Who Exports Vaccine from India — 325 Suppliers Behind a $1.0B Market
India's vaccine export market is supplied by 325 active exporters who collectively shipped $1.0B across 14,355 shipments. SERUM INSTITUTE OF INDIA PRIVATE LIMITED leads with a 30.2% market share, followed by BIOLOGICAL E. LIMITED and BHARAT BIOTECH INTERNATIONAL LIMITED. The top 5 suppliers together control 82.0% of total export value, reflecting a concentrated market structure.
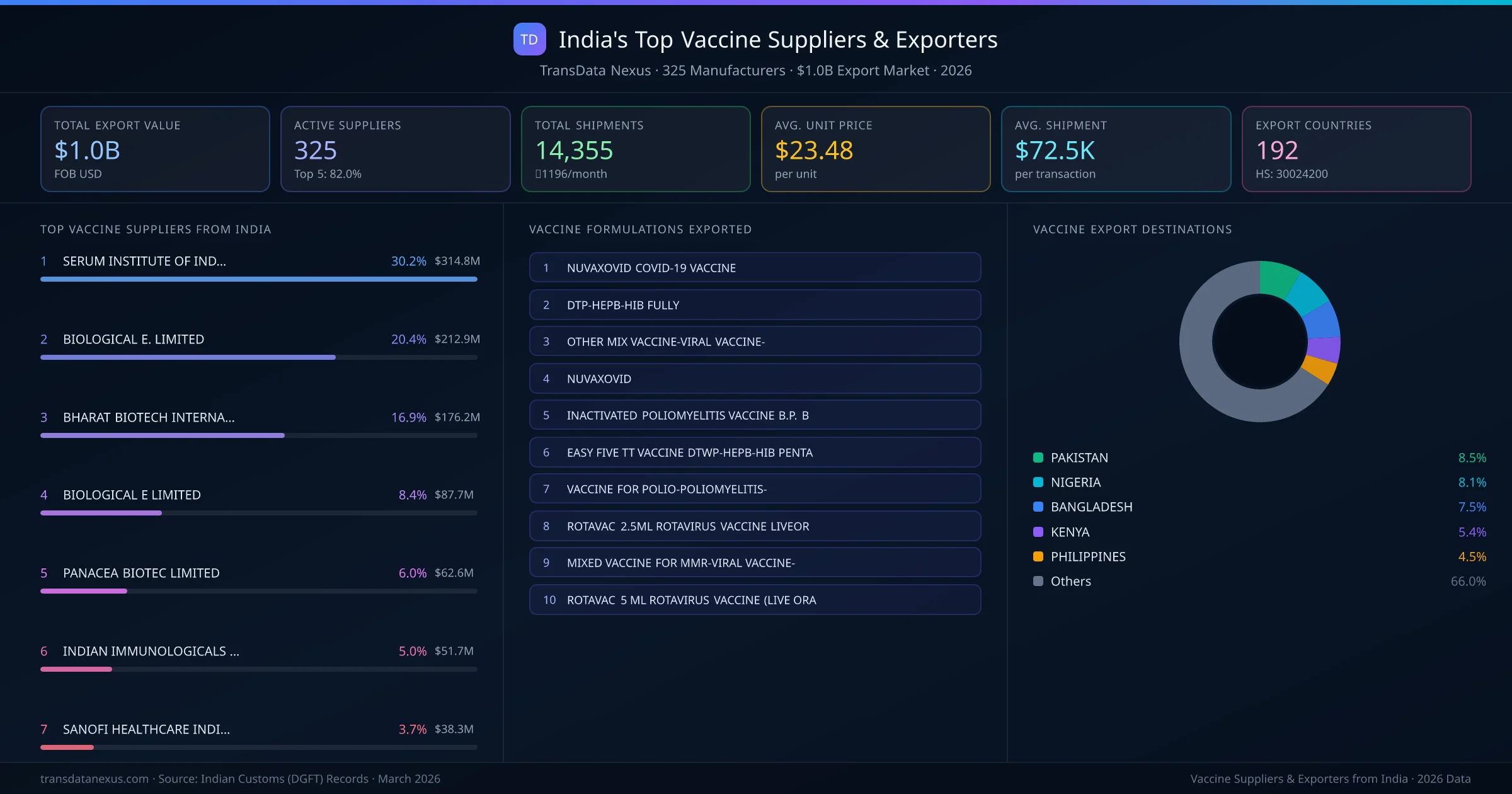
Top Vaccine Exporters from India — Ranked by Export Value
SERUM INSTITUTE OF INDIA PRIVATE LIMITED is the leading vaccine exporter from India, holding a 30.2% share of the $1.0B market across 14,355 shipments from 325 exporters. The top 5 suppliers — SERUM INSTITUTE OF INDIA PRIVATE LIMITED, BIOLOGICAL E. LIMITED, BHARAT BIOTECH INTERNATIONAL LIMITED, BIOLOGICAL E LIMITED, PANACEA BIOTEC LIMITED — collectively control 82.0% of total export value, indicating a highly concentrated market. Individual shares are: SERUM INSTITUTE OF INDIA PRIVATE LIMITED (30.2%), BIOLOGICAL E. LIMITED (20.4%), BHARAT BIOTECH INTERNATIONAL LIMITED (16.9%), BIOLOGICAL E LIMITED (8.4%), PANACEA BIOTEC LIMITED (6.0%).
