Who Exports Warfarin from India — 135 Suppliers Behind a $7.4M Market
India's warfarin export market is supplied by 135 active exporters who collectively shipped $7.4M across 1,333 shipments. AMNEAL PHARMACEUTICALS PRIVATE LIMITED leads with a 38.6% market share, followed by CIPLA LIMITED and STRIDES PHARMA SCIENCE LIMITED. The top 5 suppliers together control 81.2% of total export value, reflecting a concentrated market structure.
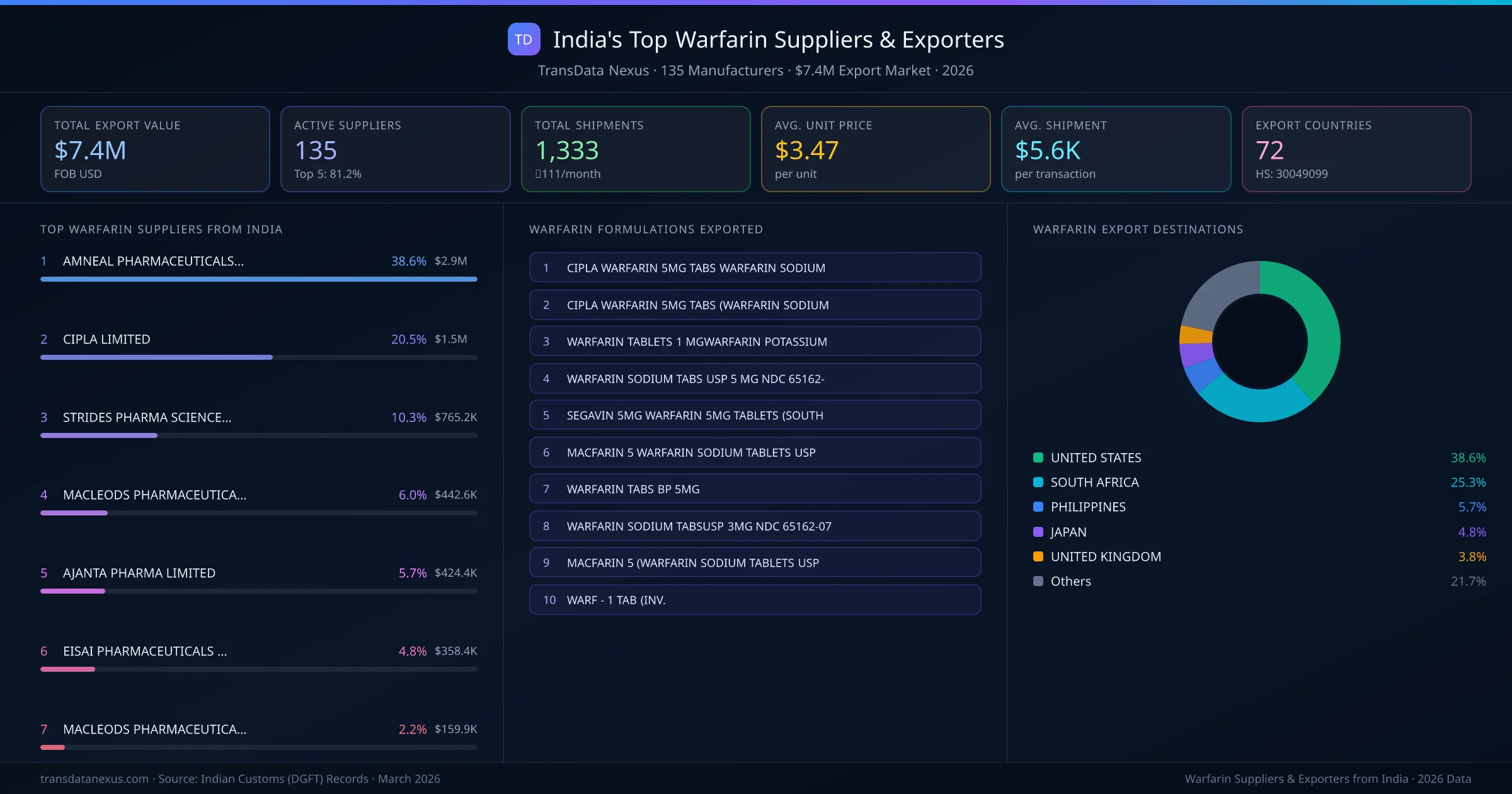
Top Warfarin Exporters from India — Ranked by Export Value
AMNEAL PHARMACEUTICALS PRIVATE LIMITED is the leading warfarin exporter from India, holding a 38.6% share of the $7.4M market across 1,333 shipments from 135 exporters. The top 5 suppliers — AMNEAL PHARMACEUTICALS PRIVATE LIMITED, CIPLA LIMITED, STRIDES PHARMA SCIENCE LIMITED, MACLEODS PHARMACEUTICALS LTD, AJANTA PHARMA LIMITED — collectively control 81.2% of total export value, indicating a highly concentrated market. Individual shares are: AMNEAL PHARMACEUTICALS PRIVATE LIMITED (38.6%), CIPLA LIMITED (20.5%), STRIDES PHARMA SCIENCE LIMITED (10.3%), MACLEODS PHARMACEUTICALS LTD (6.0%), AJANTA PHARMA LIMITED (5.7%).
