Concord Biotech Limited
Indian Pharmaceutical Exporter · Immunosuppressants Specialist · $6.9M Total Trade · DGFT Verified
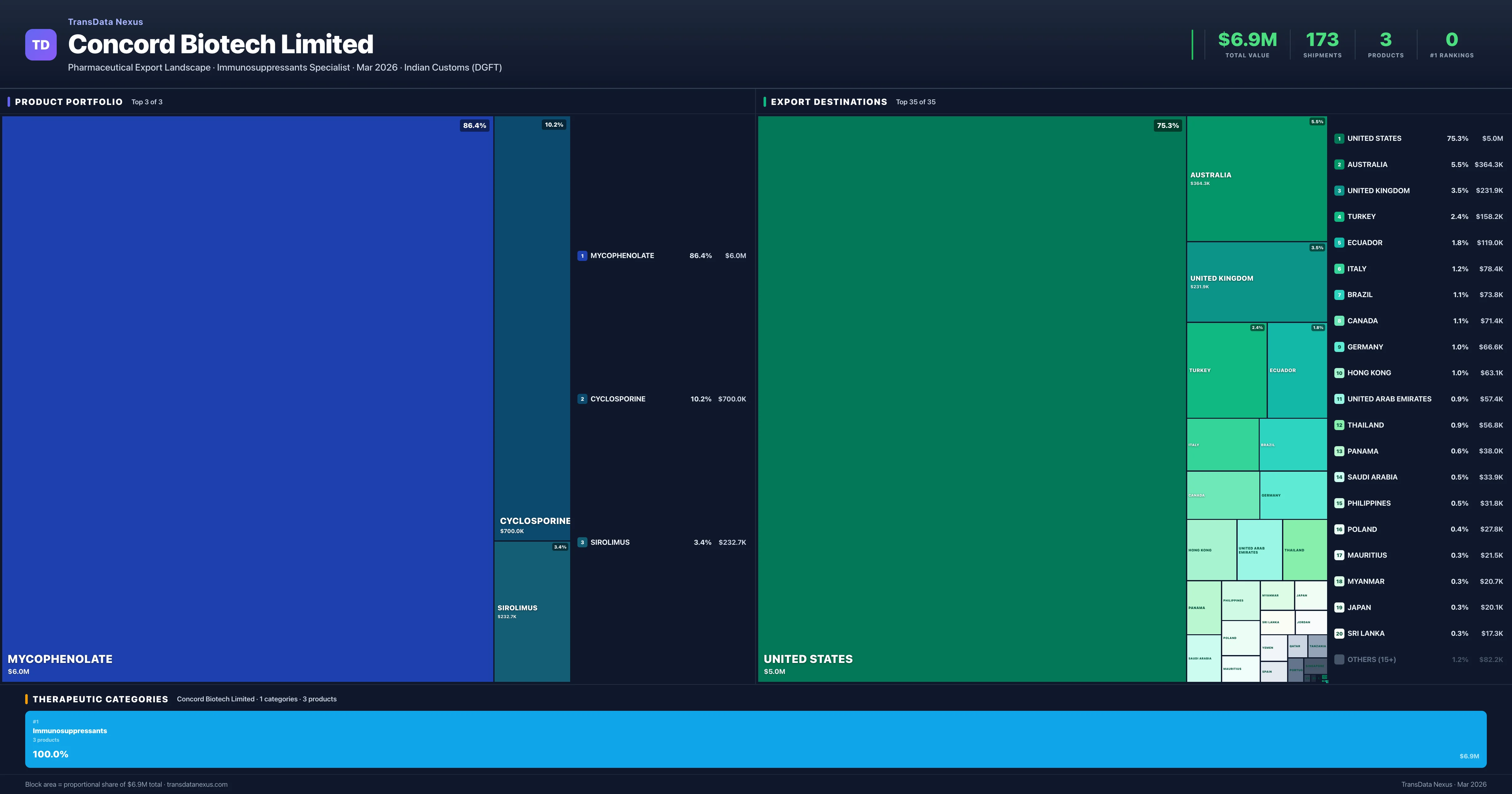
Concord Biotech Limited is an Indian pharmaceutical exporter with a total trade value of $6.9M across 3 products in 1 therapeutic categories. Based on 173 verified export shipments from Indian Customs (DGFT) records, the company actively competes across multiple product segments. Top exports include Mycophenolate ($6.0M), Cyclosporine ($700.0K), Sirolimus ($232.7K).
Concord Biotech Limited — Export Portfolio & Destination Treemap
Who is Concord Biotech Limited? — Company Overview & Market Position
Concord Biotech Limited, established in 2000, is a leading Indian biopharmaceutical company specializing in the research, development, and manufacturing of fermentation-based Active Pharmaceutical Ingredients (APIs) and finished formulations. Headquartered in Ahmedabad, Gujarat, India, the company has expanded its global footprint, serving over 70 countries with a diverse product portfolio.
The company operates with a vertically integrated business model, encompassing API production, formulation development, and contract research and manufacturing services. This integration enables Concord Biotech to maintain stringent quality control and meet the evolving demands of the global pharmaceutical market.
What Does Concord Biotech Limited Export? — Product Portfolio Analysis
Top Products by Export Value
Concord Biotech Limited Therapeutic Categories — 1 Specializations
Concord Biotech Limited operates across 1 therapeutic categories, with Immunosuppressants (100.0%), representing the largest segments by export value. The portfolio is concentrated — the top 5 products account for 100% of total exports.
Immunosuppressants
3 products · 100.0% · $6.9M
Product Portfolio — Top 3 by Export Value
Concord Biotech Limited exports 3 pharmaceutical products across 1 therapeutic categories.
| # | Product | Category | Export Value | Shipments | Market Share | Rank |
|---|---|---|---|---|---|---|
| 1 | Mycophenolate | Immunosuppressants | $6.0M | 119 | 1.8% | 6 |
| 2 | Cyclosporine | Immunosuppressants | $700.0K | 14 | 5.8% | 2 |
| 3 | Sirolimus | Immunosuppressants | $232.7K | 40 | 1.2% | 9 |
Concord Biotech Limited exports 3 pharmaceutical products across 1 therapeutic categories with a total export value of $6.9M. The top category is Immunosuppressants (100.0% of portfolio), indicating a concentrated portfolio with the top 5 products accounting for 100.0% of total export value.
Key Metrics
Top Categories
Top Export Routes
Related Analysis
Need Detailed Data?
Shipment-level records, pricing & buyer contacts for Concord Biotech Limited.
Request DemoConcord Biotech Limited — Corporate Profile & Information
Headquarters, ownership, manufacturing facilities, and key leadership
1Corporate Overview
Concord Biotech Limited, established in 2000, is a leading Indian biopharmaceutical company specializing in the research, development, and manufacturing of fermentation-based Active Pharmaceutical Ingredients (APIs) and finished formulations. Headquartered in Ahmedabad, Gujarat, India, the company has expanded its global footprint, serving over 70 countries with a diverse product portfolio.
The company operates with a vertically integrated business model, encompassing API production, formulation development, and contract research and manufacturing services. This integration enables Concord Biotech to maintain stringent quality control and meet the evolving demands of the global pharmaceutical market.
2Manufacturing Facilities
Concord Biotech Limited operates three state-of-the-art manufacturing facilities near Ahmedabad, Gujarat, India, each designed to meet international quality standards.
The Dholka facility, established in 2000, spans 112,302 square meters and comprises 22 manufacturing blocks. It specializes in the production of fermentation and semi-synthetic APIs, with a fermentation capacity of 450m³. This facility has been inspected and approved by global regulatory agencies, including the USFDA, EU GMP, Japanese PMDA, and ANVISA of Brazil.
In 2021, Concord Biotech commissioned the Limbasi facility, covering 596,309 square meters with 19 manufacturing blocks, including six fermentation blocks and 13 downstream processing blocks. This facility boasts a fermentation capacity of 800m³ and is equipped to handle complex fermentation and downstream processing, catering to major regulated markets.
The Valthera facility focuses on the production of finished formulations, including oral solids, oral liquids, and suspensions. It is designed to meet global regulatory standards such as USFDA and EU GMP, targeting both domestic and international markets.
3Key Leadership
Concord Biotech Limited is led by a team of seasoned professionals committed to driving the company's growth and innovation.
Mr. Sudhir Vaid serves as the Chairman and Managing Director. With a background in microbiology and extensive experience in the pharmaceutical industry, he has been instrumental in transforming Concord Biotech into a global leader in fermentation-based biopharmaceuticals.
Mr. Ankur Vaid, the Joint Managing Director and Chief Executive Officer, holds a B.Tech in Chemical Engineering and an MBA. With over 18 years in the pharmaceutical industry, he has been pivotal in leading key initiatives in research, development, and market strategy, contributing significantly to the company's success.
Where Does Concord Biotech Limited Export? — Destination Intelligence
Regulated market presence, emerging market penetration, and geographic strategy
1Regulated Markets
Concord Biotech Limited has established a strong presence in several regulated markets, including the United States, European Union, United Kingdom, Australia, and Japan, through rigorous regulatory filings and approvals.
In the United States, the company's Dholka facility has undergone multiple inspections by the USFDA, resulting in successful approvals for the manufacturing of various APIs. This facility is also registered with the FDA, allowing Concord Biotech to market its products in the U.S. market.
In the European Union, the Dholka facility has been inspected and approved by the EU GMP, Government of Upper Bavaria, ensuring compliance with European manufacturing standards. This approval facilitates the export of Concord Biotech's products to EU member countries.
In Japan, the company has received Drug Master File (DMF) registrations for Tacrolimus Hydrate from the Pharmaceuticals and Medical Devices Agency (PMDA), enabling the marketing of its products in the Japanese market.
Concord Biotech's commitment to quality and compliance has also led to approvals in other regulated markets, including Australia and the United Kingdom, further solidifying its global footprint.
2Emerging Markets
Concord Biotech Limited has strategically expanded its presence in emerging markets across Africa, Latin America, and Southeast Asia, leveraging WHO prequalification to enhance market access.
The company's finished formulations, including oral solids, liquids, and suspensions, are marketed under its own brands in India and distributed across emerging markets in Asia, Africa, Latin America, and the United States through established partnerships.
By obtaining WHO prequalification, Concord Biotech has facilitated the entry of its products into these markets, ensuring compliance with international quality standards and enhancing the company's reputation as a reliable supplier of pharmaceutical products.
3Geographic Strategy
Concord Biotech Limited's geographic strategy demonstrates a balanced approach to market diversification and risk management.
The company's significant presence in over 70 countries, including both regulated and emerging markets, indicates a well-diversified portfolio that mitigates concentration risks. This global footprint allows Concord Biotech to leverage growth opportunities across different regions while maintaining a stable revenue stream.
Strategically, Concord Biotech continues to focus on expanding its reach in emerging markets, capitalizing on the increasing demand for quality pharmaceutical products. Simultaneously, the company maintains a strong foothold in regulated markets, ensuring compliance with stringent regulatory standards and reinforcing its reputation as a trusted pharmaceutical manufacturer.
Concord Biotech Limited — Manufacturing & Regulatory Profile
FDA status, WHO-GMP certification, EU GMP compliance, and regulatory track record
1FDA Status
Concord Biotech Limited has a robust standing with the U.S. Food and Drug Administration (FDA), underscoring its commitment to quality and regulatory compliance.
The Dholka facility has been inspected multiple times by the USFDA, resulting in successful approvals for the manufacturing of various APIs. This facility is registered with the FDA, enabling Concord Biotech to market its products in the U.S. market.
The company's adherence to cGMP standards and its proactive approach to regulatory compliance have facilitated these approvals, allowing Concord Biotech to supply high-quality pharmaceutical products to the U.S. market.
2WHO & EU GMP
Concord Biotech Limited's commitment to quality is demonstrated through its compliance with WHO and EU GMP standards.
The Dholka facility has been inspected and approved by the EU GMP, Government of Upper Bavaria, ensuring compliance with European manufacturing standards. This approval facilitates the export of Concord Biotech's products to EU member countries.
Additionally, the Valthera formulation facility has received inspections from the WHO for GMP, further validating the company's adherence to international quality standards.
3CDSCO & Indian Regulatory
Concord Biotech Limited operates in full compliance with the Central Drugs Standard Control Organisation (CDSCO) and other Indian regulatory authorities.
The company's manufacturing facilities hold the necessary licenses and approvals from the CDSCO, ensuring adherence to national standards for pharmaceutical manufacturing. Concord Biotech also maintains state drug controller approvals and export No Objection Certificates (NOCs), facilitating the export of its products to various international markets.
4Recent Regulatory Actions
As of March 28, 2026, there have been no publicly reported Form 483 observations, warning letters, or import alerts issued to Concord Biotech Limited by the U.S. Food and Drug Administration (FDA). The company's manufacturing facilities have consistently met regulatory standards, reflecting its commitment to quality and compliance.
Concord Biotech Limited — Competitive Positioning Among Indian Pharma Exporters
Market share comparison, key differentiators, and strategic analysis
1Competitive Landscape
Concord Biotech Limited operates in a competitive landscape, with several key players in the biopharmaceutical industry offering similar products.
In the immunosuppressant segment, competitors include companies like Novartis and Roche, which have established market presence and extensive product portfolios. In oncology, firms such as Pfizer and Merck are prominent, offering a range of oncology therapeutics.
Concord Biotech's focus on fermentation-based APIs and high-quality formulations positions it as a specialized player in the market. The company's commitment to quality, regulatory compliance, and a diversified product portfolio enables it to maintain a competitive edge in the industry.
2Key Differentiators
Concord Biotech Limited distinguishes itself through several key differentiators:
- Specialized Manufacturing Capabilities: The company's expertise in fermentation and semi-synthetic processes allows for the production of complex APIs and formulations, catering to niche therapeutic segments.
- Regulatory Compliance: Concord Biotech's facilities are inspected and approved by multiple global regulatory agencies, including the USFDA, EU GMP, and WHO, ensuring adherence to international quality standards.
- Global Presence: With a footprint in over 70 countries, the company has established a robust distribution network, enabling it to serve diverse markets effectively.
Frequently Asked Questions — Concord Biotech Limited
How many pharmaceutical products does Concord Biotech Limited export from India?
Concord Biotech Limited exports 3 pharmaceutical products across 1 therapeutic categories. The top exports are Mycophenolate ($6.0M), Cyclosporine ($700.0K), Sirolimus ($232.7K). Total export value is $6.9M.
What is Concord Biotech Limited's total pharmaceutical export value?
Concord Biotech Limited's total pharmaceutical export value is $6.9M, based on 173 verified shipments recorded in Indian Customs (DGFT) data.
What therapeutic categories does Concord Biotech Limited cover?
Concord Biotech Limited exports across 1 therapeutic categories. The largest are Immunosuppressants (100.0%, 3 products).
Get Full Concord Biotech Limited Export Intelligence
Access shipment-level details, pricing data, buyer connections, product breakdown, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify manufacturer licensing, GMP certification, and export registration with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Matching: Concord Biotech Limited identified across shipments using name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 3.Market Share: Calculated per product as Concord Biotech Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 173 individual customs records matching Concord Biotech Limited.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports per product category. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.