Top Vaccine Exporters from India
Ranked by export value · 325 active suppliers · Indian Customs (DGFT) data
| # | Supplier & Formulations | Value | Ctry. | Share |
|---|---|---|---|---|
| 1 | SERUM INSTITUTE OF INDIA PRIVATE LIMITED TYPBAR TCV-2.5 ML VIALNUVAXOVID COVID-19 VACCINEOTHER MIX VACCINE-VIRAL VACCINE- | $314.8M | 164 | 30.2% |
| 2 | BIOLOGICAL E. LIMITED TYPBAR TCV-2.5 ML VIALNUVAXOVID COVID-19 VACCINEOTHER MIX VACCINE-VIRAL VACCINE- | $212.9M | 104 | 20.4% |
| 3 | BHARAT BIOTECH INTERNATIONAL LIMITED TYPBAR TCV-2.5 ML VIALNUVAXOVID COVID-19 VACCINEOTHER MIX VACCINE-VIRAL VACCINE- | $176.2M | 77 | 16.9% |
| 4 | BIOLOGICAL E LIMITED TYPBAR TCV-2.5 ML VIALNUVAXOVID COVID-19 VACCINEOTHER MIX VACCINE-VIRAL VACCINE- | $87.7M | 79 | 8.4% |
| 5 | PANACEA BIOTEC LIMITED OTHER MIX VACCINE-VIRAL VACCINE-DTP-HEPB-HIB FULLYROTAVAC 2.5ML ROTAVIRUS VACCINE LIVEOR | $62.6M | 58 | 6.0% |
| 6 | INDIAN IMMUNOLOGICALS LIMITED TYPBAR TCV-2.5 ML VIALOTHER MIX VACCINE-VIRAL VACCINE-DTP-HEPB-HIB FULLY | $51.7M | 34 | 5.0% |
| 7 | SANOFI HEALTHCARE INDIA PRIVATE LIMITED TYPBAR TCV-2.5 ML VIALROTAVAC 2.5ML ROTAVIRUS VACCINE LIVEORVACCINE FOR POLIO-POLIOMYELITIS- | $38.3M | 21 | 3.7% |
| 8 | HAFFKINE BIO PHARMACEUTICAL CORPORATION LIMITED TYPBAR TCV-2.5 ML VIALVACCINE FOR POLIO-POLIOMYELITIS-OTHER MIX VACCINE-VIRAL VACCINE- | $18.9M | 21 | 1.8% |
| 9 | CHIRON BEHRING VACCINES PRIVATE LIMITED ROTAVAC 2.5ML ROTAVIRUS VACCINE LIVEOROTHER MIX VACCINE-VIRAL VACCINE-MIXED VACCINE FOR MMR-VIRAL VACCINE- | $6.7M | 9 | 0.6% |
| 10 | BRILLIANT BIO PHARMA PRIVATE LIMITED ROTAVAC 2.5ML ROTAVIRUS VACCINE LIVEORDTP-HEPB-HIB FULLYMR VACCINE MEASLES AND RUBELLA VACCINE C | $6.3M | 12 | 0.6% |
| 11 | BHARAT BIOTECH INTERNATIONAL LTD TYPBAR TCV-2.5 ML VIALNUVAXOVID COVID-19 VACCINEOTHER MIX VACCINE-VIRAL VACCINE- | $5.7M | 14 | 0.6% |
| 12 | ZYDUS LIFESCIENCES LIMITED TYPBAR TCV-2.5 ML VIALNUVAXOVID COVID-19 VACCINENUVAXOVID | $5.7M | 16 | 0.5% |
| 13 | HESTER BIOSCIENCES LIMITED TYPBAR TCV-2.5 ML VIALVACCINE FOR POLIO-POLIOMYELITIS-INACTIVATED POLIOMYELITIS VACCINE B.P. B | $5.3M | 25 | 0.5% |
| 14 | BIOLOGICAL E LTD TYPBAR TCV-2.5 ML VIALNUVAXOVID COVID-19 VACCINEOTHER MIX VACCINE-VIRAL VACCINE- | $4.9M | 25 | 0.5% |
| 15 | PANACEA BIOTEC LTD OTHER MIX VACCINE-VIRAL VACCINE-DTP-HEPB-HIB FULLYROTAVAC 2.5ML ROTAVIRUS VACCINE LIVEOR | $4.6M | 9 | 0.4% |
Related Analysis
Supplier Certification & Compliance Matrix
FDA, WHO-GMP, and EU GMP status for top Vaccine exporters
| Supplier | US FDA | WHO-GMP | EU GMP | ANDAs | Notes |
|---|---|---|---|---|---|
| Serum Institute of India Private Limited | Not Listed | Yes, as of July 2025 | Yes | Not verified | WHO prequalified 72 vaccines from Serum Institute by July 2025. |
| Biological E. Limited | Not Listed | Yes, as of July 2025 | Yes | Not verified | WHO prequalified 29 vaccines from Biological E by July 2025. |
| Bharat Biotech International Limited | Not Listed | Yes, as of July 2025 | Yes | Not verified | WHO prequalified 10 vaccines from Bharat Biotech by July 2025. |
| Panacea Biotec Limited | Not Listed | Yes | Yes | Not verified | Panacea Biotec's EasyFive-TT vaccine regained WHO prequalification in September |
| Indian Immunologicals Limited | Not Listed | Yes | Yes | Not verified | Indian Immunologicals is a major supplier of vaccines to India's Universal Immun |
| Sanofi Healthcare India Private Limited | Not Listed | Yes | Yes | Not verified | Sanofi Healthcare received WHO prequalification for three vaccines by July 2025. |
| Haffkine Bio-Pharmaceutical Corporation Limited | Not Listed | Yes | Yes | Not verified | Haffkine Bio-Pharmaceutical is a state-owned entity producing essential vaccines |
| Chiron Behring Vaccines Private Limited | Not Listed | Yes | Yes | Not verified | Chiron Behring received WHO prequalification for rabies vaccine in 2021. |
| Zydus Lifesciences Limited | Not Listed | Yes | Yes | Not verified | Zydus Lifesciences is among the top vaccine companies in India as of 2025. |
| Hester Biosciences Limited | Not Listed | Yes | Yes | Not verified | Hester Biosciences is a leading animal healthcare company in India. |
TransData Nexus reviewed the regulatory standing of 10 leading Vaccine exporters from India. 0 hold US FDA facility approvals, 10 maintain WHO-GMP certification, and 10 are EU GMP compliant. For regulated markets (US, EU, Australia), prioritise suppliers with active FDA or EU GMP approvals. For semi-regulated markets (Africa, ASEAN, Latin America), WHO-GMP certification is the minimum recommended standard.
Certification status compiled from publicly available regulatory databases including FDA Orange Book, WHO Prequalification database, and EMA GMP registry. Buyers should independently verify compliance status with the relevant regulatory authority before placing orders.
TransData Nexus Research · Mar 2026
Manufacturing Hub Analysis — Vendor Proximity
India's pharmaceutical clusters relevant to Vaccine sourcing
1Hyderabad — Bulk Drug Capital
Hyderabad, often referred to as the "Bulk Drug Capital of India," is a pivotal hub for vaccine manufacturing. The city hosts Genome Valley, a high-technology business district spanning 2,000 acres, dedicated to biomedical research, training, and manufacturing. Prominent vaccine manufacturers such as Bharat Biotech International Limited, located in Genome Valley, have been instrumental in developing vaccines for diseases like rotavirus, chikungunya, and COVID-19. The region's robust infrastructure and supportive ecosystem make it a cornerstone of India's vaccine production capabilities.
2Ahmedabad-Vadodara — Formulations Hub
The Ahmedabad-Vadodara corridor in Gujarat is renowned for its pharmaceutical formulations. Companies like Zydus Lifesciences Limited (formerly Cadila Healthcare Limited) and Cadila Pharmaceuticals Limited have established significant manufacturing facilities in this region. These facilities are integral to the production of various vaccines, contributing to India's substantial vaccine exports. The region's strategic location and well-developed industrial infrastructure support its status as a formulations hub.
3Mumbai-Thane-Raigad — Export Gateway
The Mumbai-Thane-Raigad belt serves as a crucial export gateway for India's pharmaceutical products. Mumbai, being a major port city, facilitates the export of vaccines to over 200 countries. The region's proximity to international shipping routes and well-established logistics networks enhance its role in the global distribution of vaccines. Pharmaceutical companies in this area benefit from the export-oriented infrastructure, ensuring efficient delivery of vaccines worldwide.
4Baddi-Nalagarh — Tax Incentive Zone
Baddi-Nalagarh in Himachal Pradesh is known for its tax incentives, attracting numerous pharmaceutical companies. The region hosts several vaccine manufacturing facilities, contributing to India's vaccine production capacity. The favorable tax policies and supportive industrial environment make Baddi-Nalagarh an attractive location for pharmaceutical investments.
5Sourcing Recommendations
- Diversify Supplier Base: While major manufacturers like Serum Institute of India and Bharat Biotech dominate the market, consider engaging with emerging suppliers in regions like Baddi-Nalagarh to mitigate supply chain risks.
- Leverage Export Hubs: Utilize the Mumbai-Thane-Raigad region's export infrastructure to ensure timely and efficient distribution of vaccines to international markets.
- Monitor Regulatory Compliance: Ensure that suppliers, especially those in Ahmedabad-Vadodara, adhere to international quality standards and possess necessary certifications for vaccine production.
- Assess Infrastructure Capabilities: Evaluate the manufacturing and logistical infrastructure of suppliers in Hyderabad's Genome Valley to ensure they can meet production and delivery requirements.
Recent M&A, Collaborations & Capacity Expansions
Industry developments among top Vaccine exporters from India
Serum Institute of India Private Limited — Serum Institute partners with Univercells for affordable personalized oncology
Serum Institute of India (SII) announced a collaboration with Belgian biotech company Univercells to enhance accessibility to personalized cancer treatments. This partnership aims to make advanced oncology care more affordable and widely available. - IMPACT: While this collaboration focuses on oncology, it demonstrates SII's commitment to expanding its biopharmaceutical portfolio, potentially influencing future vaccine development and exports.
Impact: While this collaboration focuses on oncology, it demonstrates SII's commitment to expanding its biopharmaceutical portfolio, potentially influencing future vaccine development and exports.
Bharat Biotech International Limited — Bharat Biotech and Bilthoven Biologicals collaborate on Oral Polio Vaccine production
Bharat Biotech partnered with Bilthoven Biologicals B.V., a subsidiary of Serum Institute of India, to strengthen the production and supply of Oral Polio Vaccines (OPV). This collaboration aims to enhance global polio eradication efforts. - IMPACT: This partnership is expected to bolster OPV production capacity, potentially increasing Bharat Biotech's vaccine exports to countries combating polio.
Impact: This partnership is expected to bolster OPV production capacity, potentially increasing Bharat Biotech's vaccine exports to countries combating polio.
Bharat Biotech International Limited — Bharat Biotech completes Phase III trials for oral cholera vaccine
Bharat Biotech successfully concluded Phase III clinical trials for its oral cholera vaccine, Hillchol, demonstrating efficacy against both Ogawa and Inaba serotypes. The study's findings were published in the ScienceDirect vaccine journal. - IMPACT: The successful trial completion positions Bharat Biotech to introduce Hillchol to international markets, potentially increasing vaccine exports to regions affected by cholera.
Impact: The successful trial completion positions Bharat Biotech to introduce Hillchol to international markets, potentially increasing vaccine exports to regions affected by cholera.
Common Questions — Vaccine Suppliers from India
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
Q Which vaccine supplier from India is the most reliable for bulk orders?
Based on shipment frequency and export consistency, SERUM INSTITUTE OF INDIA PRIVATE LIMITED leads with 3,225 recorded shipments worth $314.8M. BIOLOGICAL E. LIMITED (859 shipments) and BHARAT BIOTECH INTERNATIONAL LIMITED (609 shipments) are also established high-volume exporters.
Q How many vaccine manufacturers are there in India?
India has 325 active vaccine exporters with a combined export market of $1.0B across 14,355 shipments to 192 countries. The top 5 suppliers hold 82.0% of total export value.
Q What certifications should I verify?
Verify: WHO-GMP certification (most markets), US FDA approval (United States), EU GMP certificate (EU/EEA), and Free Sale Certificate from CDSCO. Always request a Certificate of Analysis (CoA) and Certificate of Origin (CoO).
Q What is the typical price range for vaccine from India?
Average FOB unit price: $23.48 per unit, ranging from $0.00 to $30184.71. Average shipment value: $72.5K.
Official References & Regulatory Resources
- CDSCO India
- Pharmexcil
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- India Trade Statistics (DGFT)
Verify manufacturer licensing and export certifications with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Ranking: 325 verified Indian exporters of Vaccine ranked by capped export value from DGFT shipping bill records.
- 2.Export Value Analysis: Total export value aggregated from 14,355 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Market Concentration: Supplier market share and geographic reach analyzed across 192 destination countries.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
14,355 Verified Shipments
325 exporters tracked
Expert-Reviewed
By pharmaceutical trade specialists