Top Warfarin Exporters from India
Ranked by export value · 135 active suppliers · Indian Customs (DGFT) data
| # | Supplier & Formulations | Value | Ctry. | Share |
|---|---|---|---|---|
| 1 | AMNEAL PHARMACEUTICALS PRIVATE LIMITED WARFARIN SODIUM TABS USP 5 MG NDC 65162- | $2.9M | 1 | 38.6% |
| 2 | CIPLA LIMITED CIPLA WARFARIN 5MG TABS WARFARIN SODIUMCIPLA WARFARIN 5MG TABS (WARFARIN SODIUMSEGAVIN 5MG WARFARIN 5MG TABLETS (SOUTH | $1.5M | 3 | 20.5% |
| 3 | STRIDES PHARMA SCIENCE LIMITED CIPLA WARFARIN 5MG TABS WARFARIN SODIUMCIPLA WARFARIN 5MG TABS (WARFARIN SODIUMSEGAVIN 5MG WARFARIN 5MG TABLETS (SOUTH | $765.2K | 2 | 10.3% |
| 4 | MACLEODS PHARMACEUTICALS LTD CIPLA WARFARIN 5MG TABS WARFARIN SODIUMCIPLA WARFARIN 5MG TABS (WARFARIN SODIUMSEGAVIN 5MG WARFARIN 5MG TABLETS (SOUTH | $442.6K | 6 | 6.0% |
| 5 | AJANTA PHARMA LIMITED | $424.4K | 1 | 5.7% |
| 6 | MACLEODS PHARMACEUTICALS LIMITED CIPLA WARFARIN 5MG TABS WARFARIN SODIUMCIPLA WARFARIN 5MG TABS (WARFARIN SODIUMSEGAVIN 5MG WARFARIN 5MG TABLETS (SOUTH | $159.9K | 3 | 2.2% |
| 7 | CHANDRA BHAGAT PHARMA LIMITED | $139.3K | 1 | 1.9% |
| 8 | 3S CORPORATION | $103.0K | 4 | 1.4% |
| 9 | MYLAN LABORATORIES LIMITED | $95.4K | 1 | 1.3% |
| 10 | GENO PHARMACEUTICALS PRIVATE LIMITED WARFARIN TABS BP 5MGWARFARIN TABS 5MGWARFARIN 5MG TABLETS 28S | $90.9K | 1 | 1.2% |
| 11 | IPCA LABORATORIES LIMITED CIPLA WARFARIN 5MG TABS WARFARIN SODIUMCIPLA WARFARIN 5MG TABS (WARFARIN SODIUMSEGAVIN 5MG WARFARIN 5MG TABLETS (SOUTH | $55.6K | 2 | 0.8% |
| 12 | UNICURE REMEDIES PRIVATE LIMITED | $45.5K | 2 | 0.6% |
| 13 | BRAWN LABORATORIES LIMITED | $29.7K | 2 | 0.4% |
Related Analysis
Supplier Certification & Compliance Matrix
FDA, WHO-GMP, and EU GMP status for top Warfarin exporters
| Supplier | US FDA | WHO-GMP | EU GMP | ANDAs | Notes |
|---|---|---|---|---|---|
| Amneal Pharmaceuticals Private Limited | Approved | Yes | Yes | 149 | Received first FDA approval in 2006; operates nine facilities in India as of 202 |
| Cipla Limited | Approved | Yes | Yes | 27 | Acquired InvaGen Pharmaceuticals in 2015; operates seven manufacturing facilitie |
| Strides Pharma Science Limited | Approved | Yes | Yes | Not specified | Manufactures softgel capsules, hard-gel capsules, tablets, and injectables; oper |
| Macleods Pharmaceuticals Limited | Approved | Yes | Yes | Not specified | Recognized for compliance with international regulatory standards; operates mult |
| Ajanta Pharma Limited | Approved | Yes | Yes | 27 | Operates seven manufacturing facilities in India; exports to over 30 countries. |
| Mylan Laboratories Limited | Approved | Yes | Yes | Not specified | Part of Mylan N.V., a global pharmaceutical company; operates multiple facilitie |
| Intas Pharmaceuticals Limited | Approved | Yes | Yes | Not specified | Operates 22 manufacturing plants globally; headquartered in Ahmedabad, India. |
| Alembic Pharmaceuticals Limited | Approved | Yes | Yes | Not specified | Received multiple FDA approvals between 2024 and 2025; operates manufacturing fa |
TransData Nexus reviewed the regulatory standing of 8 leading Warfarin exporters from India. 8 hold US FDA facility approvals, 8 maintain WHO-GMP certification, and 8 are EU GMP compliant. For regulated markets (US, EU, Australia), prioritise suppliers with active FDA or EU GMP approvals. For semi-regulated markets (Africa, ASEAN, Latin America), WHO-GMP certification is the minimum recommended standard.
