United States to India Pharmaceutical Import
Bilateral Trade Intelligence · $2.7B Total Trade · 689 Foreign Suppliers · 371 Indian Buyers · DGFT Verified · Updated March 2026
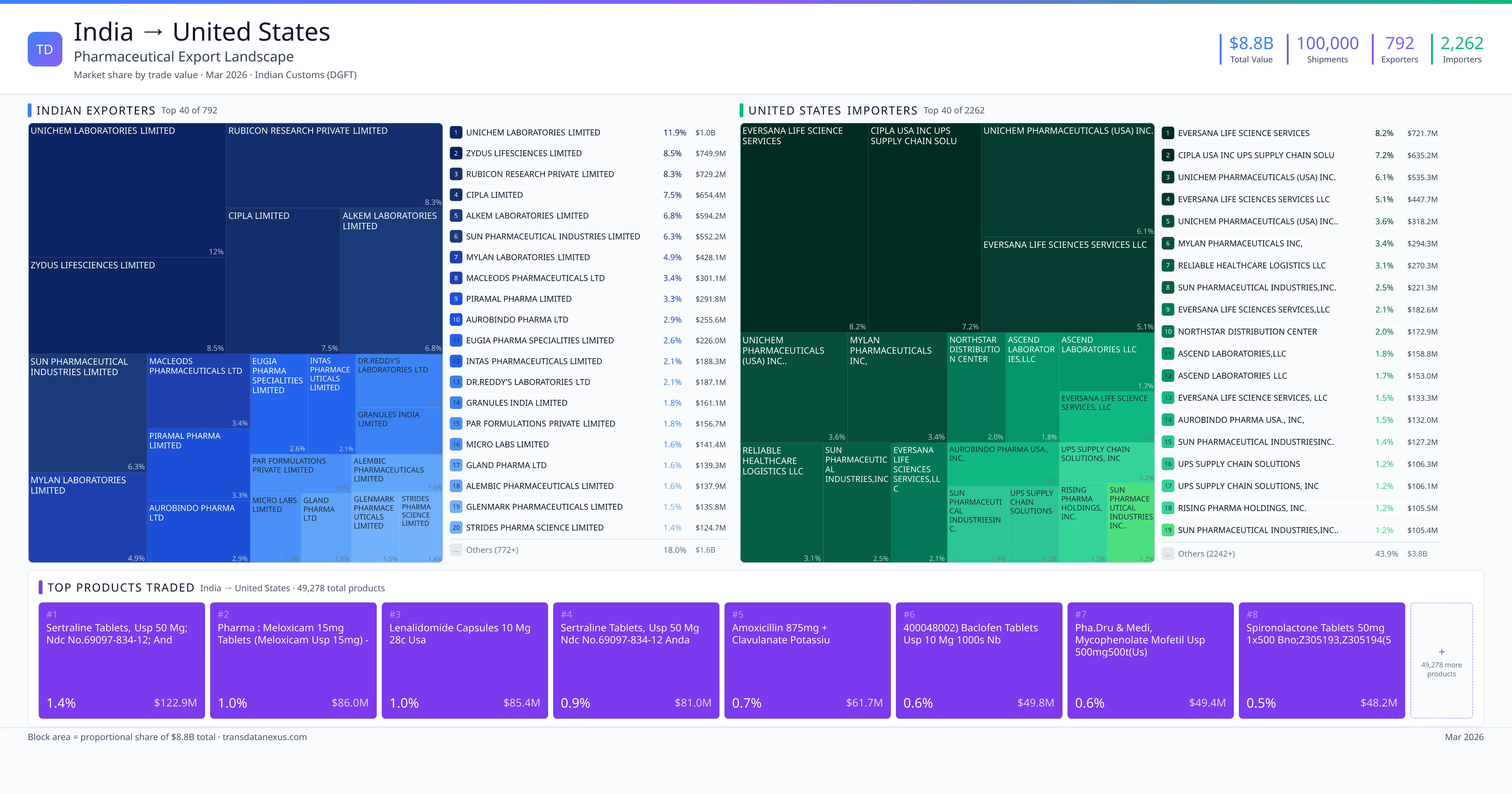
India imported $2.7B worth of pharmaceutical formulations from United States across 27,607 verified shipments, from 689 foreign suppliers to 371 Indian buyers. The top suppliers are Takeda Pharmaceuticals International AG ($48.5M) and SWORDS LABORATORIES UNLIMITED COMPANY ($45.0M). The leading products are Vaccine ($957.0M) and Serum ($774.4M). Average shipment value: $99.3K.
