions to address these issues and has continued to supply products to the U.S. market, demonstrating its commitment to compliance and quality.
Fresenius Kabi Oncology Limited — Competitive Positioning Among Indian Pharma Exporters
Market share comparison, key differentiators, and strategic analysis
1Competitive Landscape
Fresenius Kabi Oncology Limited operates in a competitive landscape, with several key players in the oncology generics segment. The company's top competitors include multinational pharmaceutical companies and specialized oncology firms that offer similar product portfolios. Fresenius Kabi Oncology Limited distinguishes itself through its comprehensive product range, including injectables, oral cytotoxics, cytostatics, intermediates, and Active Pharmaceutical Ingredients (APIs). The company's commitment to quality, as endorsed by regulatory authorities such as the Therapeutic Goods Administration (TGA), European Directorate for the Quality of Medicines & HealthCare (EDQM), and U.S. Food and Drug Administration (FDA), further strengthens its position in the market.
2Key Differentiators
Fresenius Kabi Oncology Limited's key differentiators include its comprehensive product portfolio, which spans injectables, oral cytotoxics, cytostatics, intermediates, and Active Pharmaceutical Ingredients (APIs). The company's commitment to quality is demonstrated through adherence to current Good Manufacturing Practices (cGMP) and certifications from regulatory authorities such as the Therapeutic Goods Administration (TGA), European Directorate for the Quality of Medicines & HealthCare (EDQM), and U.S. Food and Drug Administration (FDA). Additionally, the company's global reach and focus on emerging markets enable it to provide affordable and effective oncology treatments to a broad patient population.
3Strategic Position
Fresenius Kabi Oncology Limited's current strategic direction focuses on expanding its presence in both regulated and emerging markets. The company's product portfolio includes generics, specialty drugs, and biosimilars, with a strong emphasis on oncology treatments. The company continues to invest in research and development to enhance its product offerings and maintain a competitive edge in the market. Future outlooks suggest a continued focus on quality, compliance, and global expansion to meet the growing demand for affordable and effective cancer therapies.
Buyer Due Diligence Brief — Evaluating Fresenius Kabi Oncology Limited as a Supplier
Track record assessment, certifications to verify, and pre-order checks
1Supplier Assessment
Fresenius Kabi Oncology Limited has a track record of supplying high-quality pharmaceutical products globally. The company's export
Frequently Asked Questions — Fresenius Kabi Oncology Limited
How many pharmaceutical products does Fresenius Kabi Oncology Limited export from India?
Fresenius Kabi Oncology Limited exports 16 pharmaceutical products across 3 therapeutic categories. The top exports are Carboplatin ($27.5M), Paclitaxel ($25.1M), Oxaliplatin ($16.6M), Irinotecan ($10.4M), Pemetrexed ($9.0M). Total export value is $117.2M.
What is Fresenius Kabi Oncology Limited's total pharmaceutical export value?
Fresenius Kabi Oncology Limited's total pharmaceutical export value is $117.2M, based on 4,662 verified shipments recorded in Indian Customs (DGFT) data.
In which products is Fresenius Kabi Oncology Limited the #1 Indian exporter?
Fresenius Kabi Oncology Limited is the #1 Indian exporter in 7 products: Carboplatin (38.8% market share), Paclitaxel (30.2% market share), Oxaliplatin (35.7% market share), Pemetrexed (31.1% market share), Docetaxel (21.1% market share), Gemcitabine (14.8% market share), Cisplatin (21.5% market share).
What therapeutic categories does Fresenius Kabi Oncology Limited cover?
Fresenius Kabi Oncology Limited exports across 3 therapeutic categories. The largest are Oncology (73.4%, 8 products), Advanced Oncology (26.0%, 7 products), Immunosuppressants (0.7%, 1 products).
Get Full Fresenius Kabi Oncology Limited Export Intelligence
Access shipment-level details, pricing data, buyer connections, product breakdown, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify manufacturer licensing, GMP certification, and export registration with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Matching: Fresenius Kabi Oncology Limited identified across shipments using name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 3.Market Share: Calculated per product as Fresenius Kabi Oncology Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 4,662 individual customs records matching Fresenius Kabi Oncology Limited.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
16 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports per product category. For current shipment-level data, contact TransData Nexus.