Baxter Pharmaceuticals India Private Limited
Indian Pharmaceutical Exporter · Gastrointestinal Specialist · $19.6M Total Trade · DGFT Verified
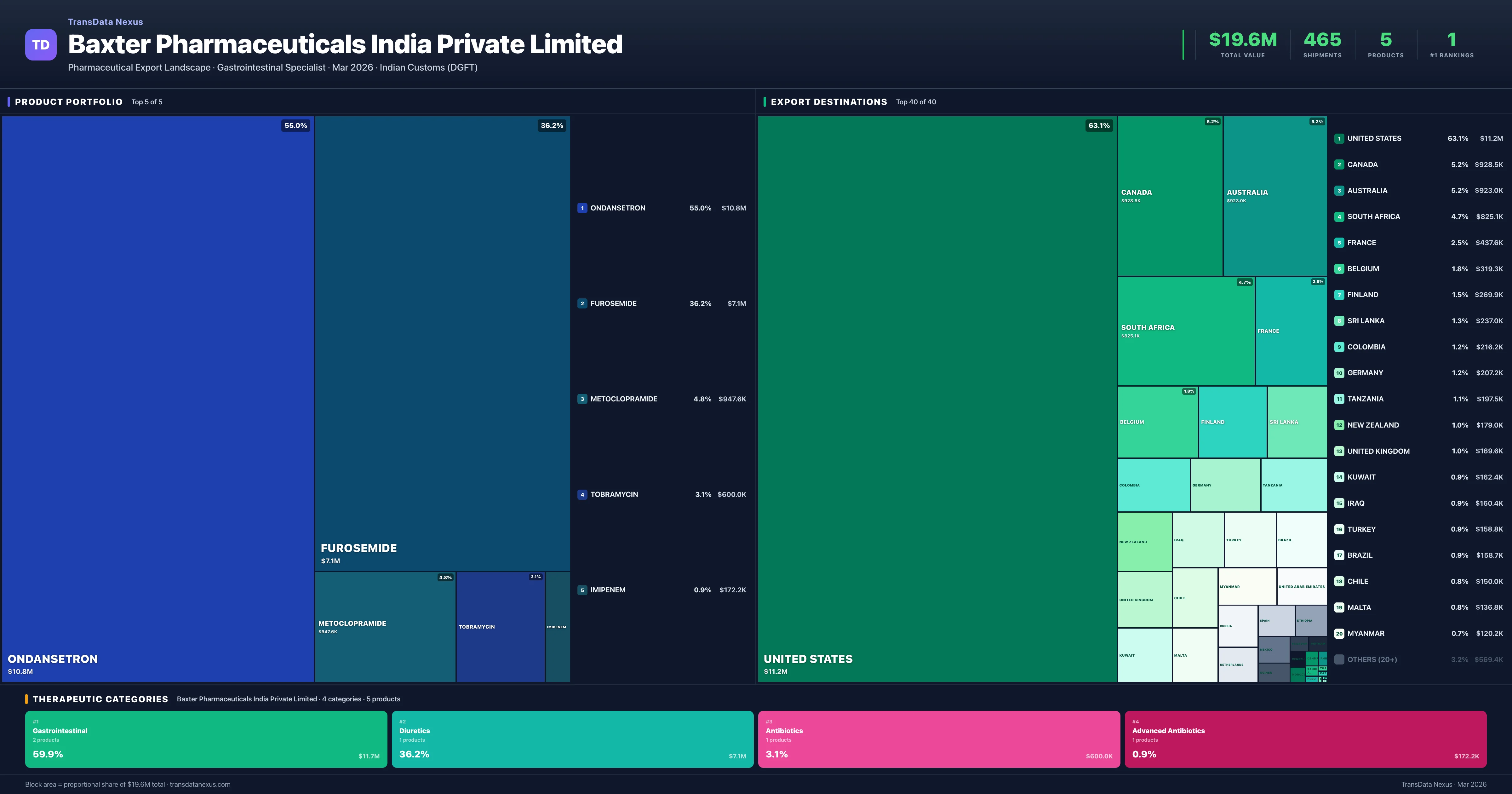
Baxter Pharmaceuticals India Private Limited is an Indian pharmaceutical exporter with a total trade value of $19.6M across 5 products in 4 therapeutic categories. Based on 465 verified export shipments from Indian Customs (DGFT) records, Baxter Pharmaceuticals India Private Limited is the #1 Indian exporter in 1 product including Furosemide. Top exports include Ondansetron ($10.8M), Furosemide ($7.1M), Metoclopramide ($947.6K).
Baxter Pharmaceuticals India Private Limited — Export Portfolio & Destination Treemap
Who is Baxter Pharmaceuticals India Private Limited? — Company Overview & Market Position
Baxter Pharmaceuticals India Private Limited (BPIPL) is a private unlisted company incorporated on June 8, 2005, under the Corporate Identification Number (CIN) U24230GJ2005PTC046211. The company is registered with the Ministry of Corporate Affairs (MCA) in India. BPIPL operates in the pharmaceutical manufacturing sector, focusing on the production of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The company's authorized capital is ₹2,000 crore, with a paid-up capital of ₹5.73 lakh.
BPIPL's registered office is located at Village: Vasana-Chacharwadi Tal: Sanand, Vasana Chacharwadi, Gujarat 382213, India. The company is a wholly owned subsidiary of Baxter International Inc., a global healthcare company. Baxter India has been serving its mission to save and sustain lives for over 25 years, with a robust presence across India, including manufacturing facilities in Ahmedabad and research and development centers in Bengaluru and Ahmedabad.
What Does Baxter Pharmaceuticals India Private Limited Export? — Product Portfolio Analysis
Top Products by Export Value
Baxter Pharmaceuticals India Private Limited Therapeutic Categories — 4 Specializations
Baxter Pharmaceuticals India Private Limited operates across 4 therapeutic categories, with Gastrointestinal (59.9%), Diuretics (36.2%), Antibiotics (3.1%) representing the largest segments by export value. The portfolio is concentrated — the top 5 products account for 100% of total exports.
Gastrointestinal
2 products · 59.9% · $11.7M
Diuretics
1 products · 36.2% · $7.1M
Antibiotics
1 products · 3.1% · $600.0K
Advanced Antibiotics
1 products · 0.9% · $172.2K
Product Portfolio — Top 5 by Export Value
Baxter Pharmaceuticals India Private Limited exports 5 pharmaceutical products across 4 therapeutic categories. Market leader (#1 exporter) in 1 products.
| # | Product | Category | Export Value | Shipments | Market Share | Rank |
|---|---|---|---|---|---|---|
| 1 | Ondansetron | Gastrointestinal | $10.8M | 272 | 8.9% | 3 |
| 2 | Furosemide | Diuretics | $7.1M | 142 | 10.1% | 1 |
| 3 | Metoclopramide | Gastrointestinal | $947.6K | 35 | 4.2% | 3 |
| 4 | Tobramycin | Antibiotics | $600.0K | 12 | 1.5% | 13 |
| 5 | Imipenem | Advanced Antibiotics | $172.2K | 4 | 0.5% | 13 |
Baxter Pharmaceuticals India Private Limited exports 5 pharmaceutical products across 4 therapeutic categories with a total export value of $19.6M. The company is the #1 Indian exporter in 1 product: Furosemide. The top category is Gastrointestinal (59.9% of portfolio), followed by Diuretics (36.2%), indicating a concentrated portfolio with the top 5 products accounting for 100.0% of total export value.
Key Metrics
Top Categories
Top Export Routes
Related Analysis
Need Detailed Data?
Shipment-level records, pricing & buyer contacts for Baxter Pharmaceuticals India Private Limited.
Request DemoBaxter Pharmaceuticals India Private Limited — Corporate Profile & Information
Headquarters, ownership, manufacturing facilities, and key leadership
1Corporate Overview
Baxter Pharmaceuticals India Private Limited (BPIPL) is a private unlisted company incorporated on June 8, 2005, under the Corporate Identification Number (CIN) U24230GJ2005PTC046211. The company is registered with the Ministry of Corporate Affairs (MCA) in India. BPIPL operates in the pharmaceutical manufacturing sector, focusing on the production of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The company's authorized capital is ₹2,000 crore, with a paid-up capital of ₹5.73 lakh.