Top Pharmaceutical Formulations — United States to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Vaccine | $957.0M | 34.9% |
| 2 | Serum | $774.4M | 28.3% |
| 3 | Plasma | $538.7M | 19.7% |
| 4 | Dapagliflozin | $92.0M | 3.4% |
| 5 | Fluticasone | $54.3M | 2.0% |
| 6 | Nivolumab | $41.5M | 1.5% |
| 7 | Immunoglobulin | $40.0M | 1.5% |
| 8 | Sodium | $29.0M | 1.1% |
| 9 | Metformin | $18.9M | 0.7% |
| 10 | Moxifloxacin | $17.0M | 0.6% |
| 11 | Metoprolol | $15.7M | 0.6% |
| 12 | Heparin | $14.8M | 0.5% |
| 13 | Diclofenac | $13.4M | 0.5% |
| 14 | Prednisolone | $9.3M | 0.3% |
| 15 | Sitagliptin | $7.3M | 0.3% |
India imports 20+ pharmaceutical formulations from United States with a combined trade value of $2.7B. Key products include Vaccine ($957.0M), Serum ($774.4M), Plasma ($538.7M), Dapagliflozin ($92.0M), Fluticasone ($54.3M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 27,607 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — United States to India Trade Routes
These are the top pharmaceutical products exported from India to United States, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, United States buyers, regulatory requirements, and logistics for that specific product corridor. Products include Vaccine ($957.0M), Serum ($774.4M), Plasma ($538.7M), Dapagliflozin ($92.0M), Fluticasone ($54.3M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top United States Suppliers to India
689 Indian pharmaceutical companies export finished formulations to United States. Leading exporters include Takeda Pharmaceuticals International Ag, Swords Laboratories Unlimited Company, Boehringer Ingelheim Vetmedica Gmbh, Intervet International B.v.. The top exporter accounts for 1.8% of total India–United States pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from United States
371 companies in United States import pharmaceutical formulations from India. Top buyers include Invitrogen Bioservices India Private Limited, Takeda Biopharmaceuticals India Private Limited, Astrazeneca Pharma India Limited, Bristol-myers Squibb India Private Limited. The largest buyer accounts for 2.7% of India–United States pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
India–United States Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade corridor between the United States and India has experienced significant growth over the past two decades. In 2005, the bilateral pharmaceutical trade was valued at approximately $500 million, with around 5,000 shipments. By 2025, this figure had escalated to $2.7 billion, encompassing 27,607 shipments. This expansion underscores the increasing interdependence of the two nations in the pharmaceutical sector.
Key milestones in this evolution include the establishment of the U.S.-India Trade Policy Forum in 2005, aimed at enhancing bilateral trade relations. The signing of the U.S.-India Civil Nuclear Agreement in 2008 also paved the way for deeper economic collaborations, indirectly benefiting sectors like pharmaceuticals. Additionally, India's participation in the World Trade Organization (WTO) since 1995 has facilitated smoother trade processes, including the import of pharmaceutical products from the U.S.
2United States's Role in India's Pharma Imports
The United States plays a pivotal role in India's pharmaceutical imports, accounting for a substantial share of the market. In 2025, the U.S. supplied a diverse range of pharmaceutical products to India, including vaccines, serums, and specialized drugs. Notably, vaccines alone constituted 34.9% of the total import value, highlighting the critical importance of immunological products in the bilateral trade.
India's importation of these products from the U.S. is driven by the need for advanced medical treatments and technologies not readily available domestically. The U.S. pharmaceutical industry is renowned for its innovation and high-quality standards, making it a preferred source for India's healthcare requirements.