Certification status compiled from publicly available regulatory databases including FDA Orange Book, WHO Prequalification database, and EMA GMP registry. Buyers should independently verify compliance status with the relevant regulatory authority before placing orders.
TransData Nexus Research · Mar 2026
Manufacturing Hub Analysis — Vendor Proximity
India's pharmaceutical clusters relevant to Warfarin sourcing
1Hyderabad — Bulk Drug Capital
Hyderabad, often referred to as the "Bulk Drug Capital of India," contributes over 40% of the nation's bulk drug production. The city hosts numerous pharmaceutical companies specializing in active pharmaceutical ingredients (APIs) and bulk drugs, supported by a robust infrastructure and a skilled workforce. The establishment of Hyderabad Pharma City further solidifies its position as a global pharmaceutical hub.
2Ahmedabad-Vadodara — Formulations Hub
The Ahmedabad-Vadodara corridor in Gujarat is renowned for its pharmaceutical formulations. Vadodara, in particular, is home to major pharmaceutical companies like Alembic Pharmaceuticals, which specializes in formulations for therapeutic areas such as cardiology and infectious diseases. The region's focus on formulations complements Hyderabad's bulk drug production, creating a comprehensive pharmaceutical supply chain.
3Mumbai-Thane-Raigad — Export Gateway
The Mumbai-Thane-Raigad belt serves as a critical export gateway for India's pharmaceutical industry. Mumbai's strategic location and well-developed port facilities facilitate efficient international distribution of pharmaceutical products. The region's infrastructure supports the export activities of companies like Amneal Pharmaceuticals Private Limited, the leading exporter of Warfarin.
4Baddi-Nalagarh — Tax Incentive Zone
The Baddi-Nalagarh region in Himachal Pradesh has emerged as a significant pharmaceutical manufacturing zone, driven by favorable tax incentives and supportive government policies. This area attracts numerous pharmaceutical companies seeking cost-effective production solutions, thereby enhancing India's overall pharmaceutical manufacturing capacity.
5Sourcing Recommendations
- Diversify Supplier Base: While Amneal Pharmaceuticals Private Limited is the leading exporter of Warfarin, consider engaging with other top exporters like Cipla Limited and Strides Pharma Science Limited to mitigate supply chain risks.
- Leverage Regional Strengths: Utilize Hyderabad's expertise in bulk drug production for sourcing APIs and Ahmedabad-Vadodara's capabilities in formulations to optimize the supply chain.
- Optimize Logistics: Capitalize on the Mumbai-Thane-Raigad region's export infrastructure to ensure efficient international distribution of Warfarin products.
- Explore Cost-Effective Manufacturing: Consider the Baddi-Nalagarh region for manufacturing opportunities, taking advantage of tax incentives and cost efficiencies.
By strategically engaging with these pharmaceutical clusters, TransData Nexus can enhance its Warfarin supply chain resilience and efficiency.
Recent M&A, Collaborations & Capacity Expansions
Industry developments among top Warfarin exporters from India
Amneal Pharmaceuticals — Amneal receives FDA approval for first generic Vyvanse
In September 2023, Amneal Pharmaceuticals received approval from the U.S. Food and Drug Administration (FDA) to produce the first generic version of Vyvanse, a medication used to treat ADHD. - IMPACT: This approval may enhance Amneal's market presence, potentially influencing its Warfarin export capabilities.
Impact: This approval may enhance Amneal's market presence, potentially influencing its Warfarin export capabilities.
Common Questions — Warfarin Suppliers from India
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
Q Which warfarin supplier from India is the most reliable for bulk orders?
Based on shipment frequency and export consistency, AMNEAL PHARMACEUTICALS PRIVATE LIMITED leads with 242 recorded shipments worth $2.9M. CIPLA LIMITED (74 shipments) and STRIDES PHARMA SCIENCE LIMITED (37 shipments) are also established high-volume exporters.
Q How many warfarin manufacturers are there in India?
India has 135 active warfarin exporters with a combined export market of $7.4M across 1,333 shipments to 72 countries. The top 5 suppliers hold 81.2% of total export value.
Q What certifications should I verify?
Verify: WHO-GMP certification (most markets), US FDA approval (United States), EU GMP certificate (EU/EEA), and Free Sale Certificate from CDSCO. Always request a Certificate of Analysis (CoA) and Certificate of Origin (CoO).
Q What is the typical price range for warfarin from India?
Average FOB unit price: $3.47 per unit, ranging from $0.01 to $95.02. Average shipment value: $5.6K.
Official References & Regulatory Resources
- CDSCO India
- Pharmexcil
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- India Trade Statistics (DGFT)
Verify manufacturer licensing and export certifications with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Ranking: 135 verified Indian exporters of Warfarin ranked by capped export value from DGFT shipping bill records.
- 2.Export Value Analysis: Total export value aggregated from 1,333 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Market Concentration: Supplier market share and geographic reach analyzed across 72 destination countries.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
1,333 Verified Shipments
135 exporters tracked
Expert-Reviewed
By pharmaceutical trade specialists