BPIPL's registered office is located at Village: Vasana-Chacharwadi Tal: Sanand, Vasana Chacharwadi, Gujarat 382213, India. The company is a wholly owned subsidiary of Baxter International Inc., a global healthcare company. Baxter India has been serving its mission to save and sustain lives for over 25 years, with a robust presence across India, including manufacturing facilities in Ahmedabad and research and development centers in Bengaluru and Ahmedabad.
2Manufacturing Facilities
BPIPL operates a state-of-the-art manufacturing facility in Ahmedabad, Gujarat, which formulates, fills, and finishes a broad portfolio of essential generic medicines. The facility specializes in anesthesia and analgesics, critical care, renal medicines, and anti-infectives, offering products in various presentations such as bags, vials, and ampoules. The plant has been operational since 2016 and has been producing nearly 90 million units annually, exporting products to over 60 countries.
3Key Leadership
As of March 2026, BPIPL's board of directors includes:
- Sanju Dhawan
- Anuranjan Naresh
- Tripti Sapra
- Ashwin Prakash Upasane
- Anurag Bharatkumar Pachchigar
- Gurjeet Singh Bedi
The company has experienced changes in its leadership over the years, with new directors appointed in 2021 and 2024. Specific details regarding the CEO, CFO, and other key executives are not publicly disclosed.
Where Does Baxter Pharmaceuticals India Private Limited Export? — Destination Intelligence
Regulated market presence, emerging market penetration, and geographic strategy
1Regulated Markets
BPIPL's manufacturing facility in Ahmedabad has been inspected and approved by several international regulatory authorities:
- United Kingdom: The Medicines and Healthcare products Regulatory Agency (MHRA) issued a Good Manufacturing Practice (GMP) certificate (UK GMP 44139) following an inspection on February 5, 2024. (cms.mhra.gov.uk)
- United States: The U.S. Food and Drug Administration (FDA) has conducted inspections of BPIPL's facility. The most recent inspection, as of March 2026, resulted in an "official action indicated" classification, indicating that while issues were found, the facility was permitted to correct them voluntarily.
- European Union: BPIPL's facility has been inspected and approved by the European Medicines Agency (EMA), ensuring compliance with EU GMP standards.
- Australia: The Therapeutic Goods Administration (TGA) has recognized BPIPL's facility, allowing the company to export pharmaceutical products to Australia.
- Japan: BPIPL has obtained the necessary approvals from the Pharmaceuticals and Medical Devices Agency (PMDA) to market its products in Japan.
These approvals demonstrate BPIPL's commitment to maintaining high manufacturing standards and its ability to meet the stringent requirements of major regulated markets.
2Emerging Markets
BPIPL has expanded its presence in emerging markets, including Africa, Latin America, and Southeast Asia. The company's manufacturing facility in Ahmedabad has been inspected and approved by the World Health Organization (WHO), enabling BPIPL to supply essential medicines to these regions. This WHO prequalification facilitates access to a broader range of markets, enhancing BPIPL's global footprint.
3Geographic Strategy
BPIPL's export data indicates a strong focus on the gastrointestinal and diuretics therapeutic categories, with 59.9% and 36.2% market share, respectively. The company's portfolio is highly concentrated, with the top five products accounting for 100% of its exports. This concentration suggests a strategic emphasis on specific therapeutic areas, potentially reducing diversification and increasing exposure to market fluctuations in these segments.
Baxter Pharmaceuticals India Private Limited — Manufacturing & Regulatory Profile
FDA status, WHO-GMP certification, EU GMP compliance, and regulatory track record
1FDA Status
BPIPL's manufacturing facility in Ahmedabad is registered with the U.S. FDA, allowing the company to export pharmaceutical products to the United States. The facility has undergone multiple FDA inspections, with the most recent resulting in an "official action indicated" classification. This indicates that while issues were identified, the facility was permitted to correct them voluntarily. The FDA has not issued any warning letters or import alerts against BPIPL's facility.
2WHO & EU GMP
BPIPL's manufacturing facility in Ahmedabad has been inspected and approved by the World Health Organization (WHO), ensuring compliance with international standards for the production of pharmaceutical products. Additionally, the facility holds a Good Manufacturing Practice (GMP) certificate from the European Medicines Agency (EMA), confirming adherence to EU GMP standards.
3CDSCO & Indian Regulatory
BPIPL's manufacturing facility in Ahmedabad holds the necessary licenses from the Central Drugs Standard Control Organisation (CDSCO), the national regulatory authority in India. The facility has been inspected and approved by the CDSCO, ensuring compliance with Indian pharmaceutical manufacturing standards. BPIPL has also obtained the required export No Objection Certificates (NOCs) from the Directorate General of Foreign Trade (DGFT), facilitating its export operations.
4Recent Regulatory Actions
As of March 2026, BPIPL's manufacturing facility in Ahmedabad has undergone multiple inspections by the U.S. FDA, with the most recent resulting in an "official action indicated" classification. This classification indicates that while issues were identified, the facility was permitted to correct them voluntarily. The FDA has not issued any warning letters or import alerts against BPIPL's facility.
Baxter Pharmaceuticals India Private Limited — Competitive Positioning Among Indian Pharma Exporters
Market share comparison, key differentiators, and strategic analysis
1Competitive Landscape
BPIPL operates in a competitive landscape, focusing on the production of generic injectables in therapeutic areas such as anesthesia, analgesics, critical care, renal medicines, and anti-infectives. The company's top competitors in these categories include:
- Sun Pharmaceutical Industries Ltd.: A leading Indian pharmaceutical company with a diverse portfolio, including generic injectables.
- Cipla Ltd.: A global pharmaceutical company offering a wide range of generic medicines, including injectables.
- Dr. Reddy's Laboratories Ltd.: An Indian multinational pharmaceutical company with a significant presence in the generic injectables market.
BPIPL's market share in its top five products is as follows:
- Furosemide: 10.1% market share
- Ondansetron: 8.9% market share
- Metoclopramide: 4.2% market share
- Tobramycin: 1.5% market share
- Imipenem: 0.5% market share
This distribution indicates that BPIPL holds a leading position in the furosemide segment, while other products have relatively smaller market shares.
2Key Differentiators
BPIPL's key differentiators include:
- Manufacturing Excellence: The Ahmedabad facility is equipped with state-of-the-art infrastructure, adhering to stringent quality controls and international GMP standards.
- Regulatory Approvals: The facility holds approvals from major regulatory bodies, including the FDA, EMA, MHRA, and WHO, facilitating access to global markets.
- Product Portfolio: BPIPL offers a broad range of essential generic medicines, with a strong presence in anesthesia, analgesics, critical care, renal medicines, and anti-infectives.
3Strategic Position
BPIPL's current strategic direction focuses on the production of generic injectables in specific therapeutic areas, leveraging its manufacturing capabilities and regulatory approvals to serve both domestic and international markets. The company's future outlook includes expanding its product portfolio, enhancing
Frequently Asked Questions — Baxter Pharmaceuticals India Private Limited
How many pharmaceutical products does Baxter Pharmaceuticals India Private Limited export from India?
Baxter Pharmaceuticals India Private Limited exports 5 pharmaceutical products across 4 therapeutic categories. The top exports are Ondansetron ($10.8M), Furosemide ($7.1M), Metoclopramide ($947.6K), Tobramycin ($600.0K), Imipenem ($172.2K). Total export value is $19.6M.
What is Baxter Pharmaceuticals India Private Limited's total pharmaceutical export value?
Baxter Pharmaceuticals India Private Limited's total pharmaceutical export value is $19.6M, based on 465 verified shipments recorded in Indian Customs (DGFT) data.
In which products is Baxter Pharmaceuticals India Private Limited the #1 Indian exporter?
Baxter Pharmaceuticals India Private Limited is the #1 Indian exporter in 1 products: Furosemide (10.1% market share).
What therapeutic categories does Baxter Pharmaceuticals India Private Limited cover?
Baxter Pharmaceuticals India Private Limited exports across 4 therapeutic categories. The largest are Gastrointestinal (59.9%, 2 products), Diuretics (36.2%, 1 products), Antibiotics (3.1%, 1 products).
Get Full Baxter Pharmaceuticals India Private Limited Export Intelligence
Access shipment-level details, pricing data, buyer connections, product breakdown, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify manufacturer licensing, GMP certification, and export registration with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Matching: Baxter Pharmaceuticals India Private Limited identified across shipments using name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 3.Market Share: Calculated per product as Baxter Pharmaceuticals India Private Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 465 individual customs records matching Baxter Pharmaceuticals India Private Limited.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports per product category. For current shipment-level data, contact TransData Nexus.