3Recent Developments
Between 2024 and 2026, several developments have influenced the pharmaceutical trade between the U.S. and India. In March 2025, the Indian Pharmaceutical Alliance (IPA) advocated for the reduction of import duties on U.S. pharmaceutical products to zero, aiming to bolster the domestic industry by lowering costs.
Additionally, in September 2025, the U.S. imposed a 100% tariff on all branded or patented pharmaceutical imports, effective October 1, 2025, unless the manufacturer is actively building a production facility in the U.S. This policy shift has significant implications for Indian drugmakers, who rely heavily on the U.S. market for generic drugs.
United States Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1CDSCO Registration Process
For U.S. companies aiming to export pharmaceutical products to India, compliance with the Central Drugs Standard Control Organisation (CDSCO) regulations is mandatory. The process involves obtaining an import license under Form 10 of the Drugs and Cosmetics Rules, 1945, and registering the product with CDSCO under Form 41.
The registration process requires submission of comprehensive documentation, including a Certificate of Analysis (CoA) for each batch, a Good Manufacturing Practice (GMP) certificate from the exporting country, and an Importer Exporter Code (IEC) from the Directorate General of Foreign Trade (DGFT). Additionally, an authorization letter from the manufacturer or marketing authorization holder (MAH) is necessary, authorizing the Indian importer to import specific pharmaceutical products on their behalf.
2GMP & Manufacturing Standards
India mandates that pharmaceutical products imported from the U.S. be manufactured in facilities compliant with Good Manufacturing Practice (GMP) standards as prescribed by CDSCO. The manufacturing facilities must meet specific requirements concerning premises, plant, machinery, and quality control processes. Compliance with these standards ensures the safety, efficacy, and quality of pharmaceutical products available in the Indian market.
3Import Documentation
Importing pharmaceutical products into India necessitates several key documents:
- Import License (Form 10): Issued by CDSCO, this license authorizes the importation of specific pharmaceutical products.
- Registration Certificate (Form 41): Also issued by CDSCO, this certificate confirms that the imported product is registered for sale in India.
- No Objection Certificate (NOC): Required for certain products, this certificate indicates that the product does not violate any existing patents or trademarks in India.
- Test License: Permits the import or manufacture of drugs for testing, analysis, or clinical trial purposes.
- Customs Documentation: Includes the Bill of Entry and other customs declarations necessary for clearing goods through Indian customs.
Ensuring all documentation is accurate and complete is crucial for the smooth clearance of pharmaceutical imports.
Product Categories & Therapeutic Trends — India to United States
Dominant categories, emerging opportunities, and demand drivers
1Dominant Import Categories
The pharmaceutical imports from the United States to India are predominantly in the categories of vaccines, serums, and specialized drugs. Vaccines alone accounted for 34.9% of the total import value, underscoring their critical role in India's healthcare system. Serums and plasma products also constitute significant portions of the imports, reflecting the demand for advanced biological products.
Specialized drugs, including Dapagliflozin, Fluticasone, and Nivolumab, are imported to address specific therapeutic needs not met by domestic products. These imports are essential for treating complex health conditions and contribute to the advancement of medical treatments in India.
2Innovation & Specialty Imports
India's importation of innovator drugs and novel formulations from the United States is driven by the need for advanced therapies not manufactured domestically. Products like Dapagliflozin, Fluticasone, and Nivolumab are examples of specialized drugs imported to meet specific therapeutic requirements. These imports are crucial for addressing complex health conditions and advancing medical treatments in India.
3Import Demand Drivers
India's importation of pharmaceutical products from the United States is influenced by several factors:
- Patent Protection: The U.S. offers strong patent protection, ensuring that innovative drugs are available for import without generic competition.
- Technology Gaps: Certain advanced pharmaceutical technologies and formulations are not yet developed domestically, necessitating imports to meet healthcare needs.
- Quality Requirements: The U.S. pharmaceutical industry is renowned for its stringent quality standards, making its products highly desirable in the Indian market.
