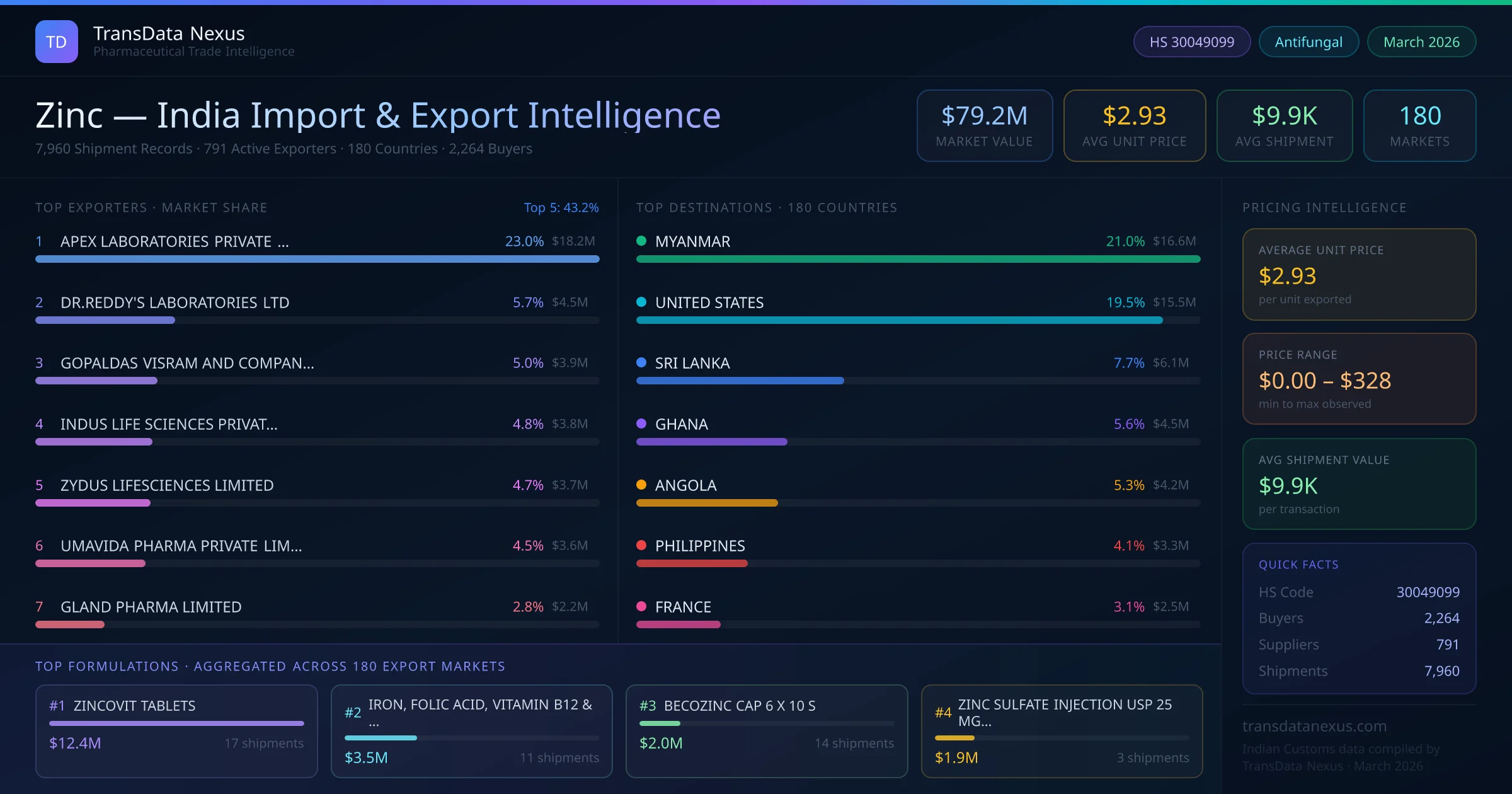
Importing finished pharmaceutical formulations containing zinc into India requires compliance with the Central Drugs Standard Control Organization (CDSCO) regulations. The importing company must obtain a valid Importer License from the Directorate General of Foreign Trade (DGFT). Additionally, the product must be registered with CDSCO, which involves submitting an application along with necessary documents such as the Certificate of Pharmaceutical Product (CoPP), Free Sale Certificate, and stability data. The registration process typically takes 6 to 12 months, depending on the completeness of the application and the product's compliance with Indian standards. For formulations under HS Code 30049099, specific requirements include demonstrating the product's safety, efficacy, and quality as per the Indian Pharmacopoeia standards. The importer must also ensure that the product complies with the Drugs and Cosmetics Act, 1940, and the rules thereunder.