- Disease Burden: The prevalence of specific diseases in India creates a demand for specialized treatments that are often sourced from the U.S.
These factors collectively drive the sustained demand for U.S. pharmaceutical imports into India.
Trade Policy & Tariff Intelligence — India and United States
Tariff structure, trade agreements, IP and patent landscape
1India's Import Tariff Structure
India imposes a 10% import duty on pharmaceutical products imported from the United States. This duty is part of the Basic Customs Duty (BCD) and is applicable to a wide range of pharmaceutical products. Additionally, the Integrated Goods and Services Tax (IGST) and health cess may apply, depending on the specific product and its classification under the Harmonized System (HS) code.
In March 2025, the Indian Pharmaceutical Alliance (IPA) proposed reducing the import duty on U.S. pharmaceutical products to zero, aiming to support the domestic industry by lowering costs.
2Trade Agreements & Preferences
As of March 2026, there is no Free Trade Agreement (FTA) between India and the United States specifically addressing pharmaceutical products. However, both countries are members of the World Trade Organization (WTO), which facilitates trade through agreed-upon rules and regulations. Additionally, India has been involved in discussions regarding the Regional Comprehensive Economic Partnership (RCEP), which could influence future trade dynamics, including pharmaceutical imports.
3IP, Patents & Price Control
India's patent regime, particularly Section 3(d) of the Indian Patents Act, aims to prevent evergreening of patents by restricting the patentability of new forms of known substances unless they result in enhanced efficacy. This provision has implications for the importation of certain pharmaceutical products. The National Pharmaceutical Pricing Authority (NPPA) regulates the prices of essential medicines in India, which can affect the pricing of imported drugs. Compulsory licensing provisions allow the government to grant licenses to produce patented products without the consent of the patent holder under specific circumstances, such as public health emergencies.
Supply Chain & Logistics — India to United States Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical products imported from the United States to India are typically transported via major sea and air routes. Sea shipments often depart from ports like New York and Los Angeles, arriving at Indian ports such as Mumbai and Chennai. Air shipments are usually routed through major international airports, including John F. Kennedy International Airport (JFK) and Los Angeles International Airport (LAX), with arrivals at Indira Gandhi International Airport (DEL) in Delhi and Chhatrapati Shivaji Maharaj International Airport (BOM) in Mumbai.
FAQ — India to United States Pharmaceutical Trade
What is the total value of India's pharmaceutical export to United States?
India exported pharmaceuticals worth $2.7B to United States across 27,607 verified shipments.
Who are the top Indian pharmaceutical exporters to United States?
1. Takeda Pharmaceuticals International AG — $48.5M. 2. SWORDS LABORATORIES UNLIMITED COMPANY — $45.0M. 3. BOEHRINGER INGELHEIM VETMEDICA GMBH — $36.0M. Total: 689 suppliers.
Which companies in United States import pharmaceuticals from India?
1. INVITROGEN BIOSERVICES INDIA PRIVATE LIMITED — $74.9M. 2. TAKEDA BIOPHARMACEUTICALS INDIA PRIVATE LIMITED — $44.0M. 3. ASTRAZENECA PHARMA INDIA LIMITED — $43.9M. 371 buyers total.
What pharmaceutical products does India export most to United States?
1. Vaccine ($957.0M, 34.9%); 2. Serum ($774.4M, 28.3%); 3. Plasma ($538.7M, 19.7%); 4. Dapagliflozin ($92.0M, 3.4%); 5. Fluticasone ($54.3M, 2.0%)
Which ports handle pharmaceutical shipments from India to United States?
Export: . Import: .
Why does United States import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $2.7B corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply United States?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to United States pharmaceutical trade?
$99.3K per consignment across 27,607 shipments.
How many Indian pharmaceutical companies export to United States?
689 Indian companies. Largest: Takeda Pharmaceuticals International AG with $48.5M.
How can I find verified Indian pharmaceutical suppliers for United States?
TransData Nexus covers 689 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to United States Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 27,607 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to United States identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 27,607 verified shipments from 689 Indian exporters to 371 United States buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
689 Exporters
371 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists